- Parham Cyclization
-
Parham Cyclization
W. E. Parham et al., J. Org. Chem. 40, 2394 (1975).
Four- to seven-membered ring annulation of aryl bromides bearing ortho side chains having an electrophilic moiety, accomplished by halogen-metal exchange and subsequent nucleophilic ring closure:
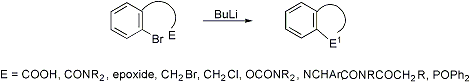
Synthetic application: M. R. Paleo et al., J. Org. Chem. 40, 2029 (1975); A. Couture et al., Chem. Commun. 1994; 1329; M. I. Collado et al., Tetrahedron Letters 37, 6193 (1996); S. D. Larsen, Synlett 1997; 1013; A. Ardeo et al., Tetrahedron Letters 41, 5211 (2000). Review: W. E. Parham, C. K. Bradsher, Accts. Chem. Res. 15, 300-305 (1982).
Prev: Passerini Reaction
Next: Overman Rearrangement - 【Back】【Close 】【Print】【Add to favorite 】
-
Health and Chemical more >
-
Hot Products
- 7789-59-5 Phosphorus oxybromide
- 24697-74-3 Leonurine hydrochloride
- 107-21-1 Ethylene glycol
- 845866-53-7 3-(2-Methylbenzyloxy)bromobenzene
- 2439-10-3 Guanidine,N-dodecyl-
- 881386-01-2 2-Piperidinone, 3,3-dichloro-1-(4-nitrophenyl)-
- 85-41-6 O-Phthalimide
- 3383-21-9 3,5-DI-TERT-BUTYL-O-BENZOQUINONE
- 2627-86-3 Benzenemethanamine, a-methyl-, (aS)-
- 68555-98-6 Phenol, 4-(1,1-dimethylpropyl)-, polymer with sulfur chloride (S2Cl2)
- 52504-24-2 Softigen 767
- 889939-67-7 3-(4-Ethoxy-3-methoxyphenyl)propan-1-amine


