- Quelet Reaction
-
Quelet Reaction
R. Quelet, Compt. Rend. 195, 155 (1932).
Passage of dry hydrochloric acid through a solution in ligroin of a phenolic ether and an aliphatic aldehyde in the presence or absence of a dehydration catalyst to yield α-chloroalkyl derivatives by substitution in the para position to the ether group or in the ortho position in para-substituted phenolic ethers:
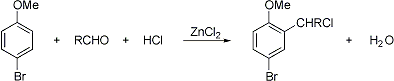
R. Quelet, ibid. 196, 1411 (1933); 198, 102 (1934); 199, 150 (1934); 202, 956 (1936); Bull. Soc. Chim. France 7, 196 (1940); U. Neda, R. Oda, J. Soc. Chem. Ind. Japan 47, 565 (1944). Cf. Blanc (Chloromethylation) Reaction.
Prev: Raschig Phenol Process
Next: Pummerer Rearrangement - 【Back】【Close 】【Print】【Add to favorite 】
- Related information
- Reaction routes of Metronidazole
- Reaction Kettle_008
- Production of 4-(2-Bromo-2-propenyl)-4-methyl-γ-butyrolactone by the Reaction of Ethyl levulinate with (2-bromoallyl)diisopropoxyborane
- Double Hydroxylation Reaction: 16α-Methylcortexolone
- Diels-Alder reaction: (1S-endo)-3-(Bicyclo[2.2.1]hept-5-en-2-ylcarbonyl)-2-oxazolidinone
- 4-(2-Bromo-2-propenyl)-4-methyl-gamma-butyrolactone: from the reaction of Ethyl levulinate and (2-Bromoallyl)diisopropoxyborane
- Synthesis of trans-4,4'-Dibromostilbene by a Double Heck Reaction of (Arylazo)amines with Vinyltriethoxysilane
- Single Displacement Reactions
- Reaction Design
- Chemical Reaction Stoichiometry (CRS, Tutorial, Java Applet, and Resources)
-
Health and Chemical more >
-
Hot Products
- 64228-81-5 Atracurium besylate
- 2235-54-3 Ammonium lauryl sulfate
- 24979-70-2 Poly(p-vinylphenol)
- 7440-59-7 Helium
- 3238-40-2 2,5-Furandicarboxylic acid
- 40501-41-5 METHYL 4'-HYDROXY[1,1'-BIPHENYL]-4-CARBOXYLATE
- 955964-94-0 3,5-dimethyl-1-(3-methylbenzyl)-1H-pyrazol-4-amine
- 108-73-6 Phloroglucinol
- 57773-65-6 Deslorelin
- 9004-78-8 Poly(oxy-1,2-ethanediyl), .alpha.-phenyl-.omega.-hydroxy-
- 99899-09-9 Phenol, 2-[1-[[2-[(2-hydroxyethyl)amino]ethyl]imino]ethyl]-
- 85700-55-6 Scopine hydrochloride


