
Synthetic Communications p. 3272 - 3280 (2013)
Update date:2022-08-11
Topics:
 Waghmode, Suresh B.
Waghmode, Suresh B.
 Mahale, Ganesh
Mahale, Ganesh
 Patil, Viraj P.
Patil, Viraj P.
 Renalson, Kartik
Renalson, Kartik
 Singh, Dharmendra
Singh, Dharmendra
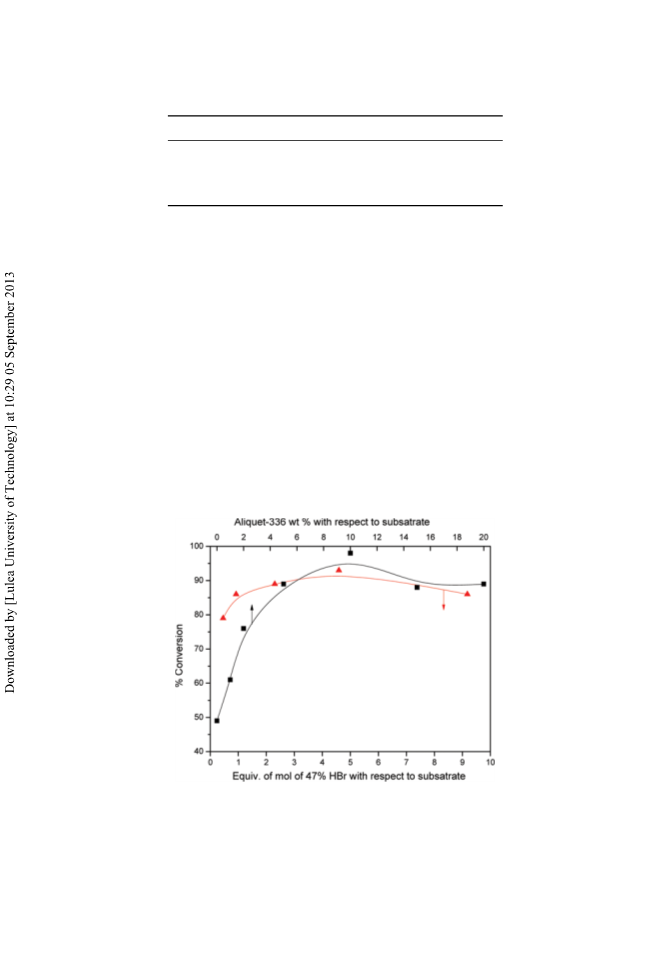
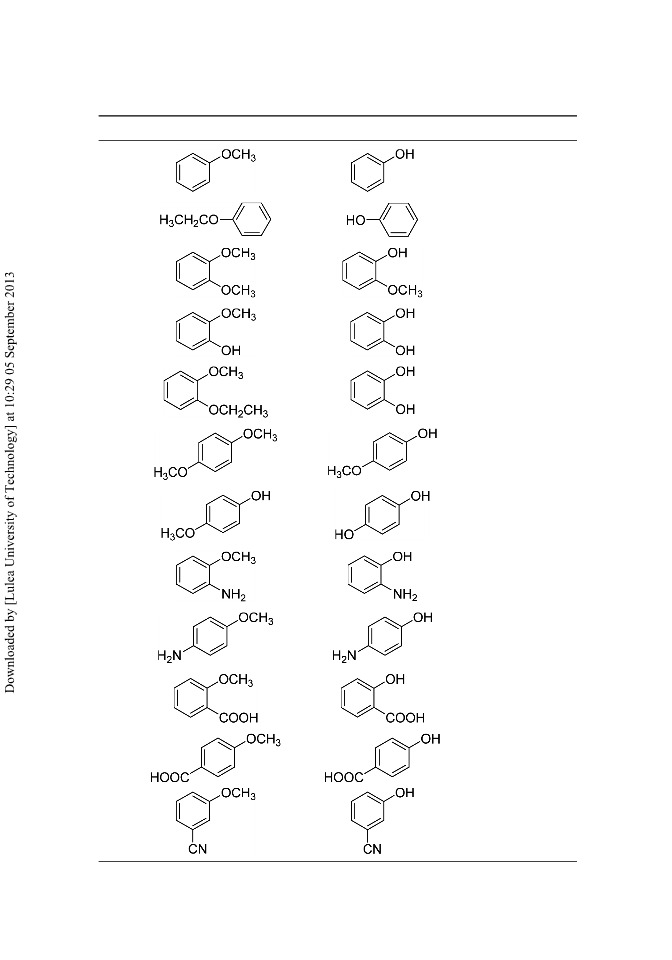
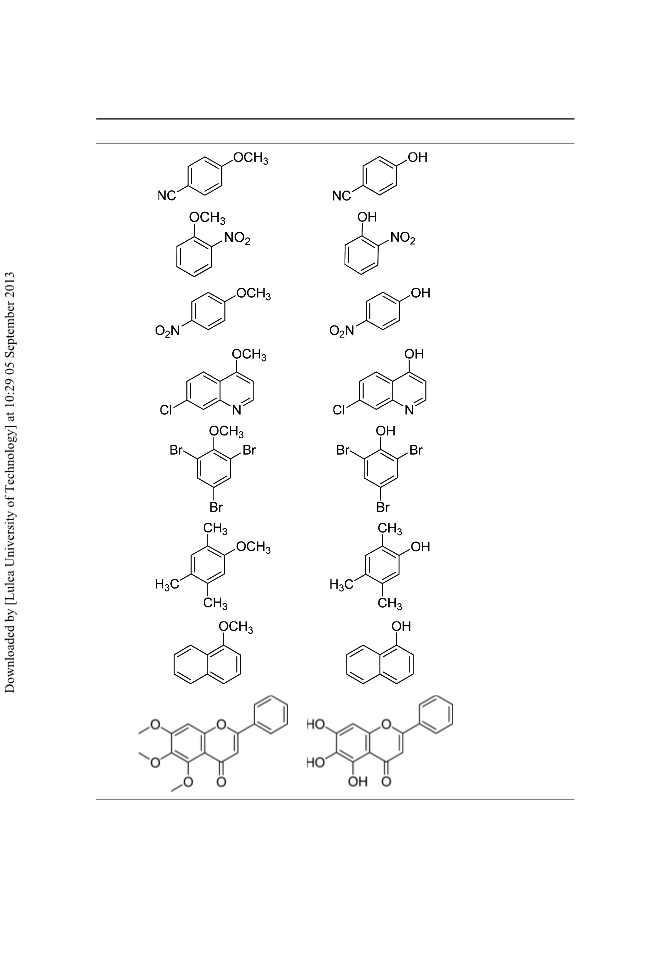
A rapid method for selective cleavage of aryl methylethers can be achieved in the presence of a protic acid and a catalytic amount of phase-transfer catalyst (Aliquat-336). Aliquat-336 accelerates the rate of reaction and affords the corresponding phenols in excellent to good yields on a wide variety of substrates. [Supplementary materials are available for this article. Go to the publisher's online edition of Synthetic Communications for the following free supplemental resource(s): Full experimental and spectral details.]
View More







Anhui Asahikasei Chemical Co., Ltd
Contact:86-551-4259770
Address:No. 88 Linquan Road Hefei Anhui China
Contact:86-536-8866010
Address:NO.4455 EAST FUSHOU ST. WEIFANG DEVELOPMENT ZONE, SHANDONG, CHINA
Contact:+91 9963263336
Address:Plot#146A, IDA Mallapur, Hyderabad - 500072
Shanghai Hanhong Scientific Co.,Ltd.
website:http://www.chemvia.com
Contact:+86-21-64541543,54280654,13918533501
Address:Jiachuan Road 245
ShiJiaZhuang Dowell Chemical Co.,Ltd.
website:http://www.dowechem.com
Contact:+86-13463963265,+86-311-89805679
Address:Xiyangling village, high tech Zone, Shijiazhuang,Hebei, China
Doi:10.1016/j.bmc.2016.11.057
(2017)Doi:10.1016/S0040-4020(02)00008-X
(2002)Doi:10.1016/S0040-4039(00)87634-0
(1982)Doi:10.1021/ol034217k
(2003)Doi:10.1007/BF02962216
(1935)Doi:10.1016/S0957-4166(98)00384-X
(1998)