DOI: 10.1002/anie.201007389
Enzyme Assays
Detection of Enzyme Activity through Catalytic Signal Amplification
with Functionalized Gold Nanoparticles**
Renato Bonomi, Alessandro Cazzolaro, Anna Sansone, Paolo Scrimin,* and Leonard J. Prins*
The detection of low levels of proteins and other biomarkers
is of crucial importance for the early diagnosis of diseases.[1]
The development of chemical-sensing methodologies as an
alternative to biological assays is of strong current interest,
because such methods involve simple detection protocols. In
addition, such systems can be adapted through straightfor-
ward structural modifications for use with a wide variety of
targets.[2–5] Nevertheless, a common feature of these assays is
that the amount of generated signal is proportional to the
amount of substrate converted by the enzyme. The sensitivity
of such assays would be significantly increased if the
enzymatic conversion of a single substrate molecule led to
the formation of a multitude of reporter molecules through a
cascade of chemical events, each of which magnified the
previous event. Examples of chemical systems able to amplify
originally weak input signals have been reported.[6–9] Herein,
we report the application of a catalytic amplification process
for the detection of proteases. The strategy relies on a cascade
of two catalytic events for signal generation, whereby a gold
nanoparticle covered with a catalytic self-assembled organic
monolayer (Au-MPC) has a crucial central role. In the first
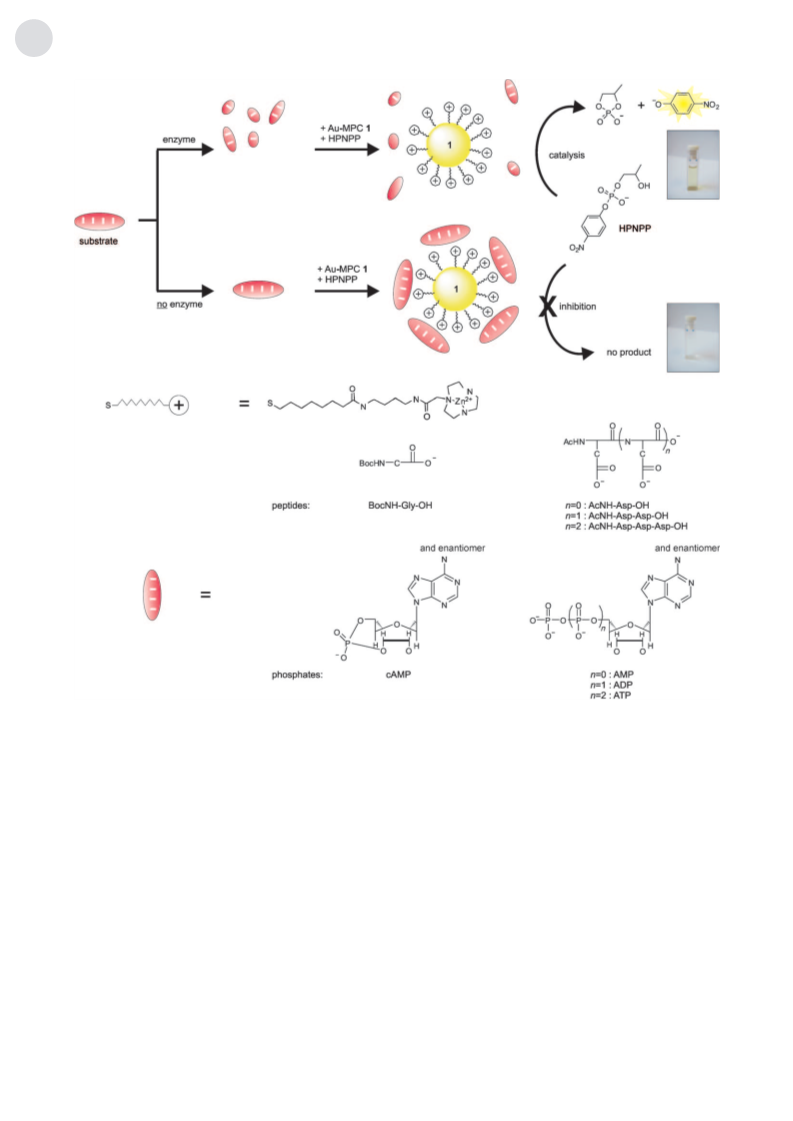
event, an enzyme hydrolyzes a peptide substrate, which acts
as an inhibitor for the catalytic monolayer (Figure 1). Upon
hydrolysis, the catalytic activity of the monolayer is restored,
and large quantities of a yellow reporter molecule are
produced. The Au nanoparticles are important for two
reasons. First, they enable the facile, spontaneous formation
of dinuclear catalytic sites on the periphery of the mono-
layer.[10] Second, their multivalent nature permits the occur-
rence of multipoint interactions with (biological) targets.[11]
The latter aspect, together with the intrinsic physical and
chemical properties of the nanoparticles and the ease of their
preparation and functionalization,[12] has led to extensive
development of assays based on Au nanoparticles that also
occasionally rely on various forms of signal amplification.[13–20]
Previously, we showed that Au-MPC 1 catalyzes the
transphosphorylation of 2-hydroxypropyl-4-nitrophenyl
phosphate (HPNPP) highly efficiently.[10] HPNPP is an
activated RNA model substrate. Detailed kinetic studies
revealed that catalysis results from the cooperative action of
two triazacyclononane·ZnII (TACN·ZnII) complexes localized
on the surface of the monolayer.[21,22] Au-MPC 1, which is
fully covered with the TACN·ZnII complex, displays enzyme-
like saturation behavior with the “overall” values kcat = 6.7 ꢀ
10À3 sÀ1 and KM = 0.31 mm at pH 7.5 in H2O.[23] This system is
intriguing for the following reasons: a) under the experimen-
tal conditions, there is practically no background reaction,
since kuncat under the same conditions is of the order of
10À7 sÀ1; b) the reaction can be monitored visibly by measur-
ing the absorbance of the p-nitrophenol product at 400 nm;
c) a surprisingly high affinity is observed for the binding of
HPNPP to 1. Since Au-MPC 1 has a multitude of positively
charged TACN·ZnII complexes on its surface, we anticipated
that the system would have a high affinity for oligoanions
owing to multivalent interactions. This hypothesis was also
supported by the contributions by Hamachi and co-workers,
who demonstrated that oligophosphates and oligoaspartates
bind a bis(zinc(II) dipicolylamine) complex with very high
affinity.[24–26] In our system, such oligoanions would act as
competitive inhibitors for HPNPP and thus turn off the
catalytic activity of the system.
To verify whether we could use the catalytic production of
p-nitrophenol as a tool to detect binding events on the Au-
MPC surface, we studied two series of biologically important
oligoanions (peptides and phosphates) with negative charges
increasing from 1 to 4. The peptide series comprised BocNH-
Gly-OH (1À), AcNH-Asp-OH (2À), AcNH-Asp-Asp-OH
(3À), and AcNH-Asp-Asp-Asp-OH (4À), and the phosphate
series cAMP (1À), AMP (2À), ADP (3À) and ATP (4À;
Figure 1). Increasing amounts of each compound were added
to a solution of Au-MPC 1 in H2O buffered at pH 7.0 at 408C
with the concentration of TACN·ZnII headgroups equal to
5 mm. This value implies a Au-MPC 1 concentration of around
100 nm, on the basis of the knowledge that a 1.6 nm sized
nanoparticle contains roughly 50 thiols.[27] A kinetic ZnII
titration confirmed that at these concentrations, the ZnII
ions are quantitatively bound to the TACN ligand (see the
Supporting Information). Subsequently, HPNPP was added
to give an initial concentration of 1 mm in the mixture, and the
initial rate of cleavage, ninit, was measured for 30 min by
monitoring the increase in absorbance at 400 nm.[28] A plot of
the initial rate (n1), normalized with respect to the initial rate
in the absence of an inhibitor (n0), as a function of the
concentration of added inhibitor, gave the inhibition curves
depicted in Figure 2 for the peptides (the data for the
[*] Dr. R. Bonomi, A. Cazzolaro, Dr. A. Sansone, Prof. Dr. P. Scrimin,
Dr. L. J. Prins
Department of Chemical Sciences
University of Padova
Via Marzolo 1, 35131 Padova (Italy)
Fax: (+39)049-827-5239
E-mail: paolo.scrimin@unipd.it
[**] Financial support from the European Research Council under the
Seventh Framework Programme (FP7/2007–2013)/ERC of the
European Community (Starting Grant agreement no. 239898) is
acknowledged.
Supporting information for this article is available on the WWW
Angew. Chem. Int. Ed. 2011, 50, 2307 –2312
ꢀ 2011 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
2307

 Bonomi, Renato
Bonomi, Renato
 Cazzolaro, Alessandro
Cazzolaro, Alessandro
 Sansone, Anna
Sansone, Anna
 Scrimin, Paolo
Scrimin, Paolo
 Prins, Leonard J.
Prins, Leonard J.