2
M. Rojas et al. / Journal of Molecular Liquids 310 (2020) 113206
based on pyridinium, imidazolium and cholinium cations and varying
alkyl chain from 2 to 16 carbon atoms (C2-C16). They demonstrated
that the length of the alkyl chain and lipophilicity parameter for these
cations (log k0) are important predictors of ecotoxicity of these surfac-
tants [14]. In particular for the surfactants based on imidazolium cat-
ions, the inclusion of functional groups (including heteroatoms O and
N) into the structure of the imidazolium ring can reduce toxicity and im-
prove biodegradability of them, for example the substitution with esters
and amide functional groups gave surfactants which presented lower
toxicity [28].
reactivities in these micellar systems can be expected. This also com-
prises a promising tool for strategically modulating desired reaction
pathways.
2. Experimental
2.1. Reagents and materials
L-Alanine, L-Proline and L-Phenylalanine were high quality and
were purchased from Sigma Aldrich and used as received without fur-
ther purification. Amberlite IRA400Cl, N-methylimidazole and 1-
tetradecyl bromide, were purchased from Sigma Aldrich and used as re-
ceived. Doubly distilled deionized water was obtained from a Millipore
Milli-Q water purification system (Millipore) and the resistance was
18.2 MΩ. Solutions were prepared with doubly distilled Millipore
Milli-Q water. Borate buffer and britton-robinson buffer were prepared
according to reported literature [34,35].
On the other hand, halogenated anions ([Br]− and [Cl]−) and
perfluorinated anions such as [NTF2]−, [BF4]− and [PF6]− have also
been proved to be hazardous due to their hydrolytically unstable prop-
erties. Most of these anions are thermally, chemically, and hydrolytically
stable and, so, are presumably resistant to degradation [3].
Considering above, the synthesis of surfactants from building blocks
derived from renewable sources seems to be a promising proposal in
order to improve the green credentials of the surfactants. In this context,
the amino acids (AAs), one of the most abundant sources in nature and
more susceptible to microbial degradation, would be excellent feed-
stock for the synthesis of new surfactants [29]. In addition, these groups
are also useful by their ability to introduce functional group(s) [30].
Lately, studies about toxicity and biodegradability of surfactants
with L-phenylalanine and L-proline as counterions, have been reported
[14,31,32]. Nevertheless, the micellar effect of these surfactants based
on amino acids has not been reported yet.
Considering both challenges, the development of surfactants with
low toxicity and the development of micellar systems to conduct or-
ganic transformations in aqueous medium instead of organic solvents
[33], in this work, as a first approached, we have examined the micellar
properties of a new class of surfactants based on imidazolium using 1-
Tetradecyl-3-methylimidazolium cation ([C14mim]+) and changing
the nature of the counterion by AAs, like L-alanine (L-Ala), L-proline
(L-Pro) and L-phenylalanine (L-Phe), shown in Table 1. Moreover, we
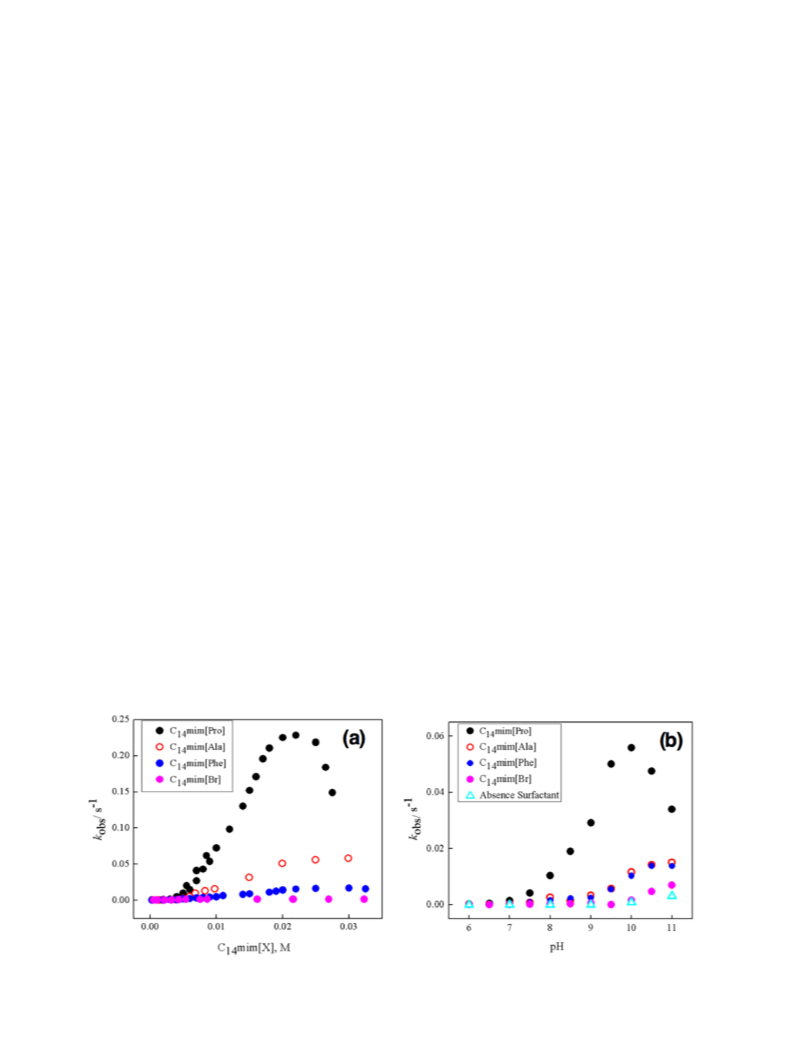
studied kinetically the micellar effect, on a classical organic reaction
such as a hydrolysis reaction. Therefore, we studied the reaction of 4-
nitrophenyl acetate (NPA) in presence of these surfactants based on
imidazolium. In order to compare the micellar effect promoted by the
surfactants with organic counterion against a traditional counterion
we have included in this study a surfactant with a simple bromide as
anion (see Table 1). Considering the presence of functionalized anions
in the structure of these surfactants based on imidazolium, novel
2.2. Synthetic procedures
Synthesis of [C14mim][Br] surfactant. The 1-Tetradecyl-3-
methylimidazolium bromide was synthesized according to reported
procedure [9], by reacting N-methylimidazole (0.1 mol) with 1-
tetradecyl bromide (0.1 mol) in toluene for 24 h, at 80 °C. The resulting
[C14mim][Br] was recrystallized from ethyl acetate five times in order to
obtain colorless solid, in 97% yield. The product was dried in vacuum for
2 days prior to use.
1NMR (CDCl3) δ, ppm: 9,86 (s, 1H), 7,55 (d, J = 2,0 Hz, 1H), 7,36 (d,
J = 1,8 Hz, 1H), 4,11 (t, J = 7,4 Hz, 2H), 3,90 (s, 3H), 1,71 (h, J = 8,5 Hz,
7,2 Hz, 2H), 1,02 (s, 22H), 0,74–0,57 (m, 3H). NMR signals according to
reported literature [9].
Synthesis of [C14mim][AA], surfactants AA (amino acids) = L-Al-
anine ([L-Ala]), L-Proline ([L-Pro]) and L-Phenylalanine ([L-Phe]).
The 1-Tetradecyl-3-methylimidazolium amino acids derivatives were
synthetized using an ionic exchange column, in the presence of
Amberlite®. Prior to exchanging the bromide anion with AA anions,
the column was activated in presence of NaOH, then [C14mim][Br]
(0.02 mol) was eluded drop wise and was collected in an equimolar
amino acid solution. The reaction mixture was then stirred for 24 h
until complete reaction. The aqueous solution was removed under re-
duce pressure.
Finally, the products were purified in presence of acetonitrile/meth-
anol mixture, then, the resulting [C14mim][AA] products were filtrated
and the mixture solvents were removed under reduce pressure to ob-
tain 94% yield. The products were dried in vacuum for 2 days prior to
use.
Table 1
Surfactants synthetized and reaction studied of substrate 4-nitrophenyl acetate (NPA) in
presence of micelles.
1-Tetradecyl-3-methylimidazolium alanine ([C14mim][Ala] sur-
factant): 1H-NMR (CDCl3) δ, ppm: 8.86 (s, 1H), 7.48 (d, J = 1.9 Hz,
1H), 7.42 (d, J = 1.9 Hz, 1H) 4.16 (t, 2H), 3.85 (s, 3H), 3.50 (q, J =
7.2 Hz, 1H), 1.79 (t, J = 28.2 Hz, 2H), 1.30 (d, 3H), 1.25 (s, 4H), 1.17
(s, J = 7.9 Hz, 18H), 0.75 (t, J = 6.5 Hz, 3H).
1-Tetradecyl-3-methylimidazolium proline ([C14mim][Pro] sur-
factant): 1H-NMR (CDCl3) δ, ppm: 9.35 (s, 1H), 8.00 (d, 2H), 4.71 (t,
J = 7.4 Hz, 2H), 4.43 (d, J = 1.8 Hz, 3H), 4.31 (t, J = 7.5 Hz, 1H), 3.78
(dt, J = 12.5 Hz, 6.8 Hz, 1H), 3.59 (dd, J = 12.1 Hz, 6.3 Hz, 1H),
2.74–2.65 (m, 1H), 2.39 (m, J = 25.7 Hz, 19.6 Hz, 6.4 Hz, 6H), 1.84 (s,
22H), 1.49–1.40 (m, 3H).
1-Tetradecyl-3-methylimidazolium phenylalanine ([C14mim]
[Phe] surfactant): 1H-NMR (CDCl3) δ ppm: 7.50 (s, 1H), 7.39 (s, 1H),
7.30 (d, J = 3.8 Hz, 4H), 7.20 (d, J = 4.0 Hz, 1H), 4.12 (t, J = 7.3 Hz,
2H), 3.88 (s, 3H), 3.71–3.65 (m, 1H), 3.16 (dd, J = 13.9, 4.9 Hz, 1H),
2.88 (dd, J = 13.8, 8.2 Hz, 1H), 1.76 (s, 2H), 1.33–1.16 (m, 22H), 0.88
(t, J = 6.2 Hz, 3H).
Full spectra are shown in Figs. S1–S4 in the Supporting information.
The water content for the [C14mim][Br] and [C14mim][AA]s was b3 wt%
and was determined using Karl Fisher titration (TitroMatic).

 Figueroa, Roberto
Figueroa, Roberto
 Orth, Elisa
Orth, Elisa
 Pavez, Paulina
Pavez, Paulina
 Rojas, Mabel
Rojas, Mabel
 Santos, José G.
Santos, José G.