- Wohl-Ziegler Reaction
-
Wohl-Ziegler Reaction
A. Wohl, Ber. 52, 51 (1919); K. Ziegler et al., Ann. 551, 30 (1942).
Allylic bromination of olefins with N-. Peroxides or ultraviolet light are used as initiators:
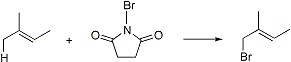
Reviews: C. Djerassi, Chem. Rev. 43, 271 (1948); L. Horner, E. M. Winkelman, Angew. Chem. 71, 349 (1959); S. S. Novikov, et al., Russ. Chem. Rev. 31, 671 (1962); A. Nechvatal, Adv. Free-Radical Chem. 4, 175-201 (1972).
Prev: Wolff Rearrangement
Next: Wohl Degradation - 【Back】【Close 】【Print】【Add to favorite 】
- Related information
- Reaction routes of Metronidazole
- Reaction Kettle_008
- Production of 4-(2-Bromo-2-propenyl)-4-methyl-γ-butyrolactone by the Reaction of Ethyl levulinate with (2-bromoallyl)diisopropoxyborane
- Double Hydroxylation Reaction: 16α-Methylcortexolone
- Diels-Alder reaction: (1S-endo)-3-(Bicyclo[2.2.1]hept-5-en-2-ylcarbonyl)-2-oxazolidinone
- 4-(2-Bromo-2-propenyl)-4-methyl-gamma-butyrolactone: from the reaction of Ethyl levulinate and (2-Bromoallyl)diisopropoxyborane
- Synthesis of trans-4,4'-Dibromostilbene by a Double Heck Reaction of (Arylazo)amines with Vinyltriethoxysilane
- Single Displacement Reactions
- Reaction Design
- Chemical Reaction Stoichiometry (CRS, Tutorial, Java Applet, and Resources)
-
Health and Chemical more >
-
Hot Products
- 323194-76-9 L-ASPARTIC ACID SODIUM SALT MONOHYDRATE
- 590-86-3 Isovaleraldehyde
- 80658-20-4 Glycyrrhizic acid 2NH4
- 57018-52-7 1-TERT-BUTOXY-2-PROPANOL
- 210357-12-3 N-Cocoyl-L-Glutamic Acid
- 762240-92-6 3-(Trifluoromethyl)-5,6,7,8-tetrahydro-[1,2,4]triazolo[4,3-a]pyrazine hydrochloride
- 1532-24-7 CLOPYRALID METHYL ESTER
- 6155-57-3 Saccharin sodium dihydrate
- 7631-90-5 Sulfurousacid, sodium salt (1:1)
- 91424-40-7 3-(tert-Butyldimethylsilyloxy)glutaric anhydride
- 142-64-3 Piperazine, hydrochloride (1:2)
- 70693-43-5 1-Octene, homopolymer, hydrogenated


