
Journal of Organic Chemistry p. 346 - 364 (2019)
Update date:2022-08-15
Topics:
 Henry, Martyn C.
Henry, Martyn C.
 Senn, Hans Martin
Senn, Hans Martin
 Sutherland, Andrew
Sutherland, Andrew
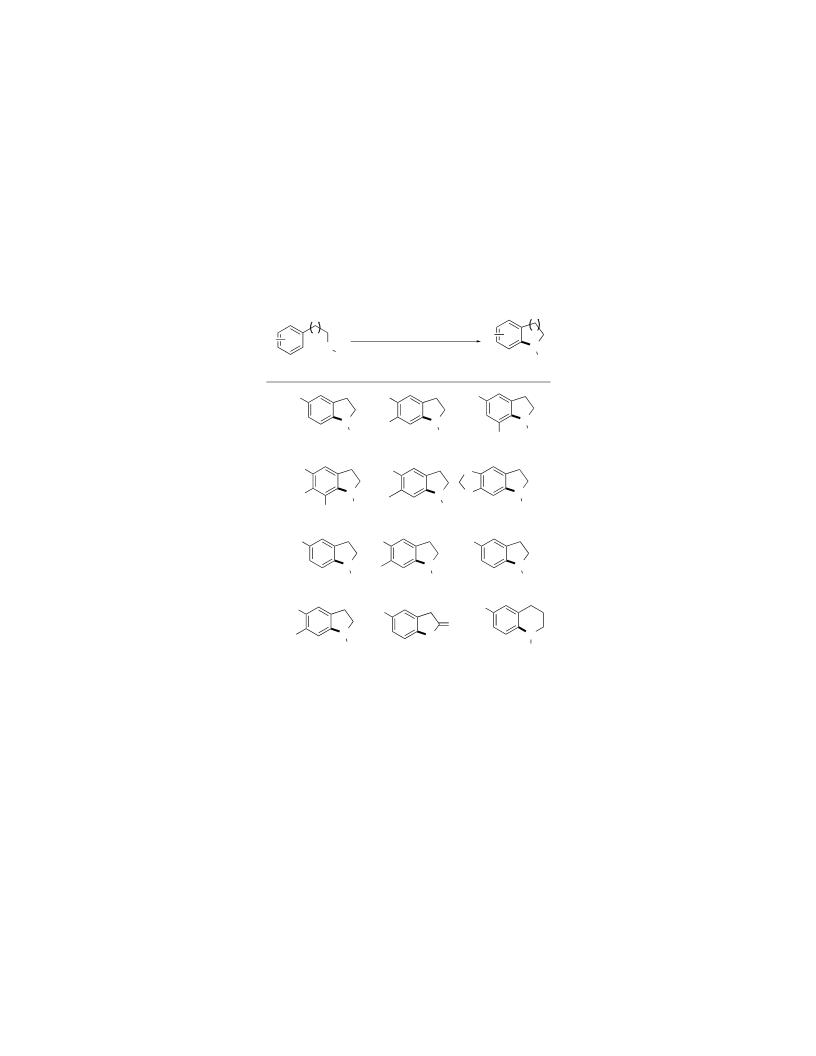
A simple and effective one-pot, two-step intramolecular aryl C-N and C-O bond forming process for the preparation of a wide range of benzo-fused heterocyclic scaffolds using iron and copper catalysis is described. Activated aryl rings were subjected to a highly regioselective, iron(III) triflimide-catalyzed iodination, followed by a copper(I)-catalyzed intramolecular N-or O-arylation step leading to indolines, dihydrobenzofurans, and six-membered analogues. The general applicability and functional group tolerance of this method were exemplified by the total synthesis of the neolignan natural product, (+)-obtusafuran. DFT calculations using Fukui functions were also performed, providing a molecular orbital rationale for the highly regioselective arene iodination process.
View More










Wuhan Sun-shine Bio-technology Corporation Limited
Contact:+862765522452
Address:武汉市东湖高新区高新二路388号生物医药加速器16栋501
TIANJIN ZHONGXIN CHEMTECH CO.,LTD.
Contact:86-022-89880739
Address:10B, Pan China International Center, No. 931 YingKou Road, TangGu, Tianjin, China, 300451
website:http://www.weichichem.com
Contact:+8613912949432
Address:Fine Chemical Industrial Base,Wujiang town,He County,Anhui China.
Wuhan Sunrise Pharmaceutical Technology Co., Ltd
Contact:+86-27-83314682
Address:Room 340, New material Industrial base No.17, Gu Tian Five Lu , Qiaokou District, Wuhan , China
Beyond Pharmaceutical Co., Ltd
Contact:+86-571-8195-3185
Address:No. 13-1, Liansheng Road, Yuhang District
Doi:10.1021/ja00977a016
(1967)Doi:10.1021/jo00319a034
(1981)Doi:10.1002/oms.1210171106
(1982)Doi:10.1007/BF00903108
(1973)Doi:10.1002/anie.201809115
(2018)Doi:10.1007/BF00760850
()