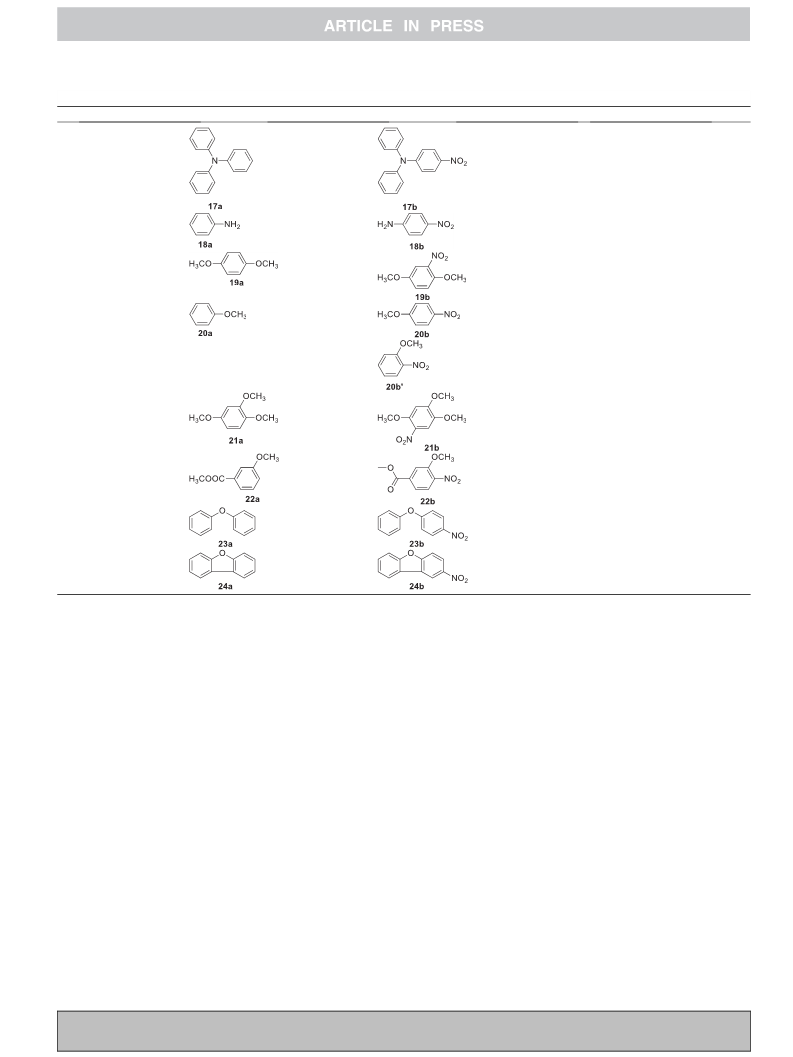2
P. Natarajan et al. / Tetrahedron Letters xxx (xxxx) xxx
profile of TS-N-IL measured under argon atmosphere exhibited
endotherm at 74 °C possibly due to the loss of NO group. These
results recommended that TS-N-IL is nonvolatile and stable up to
150 °C. Therefore, TS-N-IL is expected to decrease potential
exposure and known toxicity during use, while compared to TBN
has a low flash- and boiling points.
Later, we investigated the nitrating capacity of novel TS-N-IL
(Scheme 2). A reaction of 4-methylphenol (1a, 1 equiv.) and
TS-N-IL (2.0 equiv.) under an open-air atmosphere at room tem-
perature, after stirring the reaction mixture for 2 h, afforded the
expected 4-methyl-2-nitrophenol (1b) in quantitative yield
(Table 1 and entry 1). The product was characterized by melting
point and NMR spectroscopy (ESI), which are in close agreement
with the literature reports [3]. The bisnitrated product (4-
methyl-2,6-dinitrophenol, 1c) was possibly watched if the sub-
strate was left under the reaction conditions until well after the
beginning material was completely devoured, and then just in little
amounts (Table 1 and entry 2). The use of acetonitrile, DMF, etc., as
an additional solvent; the reaction continued at a slower rate
(Table 1 and entries 3–6). Reaction under oxygen-free conditions
is relatively sluggish (Table 1 and entry 7). Additionally, an endea-
vor to increase the reaction rate by raising the temperature to 50 °C
got failed (Table 1 and entries 8–9). With regard to the optimal
amount of TS-N-IL, the reaction in the presence of 1.3 equivalents
gave the best outcomes (Table 1 and entries 10–13). Also, no
discernible 4-methyl-2,6-dinitrophenol (1c) emerged even after
6 h with the usage of 1.3 equiv., of TS-N-IL, cf. Table 1 and entry
14. Thence, the optimized reaction conditions were as follows:
1a (1.0 equiv.,) and TS-N-IL (1.3 equiv.,) under an open air
atmosphere at room temperature for 2 h.
Scheme 1. A general strategy for the synthesis of nitro-group-containing arenes
and drugs using a novel task-specific nitrite-based ionic liquid (TS-N-IL) reported in
this paper. At the bottom is provided the chemical structure of drugs synthesized
using TS-N-IL.
tolcapone, niclofolan, flutamide, niclosamide and nitrazepam
(Scheme 1). Nonvolatile nature, easy synthesis, merely stoichio-
metric need and mildness are few advantages of TS-N-IL while
contrasted with tert-butyl nitrite a well-known and a toxic reagent
utilized in organic synthesis.
Next, we treated a series of phenols with TS-N-IL and the results
are compiled in Table 2. We found that under optimized reaction
conditions (Table 1), TS-N-IL was compelling at changing over
phenol and its derivatives into corresponding C-nitro compounds
in good yield (Table 2). At the point when both ortho and para
positions are available (Table 2 and entries 2 and 3), a mixture of
mononitrated regioisomers was acquired. It very well may be seen
from the proportion of 2b and 2b0 and 3b and 3b0 that the para-
isomer is more effectively created than the ortho-isomer.
Nevertheless, phenol with substituent just in the para-position
(1a, 4a and 5a) solely afforded corresponding ortho-nitro product
(1b, 4b and 5b) in quantitative yield. Also, salicyclic acid (6a), 2-
naphthol (7a) and 6-bromo-2-naphthol (8a) under the optimized
conditions afforded the expected nitro compounds 6b, 7b and 8b,
respectively, in good yield (Table 2). These outcomes demonstrated
that an electronic effect on the substituted group played not
significant role in the reaction, which precludes the possibility of
an electrophilic substitution reaction [3].
Results and discussion
The nitrite functionalized ionic liquid, i.e., TS-N-IL was prepared
in three steps by i) alkylation of ethyl nicotinate with 6-bromo-2-
methylhexan-2-yl acetate at reflux temperature, ii) nitrification of
resultant ionic liquid (IL, Scheme 2) with aqueous NaNO2 and HCl
at 0 °C and iii) anion exchange with lithium trifluoromethylsulfon-
imide, cf. Scheme 2. The synthesized TS-N-IL was characterized by
FT-IR, NMR, TGA, DSC and elemental analysis, cf. Electronic
Supporting Information (ESI). The IR spectra of TS-N-IL exhibit an
intense peak at 1643 cmꢀ1 corresponding to the asymmetrical
stretching of nitrite ester group, peaks between 1636–1489 cmꢀ1
and 1418–1396 cmꢀ1, respectively, are due to CAC multiple bond
stretching and aromatic CAN vibrations. The aromatic and alkyl
CAH stretching frequencies appear in the ranges of 3150–3080
and 3000–2865 cmꢀ1, respectively. Additionally, the sharp peak
shows the carbonyl stretching frequency of the ester at
1710 cmꢀ1. TGA curve of TS-N-IL measured under argon atmo-
sphere showed an initial ~6% weight loss near to ~74 °C due to
the removal of NO and was followed by a shouldering from 140
to 290 °C with maximum decomposition of organic moiety. DSC
Encouraged by these results, we then directed our concentra-
tion toward the nitration of amides using TS-N-IL as a reagent. A
reaction of 4-methyl-N-phenylbenzenesulfonamide (9a) with
TS-N-IL under the same experimental conditions (Table 1) utilized
for nitration of phenols resulted in incomplete conversion. Viz.,
Scheme 2. The synthetic route to task-specific nitrite-based ionic liquid (TS-N-IL).
Please cite this article as: P. Natarajan, R. Chaudhary, N. Rani et al., 3-(Ethoxycarbonyl)-1-(5-methyl-5-(nitrosooxy)hexyl)pyridin-1-ium cation: A green
alternative to tert-butyl nitrite for synthesis of nitro-group-containing arenes and drugs at room temperature, Tetrahedron Letters, https://doi.org/

 Chaudhary, Renu
Chaudhary, Renu
 Natarajan, Palani
Natarajan, Palani
 Rani, Neetu
Rani, Neetu
 Sakshi
Sakshi
 Venugopalan, Paloth
Venugopalan, Paloth