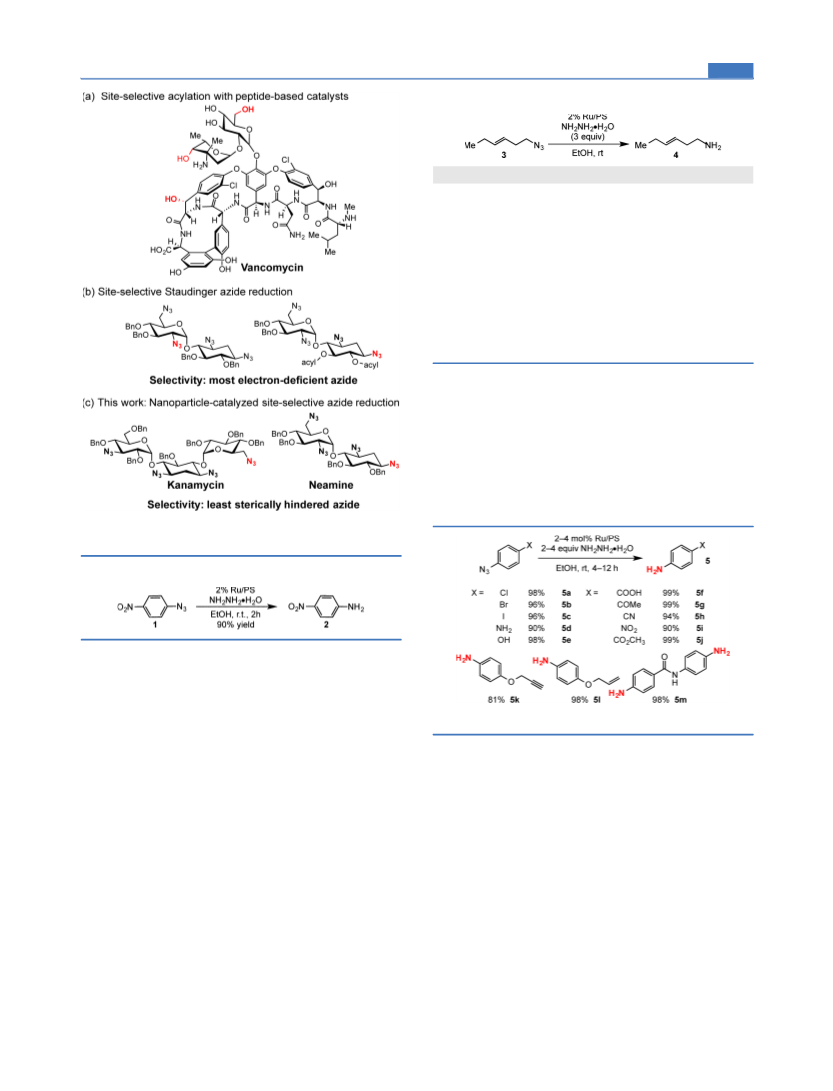
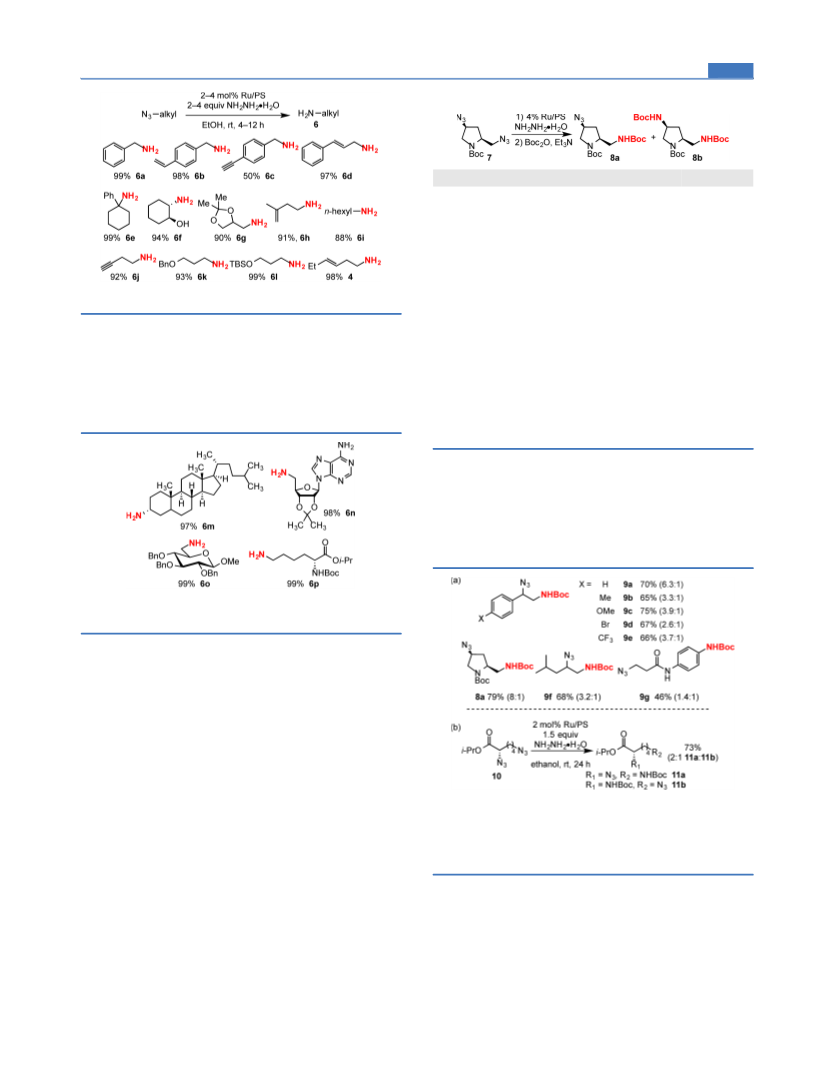
ACS Catalysis
REFERENCES
Letter
(b) Ahammed, S.; Saha, A.; Ranu, B. C. J. Org. Chem. 2011, 76, 7235−
7239.
(19) When the reaction of 6f was halted at partial conversion over
five consecutive recycles, the same partial conversion (47%−56%
conversion, 1 h) was obtained for each run. See the Supporting
■
(1) (a) Robles, O.; Romo, D. Nat. Prod. Rep. 2014, 31, 318−334.
(b) Mahatthananchai, J.; Dumas, A. M.; Bode, J. W. Angew. Chem., Int.
Ed. 2012, 51, 10954−10990. (c) Beale, T. M.; Taylor, M. S. Org. Lett.
2013, 15, 1358−1362. (d) Giuliano, M. W.; Miller, S. J. Top. Curr.
Chem. 2016, 372, 157−201.
(20) Knouzi, N.; Vaultier, M.; Carrie,
815−819.
́
R. Bull. Soc. Chim. Fr. 1985, 5,
(2) O’Connell, K. M. G.; Hodgkinson, J. T.; Sore, H. F.; Welch, M.;
Salmond, G. P. C.; Spring, D. R. Angew. Chem., Int. Ed. 2013, 52,
10706−10733.
(3) (a) Lewis, C. A.; Miller, S. J. Angew. Chem., Int. Ed. 2006, 45,
5616−5619. (b) Lewis, C. A.; Longcore, K. E.; Miller, S. J.; Wender, P.
A. J. Nat. Prod. 2009, 72, 1864−1869. (c) Yoshida, K.; Furuta, T.;
Kawabata, T. Tetrahedron Lett. 2010, 51, 4830−4832. (d) Ueda, Y.;
Mishiro, K.; Yoshida, K.; Furuta, T.; Kawabata, T. J. Org. Chem. 2012,
77, 7850−7857. (e) Yoganathan, S.; Miller, S. J. J. Med. Chem. 2015,
58, 2367−2377.
(4) Han, S.; Miller, S. J. J. Am. Chem. Soc. 2013, 135, 12414−12421.
(5) (a) Pathak, T. P.; Miller, S. J. J. Am. Chem. Soc. 2012, 134, 6120−
6123. (b) Pathak, T. P.; Miller, S. J. J. Am. Chem. Soc. 2013, 135,
8415−8422.
(6) (a) Fowler, B. S.; Laemmerhold, K. M.; Miller, S. J. J. Am. Chem.
Soc. 2012, 134, 9755. (b) Lee, D.; Williamson, C. L.; Chan, L.; Taylor,
M. S. J. Am. Chem. Soc. 2012, 134, 8260−8267.
(7) (a) Nyffeler, P. T.; Liang, C.-H.; Koeller, K. M.; Wong, C.-H. J.
Am. Chem. Soc. 2002, 124, 10773−10778. (b) Li, J.; Chen, H.-N.;
Chang, H.; Wang, J.; Chang, C.-W. T. Org. Lett. 2005, 7, 3061−3064.
(c) Li, J.; Chiang, F.-I.; Chen, H.-N.; Chang, C.-W. T. J. Org. Chem.
2007, 72, 4055−4066.
(8) (a) Liang, F. S.; Wang, S.-K.; Nakatani, T.; Wong, C.-H. Angew.
Chem., Int. Ed. 2004, 43, 6496−6500. (b) Li, J.; Wang, J.; Czyryca, P.
G.; Chang, H.; Orsak, T. W.; Evanson, R.; Chang, C.-W. T. Org. Lett.
2004, 6, 1381−1384.
(9) Organic Azides: Synthesis and Applications; Brase, S., Banert, K.,
̈
Eds.; John Wiley & Sons: West Sussex, U.K., 2010.
(10) Maddani, M. R.; Moorthy, S. K.; Prabhu, K. R. Tetrahedron
2010, 66, 329−333.
(11) (a) Gross, E.; Toste, F. D.; Somorjai, G. A. Catal. Lett. 2015,
145, 126−138. (b) Nanocatalysis: Synthesis and Applications;
Polshettiwar, V., Asefa, T., Eds.; Wiley: Hoboken, NJ, 2013.
(c) Cong, H.; Porco, J. A., Jr. ACS Catal. 2012, 2, 65−70.
(d) Yasukawa, T.; Miyamura, H.; Kobayashi, S. Chem. Soc. Rev.
́
2014, 43, 1450−1461. (e) Pachon, L. D.; Rothenberg, G. Appl.
Organomet. Chem. 2008, 22, 288−299. (f) Astruc, D. In Nanoparticles
and Catalysis; Astruc, D., Ed.; Wiley−VCH: Weinheim, Germany,
2008; pp 1−48. (g) Astruc, D.; Lu, F.; Ruiz Aranzaes, J. Angew. Chem.,
Int. Ed. 2005, 44, 7852−7872. (h) Chng, L. L.; Erathodiyil, N.; Ying, J.
Y. Acc. Chem. Res. 2013, 46, 1825−1837.
(12) (a) Akiyama, R.; Kobayashi, S. Angew. Chem., Int. Ed. 2001, 40,
3469−3471. (b) Kobayashi, S.; Akiyama, R. Chem. Commun. 2003,
449−460. (c) Akiyama, R.; Kobayashi, S. J. Am. Chem. Soc. 2003, 125,
3412−3413. (d) Nishio, R.; Sugiura, M.; Kobayashi, S. Org. Lett. 2005,
7, 4831−4834. (e) Miyamura, H.; Matsubara, R.; Miyazaki, Y.;
Kobayashi, S. Angew. Chem., Int. Ed. 2007, 46, 4151−4154.
(13) Yasukawa, T.; Suzuki, A.; Miyamura, H.; Nishino, K.; Kobayashi,
S. J. Am. Chem. Soc. 2015, 137, 6616−6623.
(14) (a) Shu, X.-Z.; Nguyen, S. C.; He, Y.; Oba, E.; Zhang, O.;
Canlas, C.; Somorjai, G. A.; Alivisatos, A. P.; Toste, F. D. J. Am. Chem.
Soc. 2015, 137, 7083−7086. (b) Gross, E.; Liu, J. H.; Alayoglu, S.;
Marcus, M. A.; Fakra, S. C.; Toste, F. D.; Somorjai, G. A. J. Am. Chem.
Soc. 2013, 135, 3881−3886.
(15) Schmidt, E.; Vargas, A.; Mallat, T.; Baiker, A. J. Am. Chem. Soc.
2009, 131, 12358−12367.
(16) Udumula, V.; Tyler, J. H.; Davis, D. L.; Wang, H.; Linford, M.
R.; Minson, P. S.; Michaelis, D. J. ACS Catal. 2015, 5, 3457−3462.
(17) (a) Hemantha, H. P.; Sureshbabu, V. V. Org. Biomol. Chem.
2011, 9, 2597−2601. (b) Cantillo, D.; Moghaddam, M. M.; Kappe, C.
O. J. Org. Chem. 2013, 78, 4530−4542.
(18) (a) Guo, H.; Yan, X.; Zhi, Y.; Li, Z.; Wu, C.; Zhao, C.; Wang, J.;
Yu, Z.; Ding, Y.; He, W.; Li, Y. Nano Res. 2015, 8, 1365−1372.
4427
ACS Catal. 2016, 6, 4423−4427

 Udumula, Venkatareddy
Udumula, Venkatareddy
 Nazari, S. Hadi
Nazari, S. Hadi
 Burt, Scott R.
Burt, Scott R.
 Alfindee, Madher N.
Alfindee, Madher N.
 Michaelis, David J.
Michaelis, David J.