Tetrahedron Letters
Ruthenium(II)–NNN complex catalyzed Oppenauer-type oxidation
of secondary alcohols
⇑
Qingfu Wang, Wangming Du, Tingting Liu, Huining Chai, Zhengkun Yu
Dalian Institute of Chemical Physics, Chinese Academy of Sciences, 457 Zhongshan Road, Dalian, Liaoning 116023, China
a r t i c l e i n f o
a b s t r a c t
Article history:
Highly efficient Oppenauer-type oxidation of secondary alcohols to the corresponding ketones has been
realized by means of the ruthenium(II) complex catalysts bearing a 2-(benzoimidazol-2-yl)-6-(3,5-dim-
ethylpyrazol-1-yl)pyridine ligand. The oxidation reaction underwent in the presence of acetone as oxi-
dant under mild conditions, reaching final TOF values up to 3960 hÀ1. The hemilability of the ligand is
attributed to the high catalytic activity of these Ru(II) complexes.
Received 31 October 2013
Revised 14 January 2014
Accepted 20 January 2014
Available online 24 January 2014
Ó 2014 Elsevier Ltd. All rights reserved.
Keywords:
Ruthenium(II)
Pyridyl–imidazolyl–pyrazolyl ligand
Oppenauer oxidation
Secondary alcohols
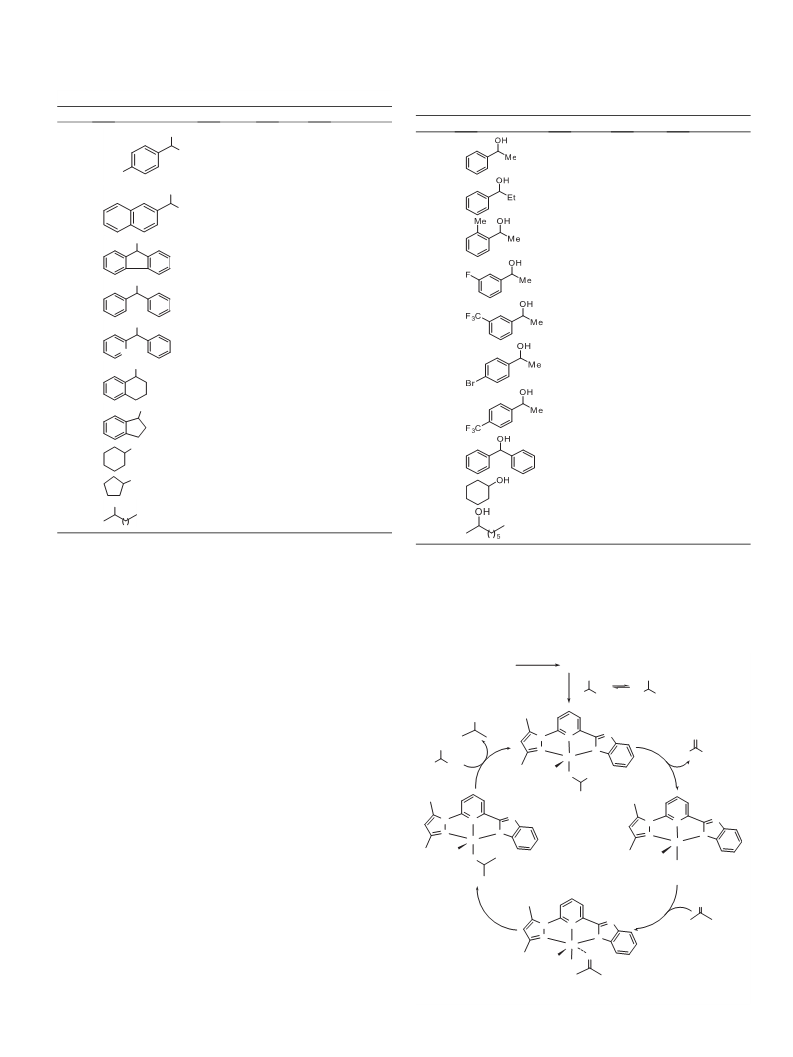
Synthesis of carbonyl compounds is of great importance in or-
ganic synthesis and chemical industry. Oxidation of alcohols to
the corresponding carbonyl compounds provides a practical route
to aldehydes and ketones. Traditional methods usually require
stoichiometric oxidants such as manganese and chromium oxides,1
which are hazardous to the environment. Considering the demand
for green methods, much effort has been devoted to explore the
transition metal catalyzed oxidation of alcohols. In this aspect,
Ru,2 Ir,3 and Fe4-catalysis has attracted considerable interest, and
mild oxidants such as H2O2,5 O2,6 and acetone2–4 have been used.
Acetone is not only employed as the solvent but also as the hydro-
gen acceptor in the homogeneous hydrogen transfer reaction,
which makes it a suitable oxidant in Oppenauer-type oxidation
of secondary alcohols to ketones.
Ruthenium(II) complexes have usually been applied as the most
useful catalysts for transfer hydrogenation (TH) of ketones.7–10
Complex 19 bearing a symmetrical NNN ligand, that is, RuCl2
(PPh3)L (L = 2,6-bis(3,5-dimethylpyrazol-1-yl)pyridine), exhibited
a good catalytic activity for the TH reactions of ketones in refluxing
isopropanol, reaching final TOFs up to 5940 hÀ1, while complex 22c
containing an unsymmetrical NNC ligand showed a higher cata-
lytic activity for the same process (final TOF = 178200 hÀ1) due to
the unsymmetrical environment around the metal center.
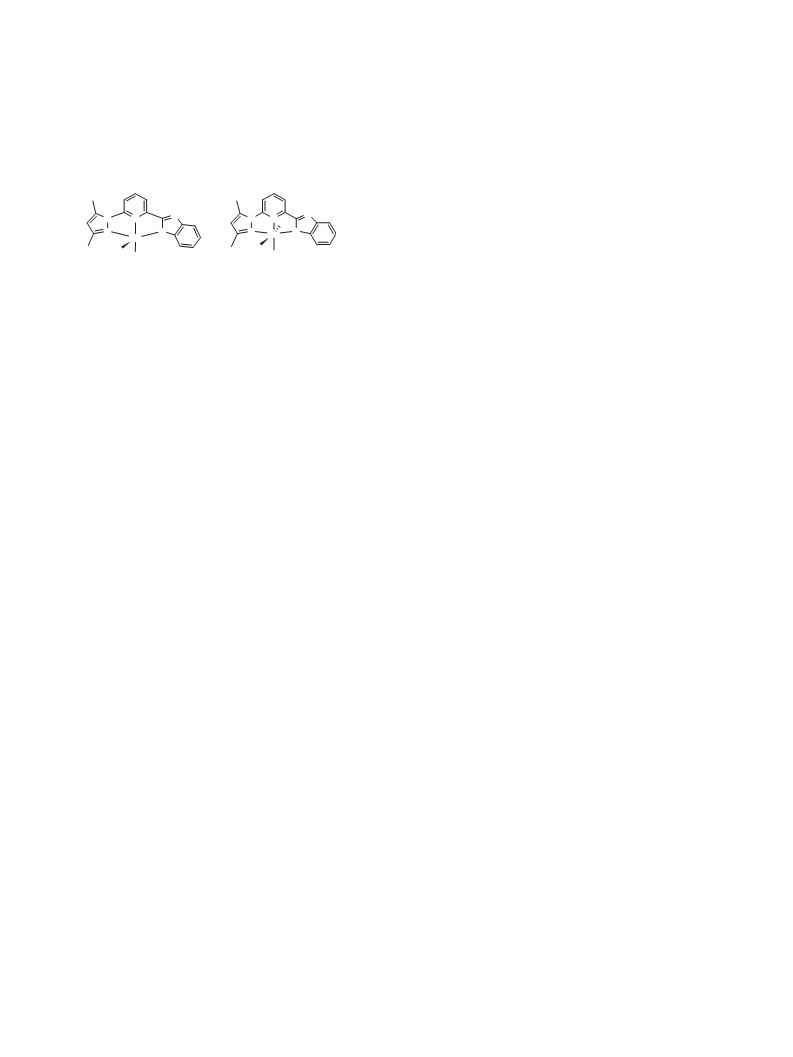
among which complex 310a bearing a pyridyl-based imidazolyl-
pyrazolyl ligand exhibited exceptionally high catalytic activity in
the TH reactions of ketones, reaching final TOFs up to
7.2 Â 105 hÀ1. Hemilability of the ligand and easy convertibility
from coordinately saturated complex 3 to coordinately unsatu-
rated precatalyst 4 under the reaction conditions are attributed
to the high catalytic activities of these complexes (Scheme 1).
We recently found that complex 2 can also effect Oppenauer-
type oxidation of secondary alcohols,2c while complex 1 exhibited
a poor catalytic activity for converting 1-phenylethanol to aceto-
phenone and reached only 11% yield with 0.5 mol % loading during
a period of one hour (Eq. 1). Such a remarkable steric and electronic
effect of the complex catalysts on Oppenauer-type oxidation of
secondary alcohols led us to explore the catalytic behaviors of
complex 3 bearing ligand L10a for the same reaction. To our delight,
3 exhibited a better catalytic activity than complex 2. Herein, we
report Oppenauer-type oxidation of secondary alcohols by means
of complexes 3 and 4 as catalysts.
0.5 mol % Ru(II) cat.
O
O
OH
10 mol % tBuOK
OH
(1)
+
+
o
56 C, N2
Ph
99%
Ph
2c
complex 2 / 5 min:
complex 1 / 1 h:
During the ongoing investigation on Ru(II) complex catalysts for
TH reactions of ketones, we also reported a family of Ru(II)-NNN
complex catalysts supported by a ligand containing an NH moiety,
11%
Initially, oxidation of 1-phenylethanol was conducted for opti-
mization of the reaction conditions (Table 1). By using complex 3
(0.5 mol %) as catalyst and tBuOK (10 mol %) as base in refluxing
⇑
Corresponding author. Tel./fax: +86 411 8437 9227.
0040-4039/Ó 2014 Elsevier Ltd. All rights reserved.

 Wang, Qingfu
Wang, Qingfu
 Du, Wangming
Du, Wangming
 Liu, Tingting
Liu, Tingting
 Chai, Huining
Chai, Huining
 Yu, Zhengkun
Yu, Zhengkun