- McLafferty Rearrangement
-
McLafferty Rearrangement
F. W. McLafferty, Anal. Chem. 31, 82 (1959).
Electron-impact-induced cleavage of carbonyl compounds having a hydrogen in the γ-position, to an enolic fragment and an olefin:
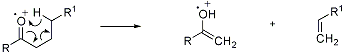
D. G. I. Kingston et al., Chem. Rev. 74, 215 (1974); K. Biemann, Mass Spectrometry (New York, 1962) p 119; Djerassi et al., J. Am. Chem. Soc. 87, 817 (1965); 91, 2069 (1969); 94, 473 (1972); M. J. Lacey et al., Org. Mass Spectrom. 5, 1391 (1971); G. Eadon, J. Am. Chem. Soc. 94, 8938 (1972); F. Turecek, V. Hanus, Org. Mass Spectrom. 15, 8 (1980). Cf. Norrish Type Cleavage.
Prev: McMurry Coupling Reaction
Next: McFadyen-Stevens Reaction - 【Back】【Close 】【Print】【Add to favorite 】
-
Health and Chemical more >
-
Hot Products
- 15207-30-4 GRAMICIDIN S HYDROCHLORIDE
- 99-96-7 4-Hydroxybenzoic acid
- 261760-24-1 1,1,1,2-Tetrafluoro-4-iodo-2-(trifluoromethyl)pentane
- 163702-01-0 Benzene, (1-methylethenyl)-, homopolymer, ar-(2-hydroxy-2-methyl-1-oxopropyl) derivs.
- 2380-27-0 METHYL 12-OXOOCTADECANOATE
- 432020-20-7 (4-Chloro-3-nitrophenyl)(4-(2-fluorophenyl)piperazin-1-yl)methanone
- 92-69-3 4-Phenylphenol
- 7292-16-2 PROPAPHOS
- 1943-83-5 2-Chloroethyl isocyanate
- 42142-52-9 3-Hydroxy-N-methyl-3-phenyl-propylamine
- 80370-42-9 Propanoic acid,2-formyl-3-oxo-, ethyl ester
- 64811-37-6 1,4-Benzenedicarboxylic acid, dimethyl ester, polymer with 1,4-butanediol, methyloxirane and oxirane


