- Cobalt
- Cobalt carbonate
- Cobalt chloride
- Cobalt chloride
- Cobalt chloride hexahydrate
- Cobalt disulfamate
- Cobalt disulfide
- Cobalt hydroxide oxide
- Cobalt iodide,dihydrate (8CI,9CI)
- Cobalt oxide
- 10026-22-9Nitric acid, cobalt(2+)salt, hydrate (2:1:6)
- 10026-24-1Sulfuricacid, cobalt(2+) salt, hydrate (1:1:7)
- 100-26-52,5-Pyridinedicarboxylicacid
- 1002-67-1Ethane,1-ethoxy-2-(2-methoxyethoxy)-
- 1002-69-3Decane, 1-chloro-
- 1002726-59-12,2-DIMETHYL-6-NITRO-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 1002726-62-66-Amino-2,2-dimethyl-2H-pyrido[3,2-b] [1,4]oxazin-3[4H]-one
- 10027-30-21,2-Benzenedicarboxylicacid, copper(2+) salt (1:1)
- 100275-94-31,4-Benzodioxin,2,3-dihydro-6-isocyanato-
- 100-27-6Benzeneethanol,4-nitro-
Hot Products
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
- 7631-86-9Silicon dioxide
- 302-79-4Tretinoin
- 77-92-9Citric acid

|
Basic Information |

|
Post buying leads |

|
Suppliers |

|
Cas Database |

| Name |
Cobalt trifluoride |
EINECS | 233-062-4 |
| CAS No. | 10026-18-3 | Density | 3.88 g/cm3 |
| PSA | 0.00000 | LogP | 1.26060 |
| Solubility | N/A | Melting Point |
927 °C |
| Formula | CoF3 | Boiling Point | 19.5oC at 760mmHg |
| Molecular Weight | 115.988 | Flash Point | N/A |
| Transport Information | UN 3260 8/PG 2 | Appearance | Brown solid |
| Safety | 26-36/37/39-45 | Risk Codes | 34 |
| Molecular Structure |
|
Hazard Symbols |
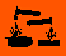 C, C, T, T, O O
|
| Synonyms |
Cobalt(III) fluoride;Cobalt fluoride;Cobalt trifluoride;Cobaltic fluoride;Cobaltic trifluoride; |
Article Data | 40 |
Cobalt trifluoride Consensus Reports
Cobalt and its compounds are on the Community Right-To-Know List.
Cobalt trifluoride Specification
The Cobalt trifluoride with CAS registry number of 10026-18-3 is also known as Cobalt fluoride. The IUPAC name is Cobalt(3+) trifluoride. It belongs to product categories of Inorganic Fluorides. Its EINECS registry number is 233-062-4. In addition, the formula is CoF3 and the molecular weight is 115.93.
Physical properties about Cobalt trifluoride are: (1)H-Bond Acceptor: 3; (2)Exact Mass: 115.92841; (3)MonoIsotopic Mass: 115.92841; (4)Heavy Atom Count: 4; (5)Covalently-Bonded Unit Count: 4.
Preparation of Cobalt trifluoride: it is prepared in the laboratory by reaction of CoCl2 with fluorine at the temperature of 250 °C.
CoCl2+3/2F2→CoF3+Cl2
Uses of Cobalt trifluoride: it is is a powerful fluorinating agent, which decomposes upon contact with water to give oxygen.
4CoF3+2H2O→4HF+4CoF2+O2
When you are using this chemical, please be cautious about it. As a chemical, it may cause burns. During using it, wear suitable protective clothing, gloves and eye/face protection. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If accident happens or you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: [F-].[F-].[F-].[Co+3]
2. InChI: InChI=1S/Co.3FH/h;3*1H/q+3;;;/p-3
3. InChIKey: WZJQNLGQTOCWDS-UHFFFAOYSA-K

