- Stollé Synthesis
-
StolléSynthesis
R. Stollé, Ber. 46, 3915 (1913); 47, 2120 (1914); J. Prakt. Chem. 105, 137 (1923); 128, 1 (1930).
Formation of indole derivatives by the reaction of arylamines with α-haloacid chlorides or oxalyl chloride, followed by cyclization of the resulting amides with aluminum chloride:
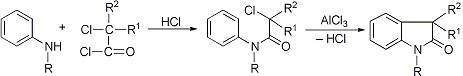
W. C. Sumpter, Chem. Rev. 34, 396 (1944); 37, 446 (1945); P. L. Julian et al., Heterocyclic Compounds 3, 142, 209 (1952); A. H. Beckett et al., Tetrahedron 24, 6093 (1968). Cf. Hinsberg Synthesis.
Prev: Stork Enamine Reaction
Next: Widman-Stoermer Synthesis - 【Back】【Close 】【Print】【Add to favorite 】
- Related information
- International Priceso of Synthesis Ammonia in the Falling Trends
- International Prices of Synthesis Ammonia in Falling Trends
- International Prices of Synthesis Ammonia in Falling Trends
- International Prices of Synthesis Ammonia in Differentiation Trends
- International Prices of Synthesis Ammonia in Differentiation Trends
- International Prices of Synthesis Ammonia in Rising Trend
- International Prices of Synthesis Ammonia in Rising Trend
- International Prices of Synthesis Ammonia in Rising Trend
- International Prices of Synthesis Ammonia in Rising Trend
- International Prices of Synthesis Ammonia in Early September
-
Health and Chemical more >
-
Hot Products
- 399-97-3 2-AMINO-4-FLUOROPHENOL
- 106-65-0 Dimethyl succinate
- 400087-31-2 2,6-dichloro-N'-(3-chloro-2-pyridinyl)benzenecarbohydrazide
- 123654-26-2 4-Amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid
- 130-40-5 Riboflavin-5-phosphate sodium
- 7727-54-0 Ammonium persulfate
- 547-64-8 Propanoic acid,2-hydroxy-, methyl ester
- 50-43-1 2,4,6-Trichlorobenzoic acid
- 4045-44-7 1,3-Cyclopentadiene,1,2,3,4,5-pentamethyl-
- 2216-34-4 4-Methyloctane
- 72-18-4 L-Valine
- 4389-45-1 3-Methylanthranilic acid


