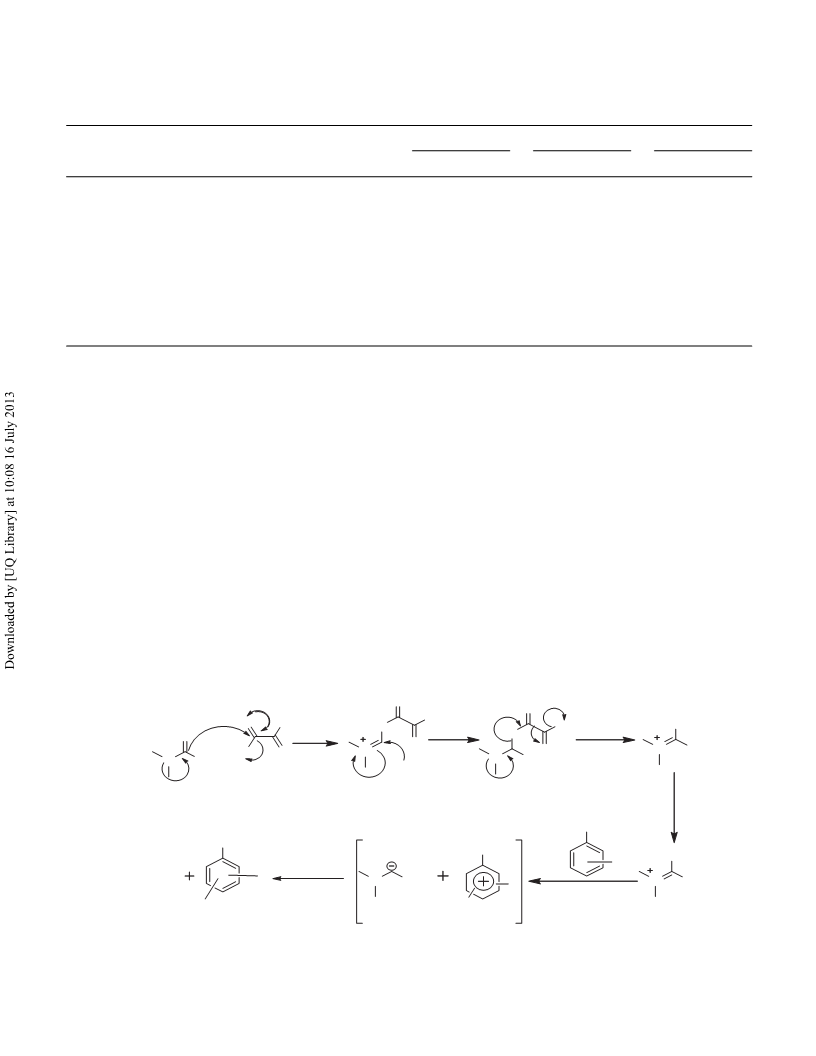
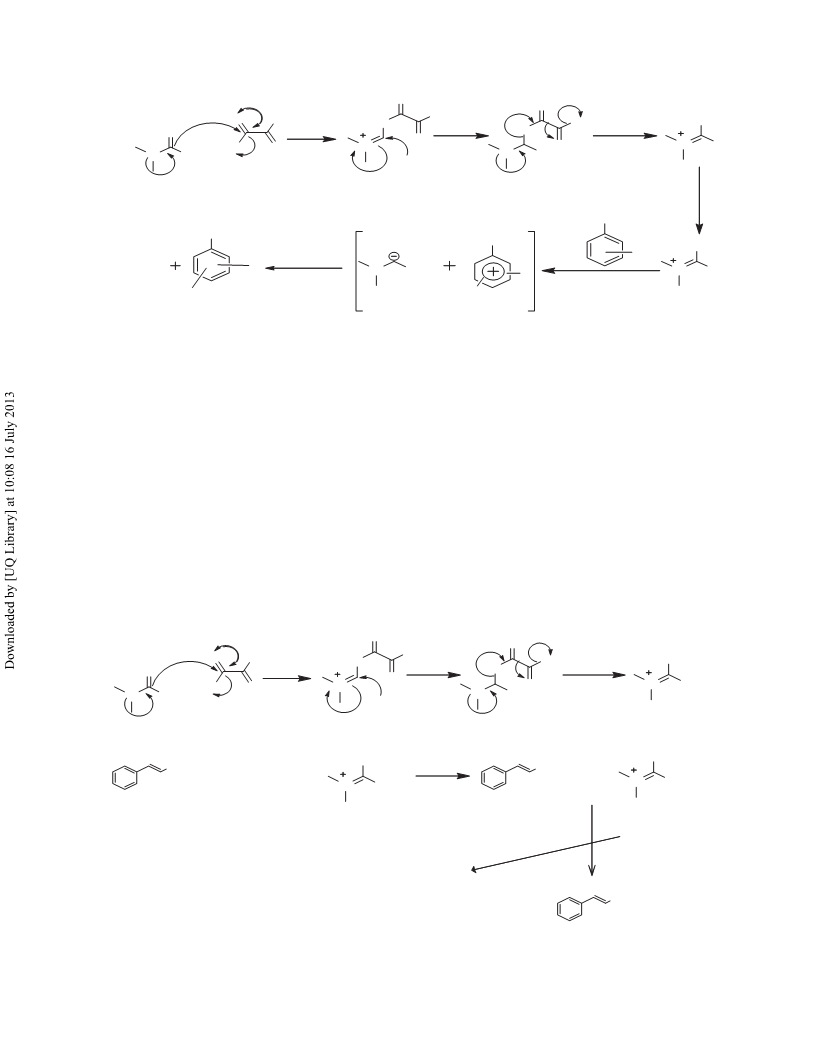
OXALYLCHLORIDE/DMF FOR NITRATION
983
reaction times are reduced from several hours to only 2–3 min 18. (a) 18 Rajanna, K.C.; Abdulla; Amina, S.; Arun Kumar, Y.; Arifuddin, M.
Synth. Comm. 2011, 41, 2946–2951. (b) Rajanna, K.C.; Moazzam Ali; Sana,
S.; Saiprakash, P.K. Chemistry Letters, 2000, 29, 48–49. (c) Ramgopal, S.;
Ramesh, K.; Chakradhar, A.; Maasi Reddy, N.; Rajanna, K.C. Tetrahedron
Lett. 2007, 48, 4043–4045.
9. Olah, G.A.; Fung, A.P.; Narang, S.C.; Olah, J.A. J. Org. Chem. 1981, 46,
3533.
0. Joan, M.; Riego, Z.; Sedin, J.; Zaldivar, M.; Nunziata, C.; Marzianot, C.T.
Tetrahedron Lett. 1996, 37, 513.
1. Das, J.P.; Sinha, P.; Roy, S. A nitro-Hunsdiecker reaction: from unsatu-
rated carboxylic acids to nitrostryrenes and nitroarenes. Org. Lett. 2002, 4,
accompanied by yield enhancements (Table 2). The ability to
rapidly heat reactions significantly above the boiling point of
the solvent has resulted in dramatic decreases in reaction times
and increases in reaction yields for a variety of chemical trans-
1
2
2
formations.[
34,35]
CONCLUSION
In this proposed protocol, we developed iminium salt
3
055–3048.
(
oxalylchloride/DMF) mediated nitration reaction of aromatic 22. (a) Ramgopal, S.; Ramesh, K.; Chakradhar, A.; Reddy, N.M.; Rajanna,
compounds and nitro decarboxylation of cinnamic acids under
sonication and microwave irradiation. Microwave and ultrason-
ically assisted reactions not only reduced the reaction times re-
markably but also enhanced the yield of products from good to
excellent as compared to those of normal protocol. The presently
K.C. Metal nitrate driven nitro Hunsdiecker reaction with α, β-unsaturated
carboxylic acids under solvent-free conditions. Tetrahedron Lett. 2007, 48,
4
043–4045. (b) Satish Kumar, M.; Venkanna, P.; Ramgopal, S.; Ramesh,
K.; Venkateswarlu, M.; Rajanna, K.C. Org. Commun. 2012, 5(2): 42–49.
c) Anna, M.; Alessandra, G.; Isidoro, G.; Fabio, T.; Benedetto, D.B.;
Antonio, F. Syn. Com. 2004, 34, 3317–3324.
(
developed work is more advantageous because the reactions 23. (a) Mason, T.J. Chemistry with ultrasound, Elsevier Science Publishers
Ltd., England, 1990; (b) Mason, T.J.; Peters, D. Practical Sono-Chemistry:
Power Ultrasound Uses and Applications, 2nd edn.; Horwood, Cambridge,
England, 2003. (c) Suslick, K.S. Ultrasound: Its Chemical, Physical and
are conducted with economically cheap and readily available
reagents.
Biological Effects; VCH, London, 1988. (d) Margulis, M.A. Advances in
Sonochemistry, Mason, T.J., editor. Greenwich, London, 1990; 1, 49.
24. (a) Chaouchi, M.; Loupy, A.; Marque, S.; Petit, A. Eur. J. Org. Chem. 2002,
1278. (b) Gedye, R.N.; Smith, F.E.; Westaway, K.C. Can. J. Chem. 1988,
66, 17. (c) Loupy, A.; Perreux, L.; Liagre, M.; Burle, K.; Moneuse, M. Pure
Appl. Chem. 2001, 73, 161.
25. Rajanna, K.C.; Satish Kumar, M.; Venkanna, P.; Ramgopal, S.;
Venkateswarlu, M. Int. J. Org Chem. 2011, 4, 250–256.
26. Vilsmeier, A.; Haack, A. Ber. 1927, 60, 119.
27. (a) Barnett, G.H.; Anderson, H.J.; Loader, C.E. Can. J. Chem. 1980, 58,
409. (b) Salmon, R. Encyclopedia of Reagents for Organic Synthesis. Wiley,
New York, 2001.
28. Al’bina, I.; Mikhaleva, A.V.; Ivanov, E.V.; Skital’tseva, I.A.; Ushakov,
A.M.; Vasil’tsov, B.A.T. Synthesis 2009, 4, 587–590.
29. (a) Mason, T.J. Applied sonochemistry. In: Kirk-Othmer Encyclopedia of
Chemical Technology, Kroschwitz, J.I.; Howe-Grant, M.; editors. Wiley,
New York, 1991. (b) Mason, T.J. Practical Sonochemistry: User’s Guide
to Applications in Chemistry and Chemical Engineering, Ellis Horwood
Series in Organic Chemistry; Horwood, New York, 1991.
30. Mason, T.J.; Cobley, A.J.; Graves, J.E.; Morgan, D. Ultrason. Sonochem.
2011, 18, 226.
REFERENCES
1
2
3
4
5
. Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford
University Press, Oxford, England, 1999.
. Olah, G.A.; Malhotra, R.; Narang, S.C. Nitration: Methods and Mechanism,
Feuer, H. editor. VCH, New York, 1989.
. Ingold, C.K. Structure and Mechanism in Organic Chemistry, 2nd edn.;
Cornell University Press, Ithaca, New York, 1969.
. Olah, G.A.; Kuhn, S.J. In Friedel-Crafts and Related Reactions, Olah, G.A.
editor. Wiley-Interscience, New York, 1964; Vol. 2.
. Olah, G.A.; Narang, S.C.; Olah, J.A.; Lammertsma, K. Proc. Natl. Acad.
Sci. 1982, 4487.
6. Thompson, M.J.; Zeeger, P.J. Tetrahedron 1991, 47, 8787.
7. Bisarya, S.C.; Joshi, S.K.; Holker, A.G. Synth. Commun. 1993, 8, 1125.
8. Yang, G.S.; Shi, J.; Li, J. Korean J. Chem. Eng. 2003, 20, 886.
9. Joshi, A.V.; Baidoosi; Mukhopadhyay, S.; Sasson, Y. Org. Proc. Res. Dev.
2
003, 7, 95.
1
1
1
0. Bahulayan, D.; Narayan, G.; Sreekumar, V.; Lalithambika, M. Synth. Com-
mun. 2002, 32, 3565.
1. Heravi, M.M.; Bakhtiari, K.; Benmorad, T.; Bamoharram, F.F.; Oskooie,
H.A.; Tehrani, M.H. Monatsh. Chem. 2007, 138, 449.
2. Kamal, A.; Kumar, B.A.; Arifuddin, M.; Patrick, M. Ultrasonics Sonochem.
31. Lew, A.; Krutzik, P.O.; Hart, M.E.; Chamberlin, A.R. J. Comb. Chem. 2002,
4, 95.
2
004, 11, 455.
32. Kaddar, H.; Hamelin, J.; Benhaoua, H. J. Chem. Res. 1999, 718.
33. (a) Perreux, L.; Loupy, A. Tetrahedron. 2001, 57, 9199. (b) Lidstr o¨ m, P.;
Tierney, J.; Wathey, B.; Westman, J. Tetrahedron. 2001, 57, 9225.
34. Loupy, A.; Petit, A.; Hamelin, J.; Texier-Boullet, F.; Jacquault, P.; Mathe,
D. Synthesis 1998, 1213.
1
1
1
1
1
3. Shi, M.; Cui, S.C.; Yin, W.P. Eur. J. Org. Chem. 2005, 2379.
4. Heal, M.R.; Harrison, M.A.J.; Cape, J.N. Atmosph. Environ. 2007, 41, 3515.
5. Mallick, S.; Parida, K.M. Catal. Commun. 2007, 8, 1487.
6. Munawar, M.A.; Khalid, M. J. Chem Soc. 2004, 26, 4.
7. Rodrigues, J.A.R.; de Oliveira Fiho, A.P.; Moran, P.J.S.; Custodio, R. Tetra- 35. Deshayes, S.; Liagre, M.; Loupy, A.; Luche, J.L.; Petit, A. Tetrahedron
hedron 1999, 55, 6733. 1999, 55, 10851.

 Kumar, M. Satish
Kumar, M. Satish
 Reddy, K. Rajendar
Reddy, K. Rajendar
 Rajanna
Rajanna
 Venkanna
Venkanna
 Krishnaiah
Krishnaiah