
Organic Letters p. 6000 - 6003 (2012)
Update date:2022-08-15
Topics:
 Lauer, Matthew G.
Lauer, Matthew G.
 Henderson, William H.
Henderson, William H.
 Awad, Amneh
Awad, Amneh
 Stambuli, James P.
Stambuli, James P.
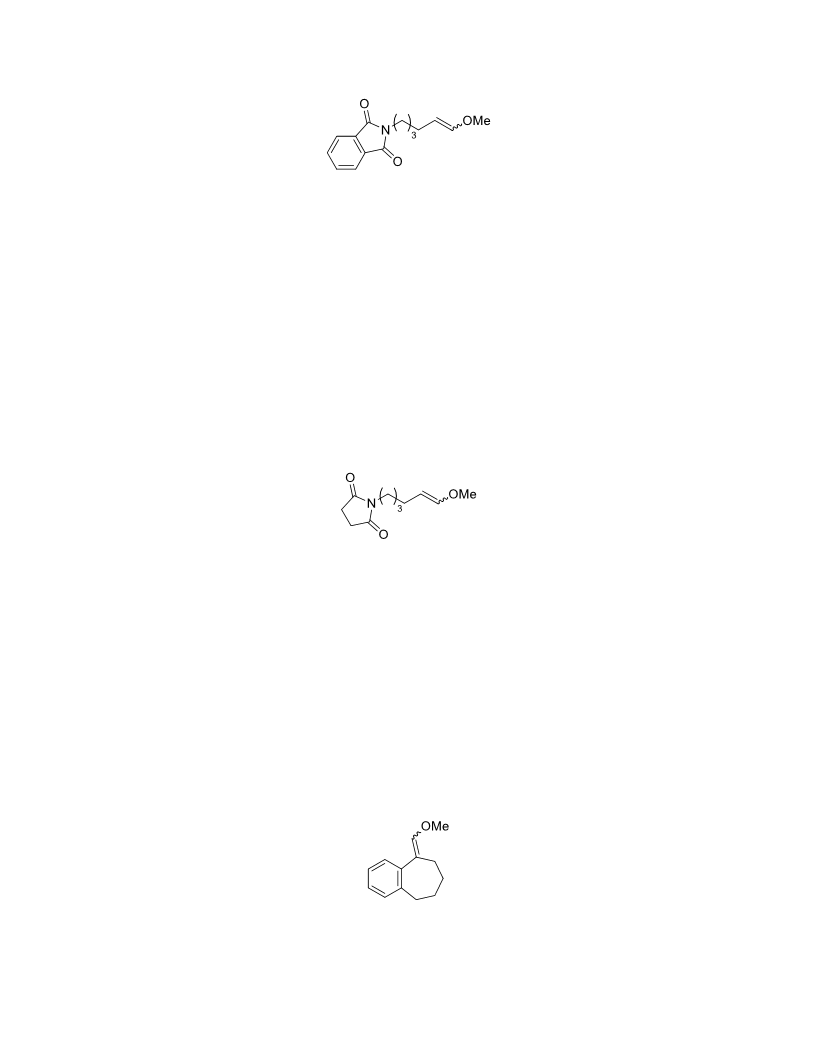
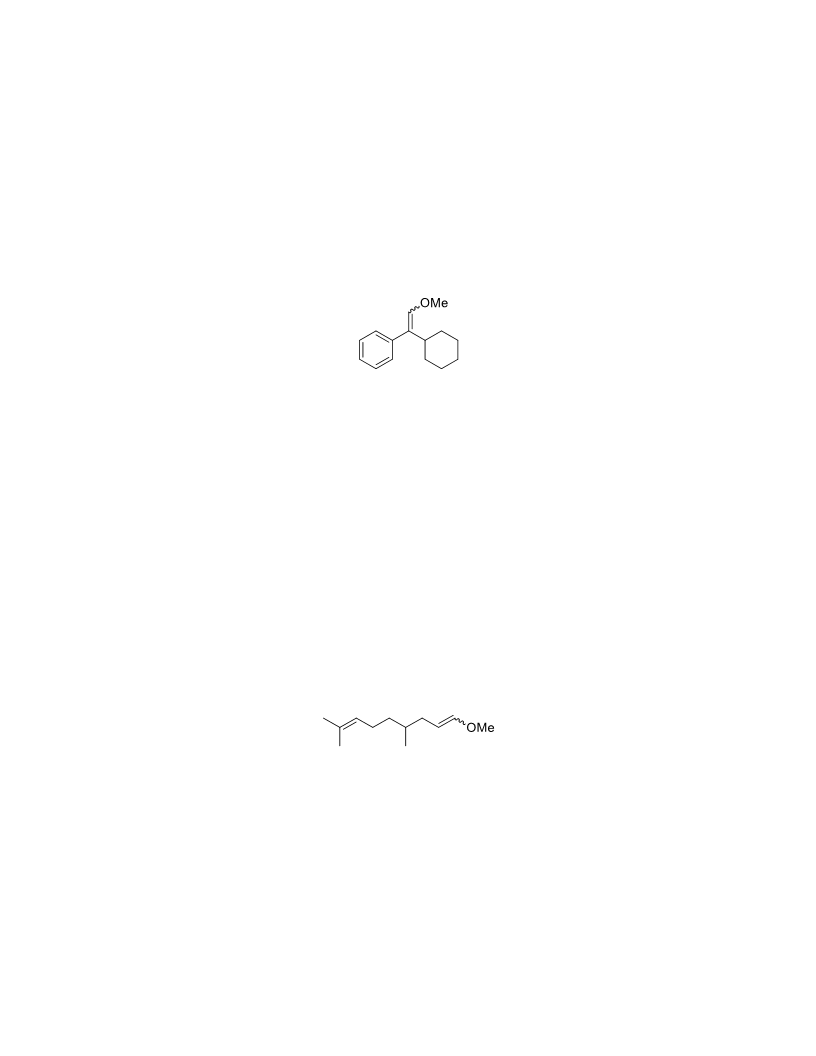
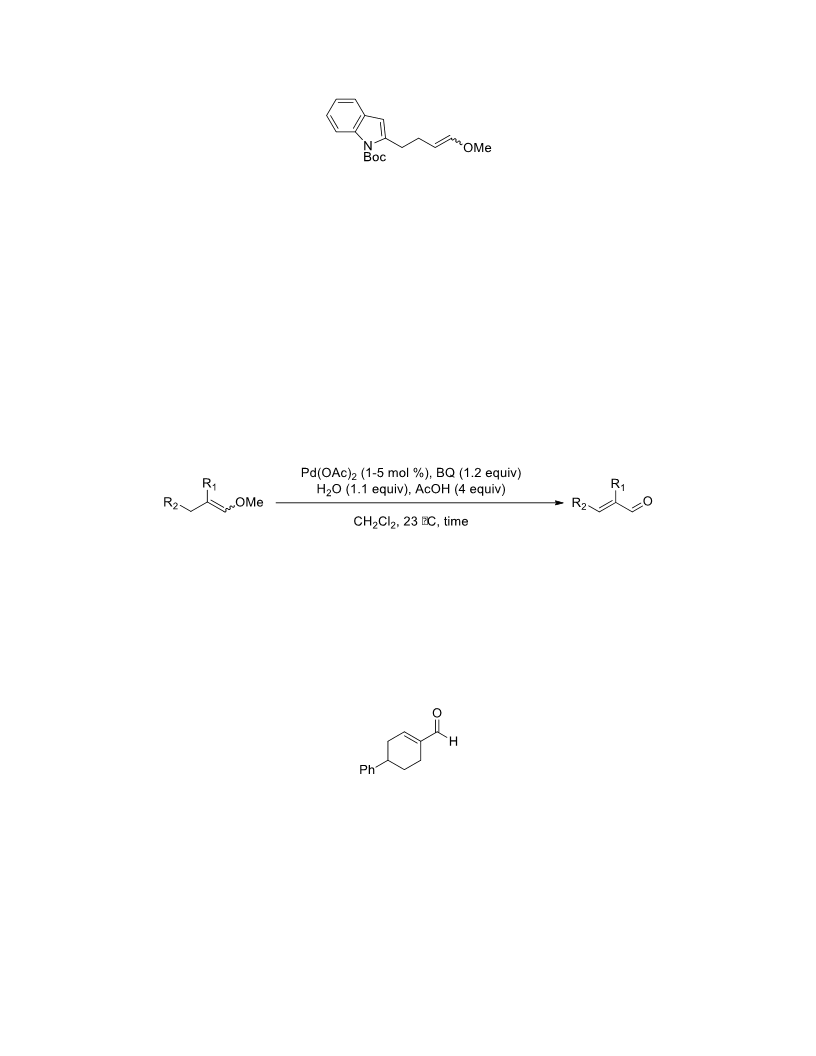
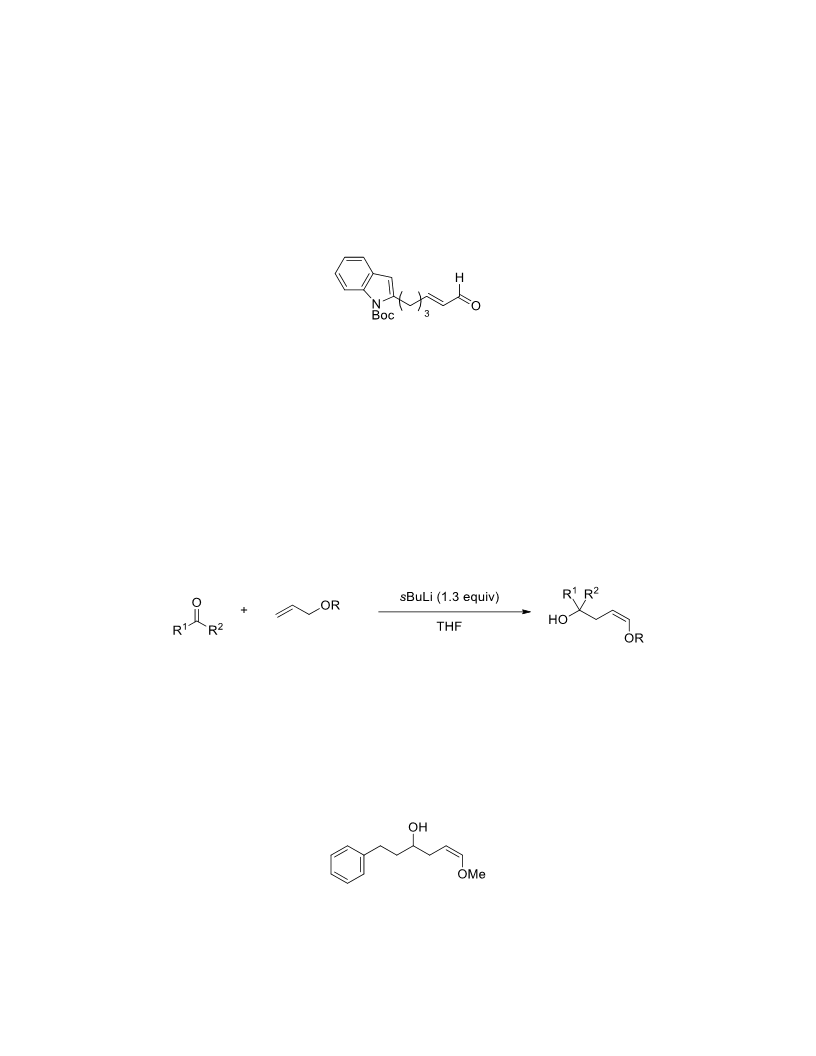
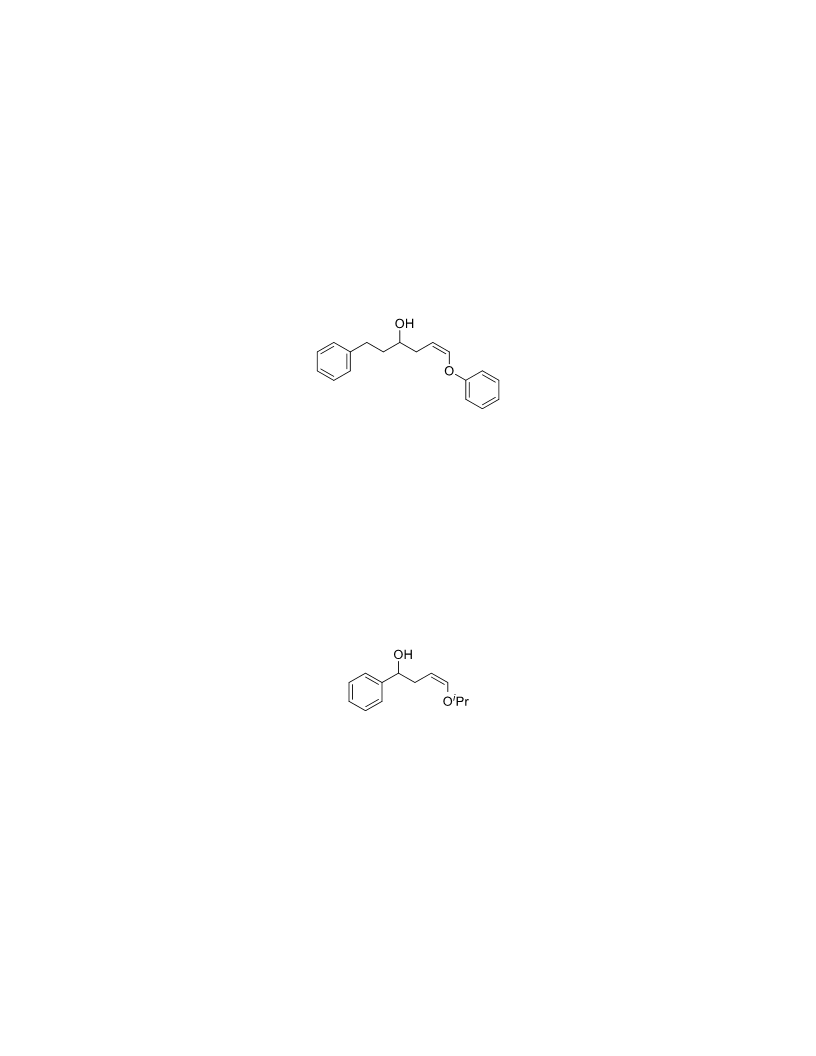
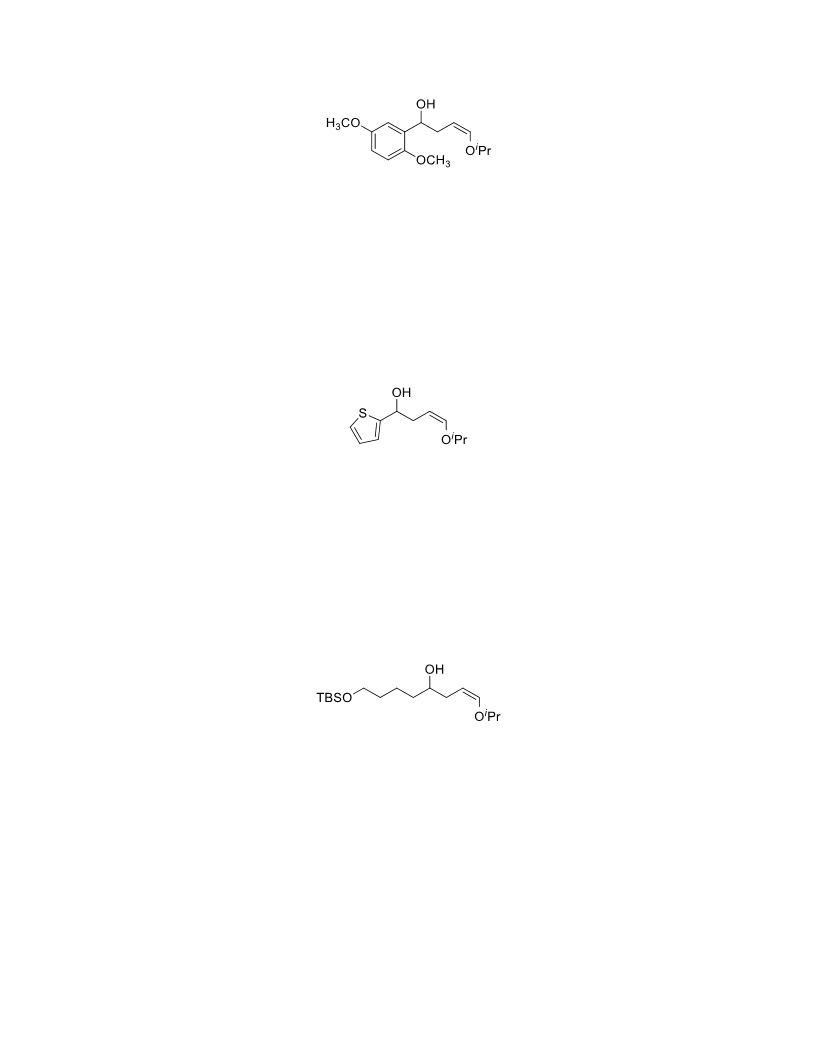
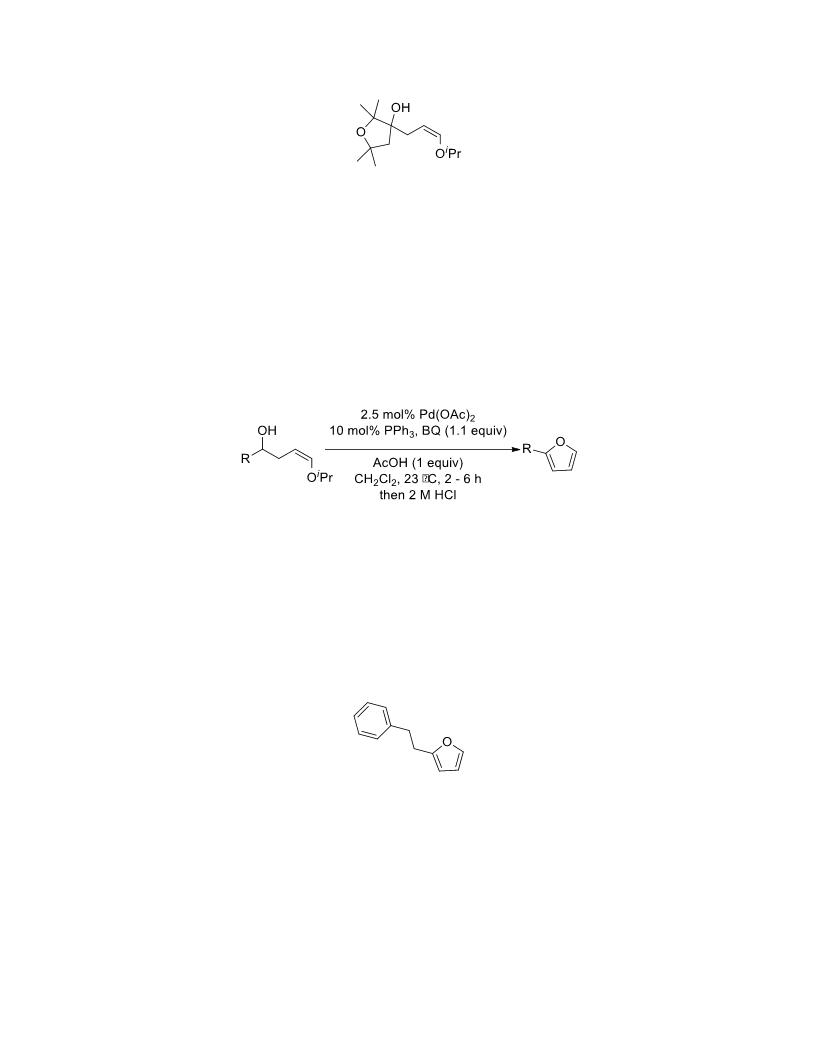
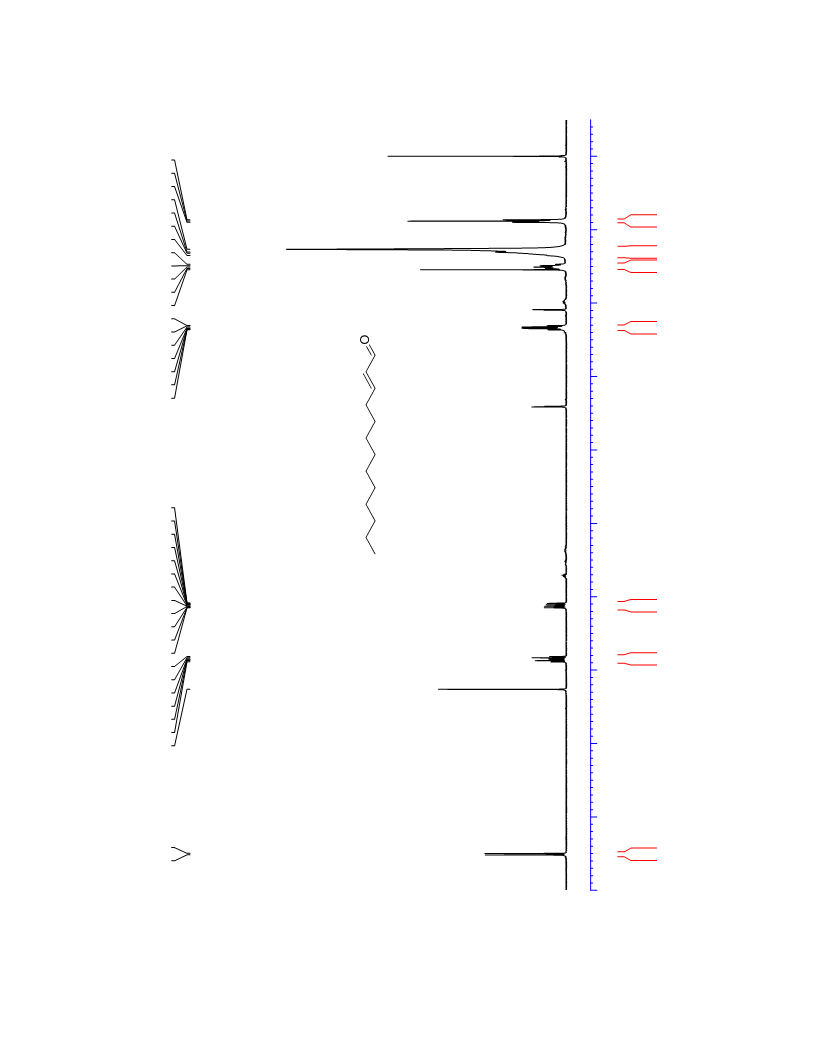
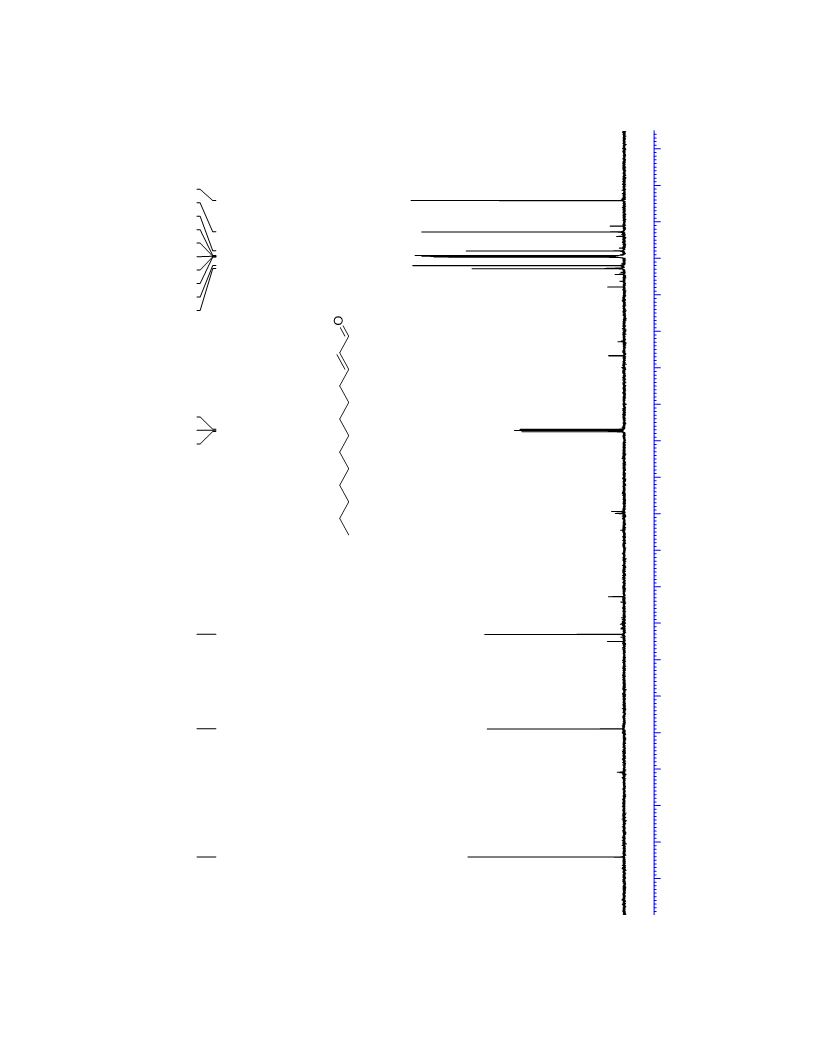
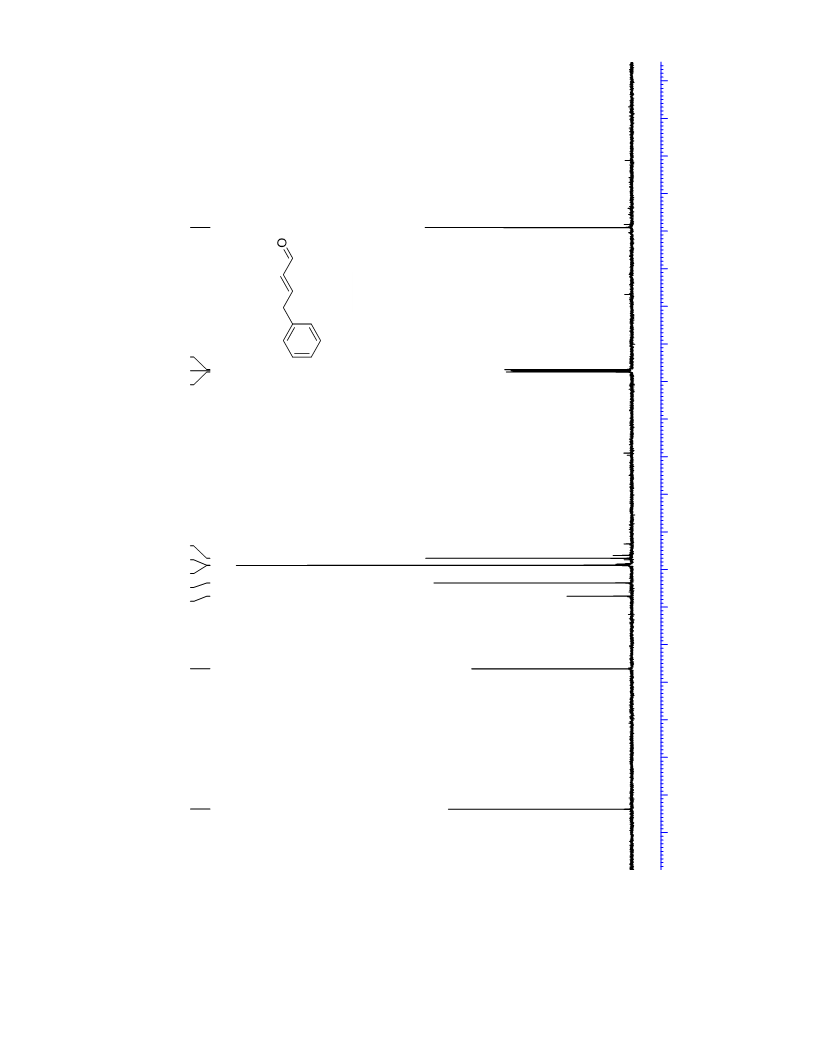
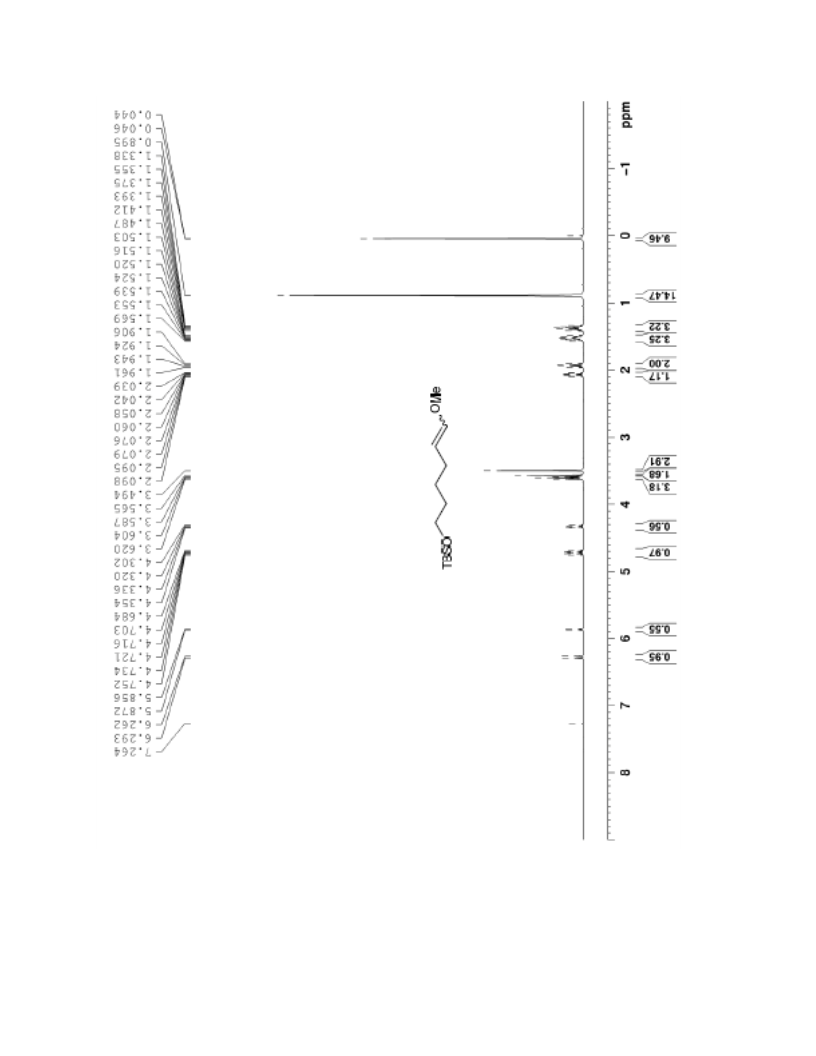
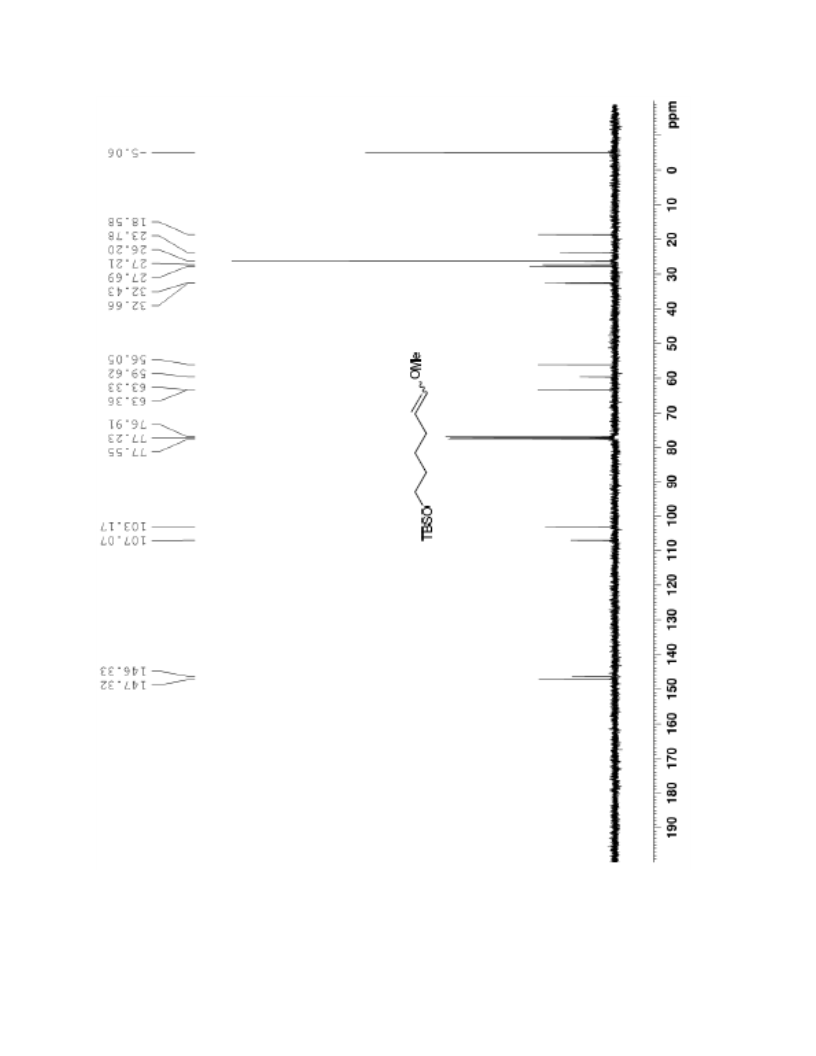
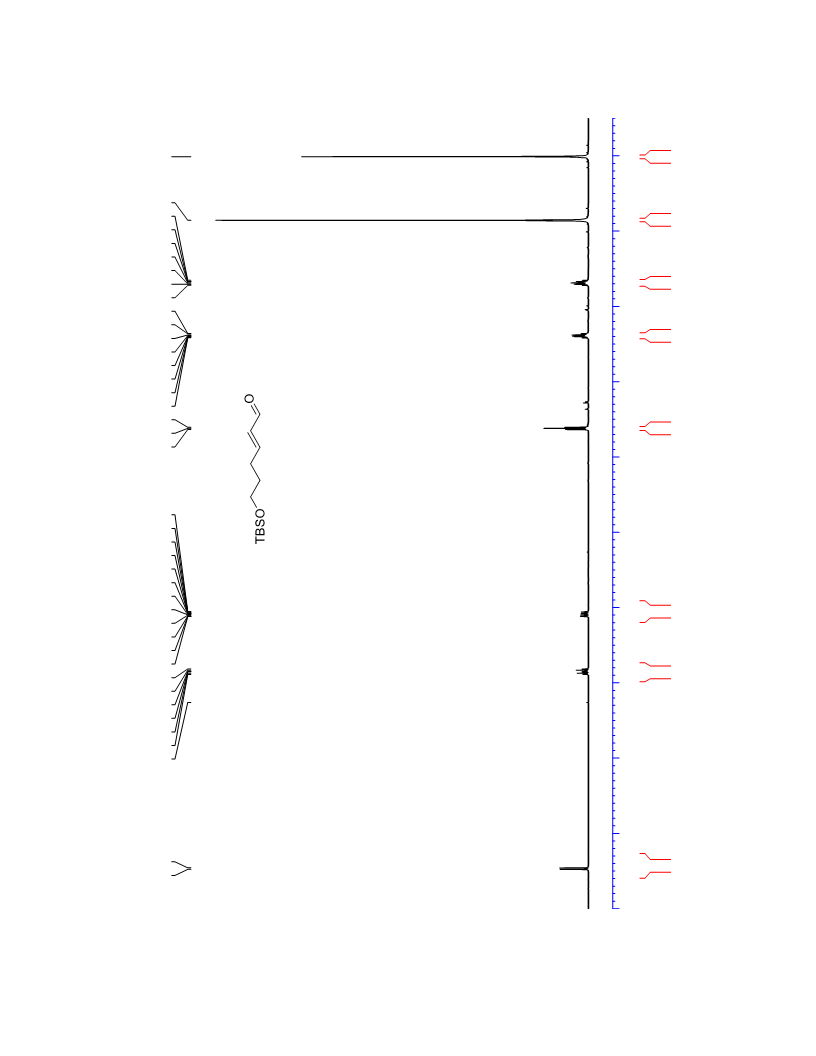
The palladium-catalyzed oxidation of alkyl enol ethers to enals, which employs low loadings of a palladium catalyst, is described. The mild oxidation conditions tolerate a diverse array of functional groups, while allowing the formation of di-, tri-, and tetrasubtituted olefins. The application of this methodology to intramolecular reactions of alkyl enol ethers containing pendant alcohols provides furan and 2,5-dihydrofuran products.
View More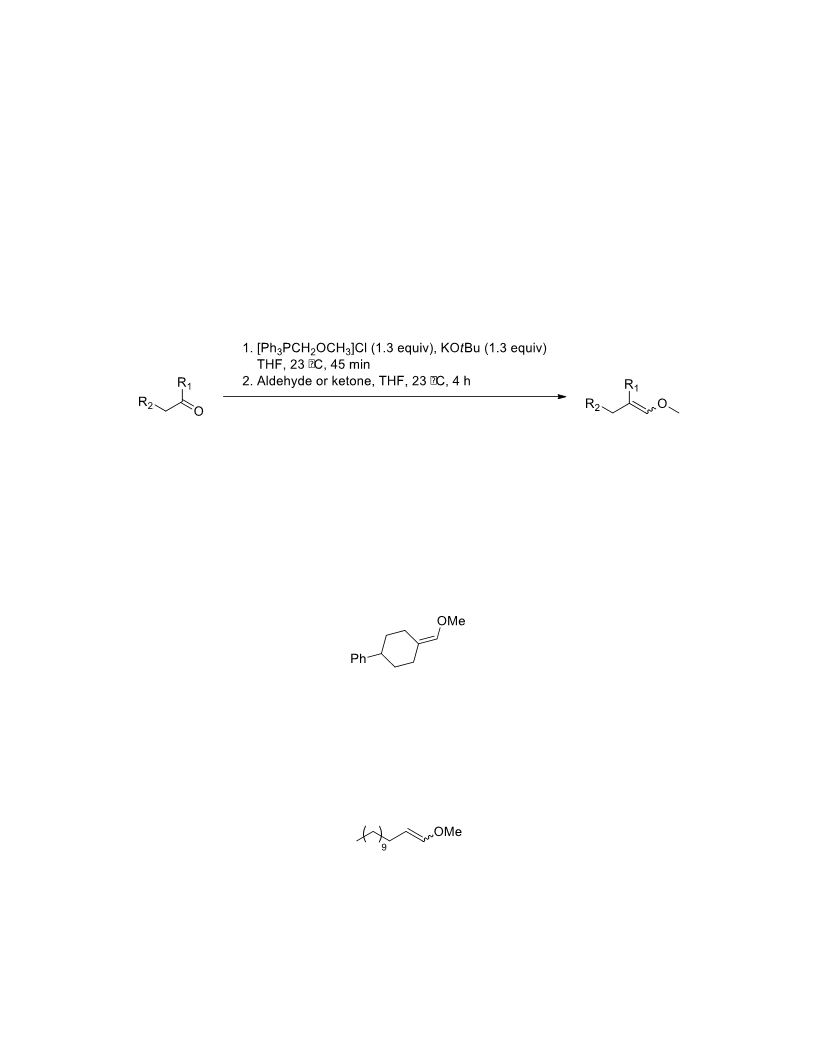













QINGDAO NEW FLOURISH INTERNATIOANAL TRADE CO., LTD.
Contact:+86 532 80861829
Address:No. 1, Yinchuan East Road, 266061, Qingdao, China
Hangzhou Think Chemical Co. Ltd
website:http://www.thinkchem.com/
Contact:86-571-89986307/81956191/81956084/81956192
Address:Room 501-502, Tower E, Yuanjian Building, Yuanyang International Center, Hangzhou-310011, China
KangZhiYuan Pharmaceutical Company Limited
Contact:(Sabrina)86-20-85273232
Address:4th floor, building B, Dadi industry zone, Tangxia, Tianhe, Guangzhou, China
Contact:13813902930 025-52714267
Address:20 Fengji Road, Yuhua Economic Development Zone, Nanjing, Jiangsu, P. R. China
Puyang Huicheng Electronic Material Co., Ltd
website:http://huichengchem.weba.testwebsite.cn/index_en.html
Contact:+86-393-8910800
Address:West Section Shengli Road, Puyang457000, China
Doi:10.1055/s-0032-1316753
(2012)Doi:10.1016/j.molstruc.2021.131154
(2021)Doi:10.1016/j.tetlet.2012.09.096
(2012)Doi:10.1016/j.ejmech.2012.09.019
(2012)Doi:10.1248/cpb.42.1784
(1994)Doi:10.1016/j.bmc.2012.10.009
(2012)