
Journal of Organic Chemistry p. 2616 - 2621 (2005)
Update date:2022-08-15
Topics:
 Buron, Frederic
Buron, Frederic
 Ple, Nelly
Ple, Nelly
 Turck, Alain
Turck, Alain
 Queguiner, Guy
Queguiner, Guy
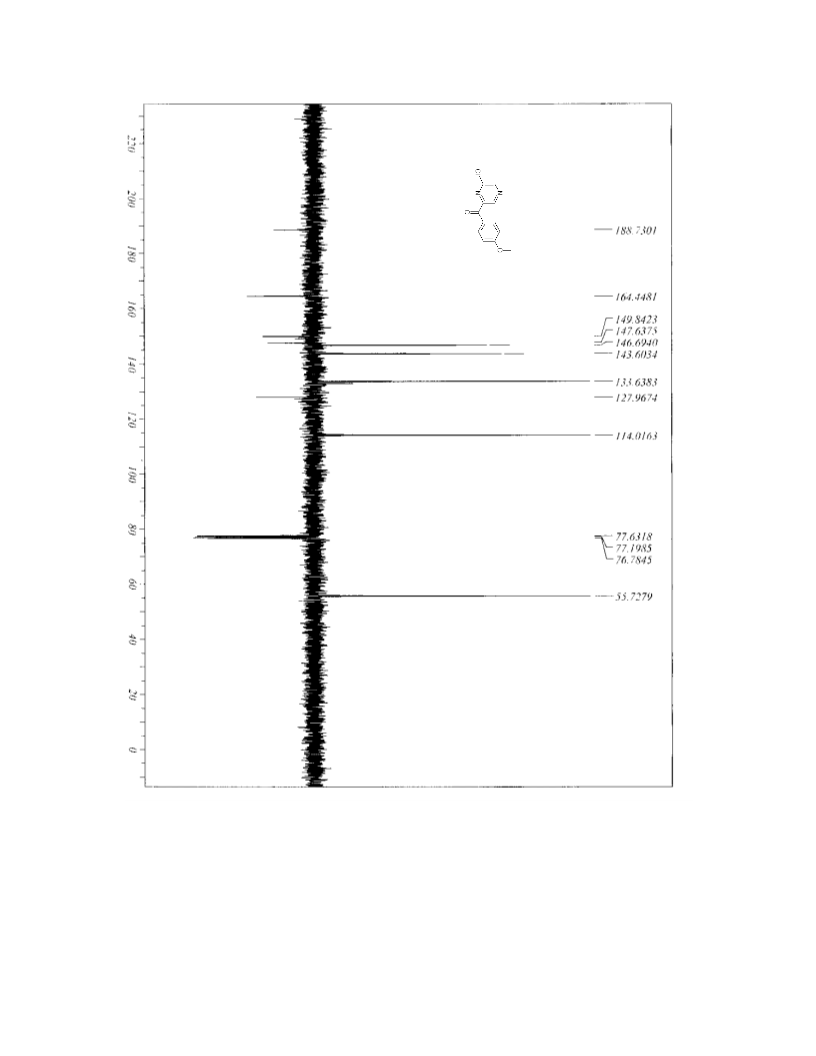
(Chemical Equation Presented) Regioselective metalation of pyrazines and cross-coupling reactions provides an easy access to botryllazines A and B and to an isomer of botryllazine A with good yields from chloropyrazine.
View More



























Contact:+86-18653358619
Address:zibo
Changzhou Ruiping Chemical Co., Ltd
website:http://www.wishchem.com
Contact:+86-519-82324280
Address:No.288-1 Huacheng Road, Jintan
Wuhan Chemwish Technology Co., Ltd
website:http://www.chemwish.com/
Contact:+86-27-67849912
Address:Room 1311, Unit 2, Block1, Innovation Road East Lake High-tech Development Zone Wuhan, Hubei,P.R. China
Goldwills Pharmaceuticals Co., Ltd.
Contact:0916-2237889 13991621155
Address:North Suburb of Hanzhong city, Shaanxi Province
Chongqing KonAo Health Co.,Ltd
Contact:13687578375
Address:wuhan hubei china
Doi:10.1007/s11172-005-0108-8
(2004)Doi:10.1016/S0040-4020(98)00014-3
(1998)Doi:10.1002/anie.200462713
(2005)Doi:10.1002/ejoc.200400605
(2005)Doi:10.1021/ol050370y
(2005)Doi:10.1023/B:COHC.0000044568.69817.bc
(2004)