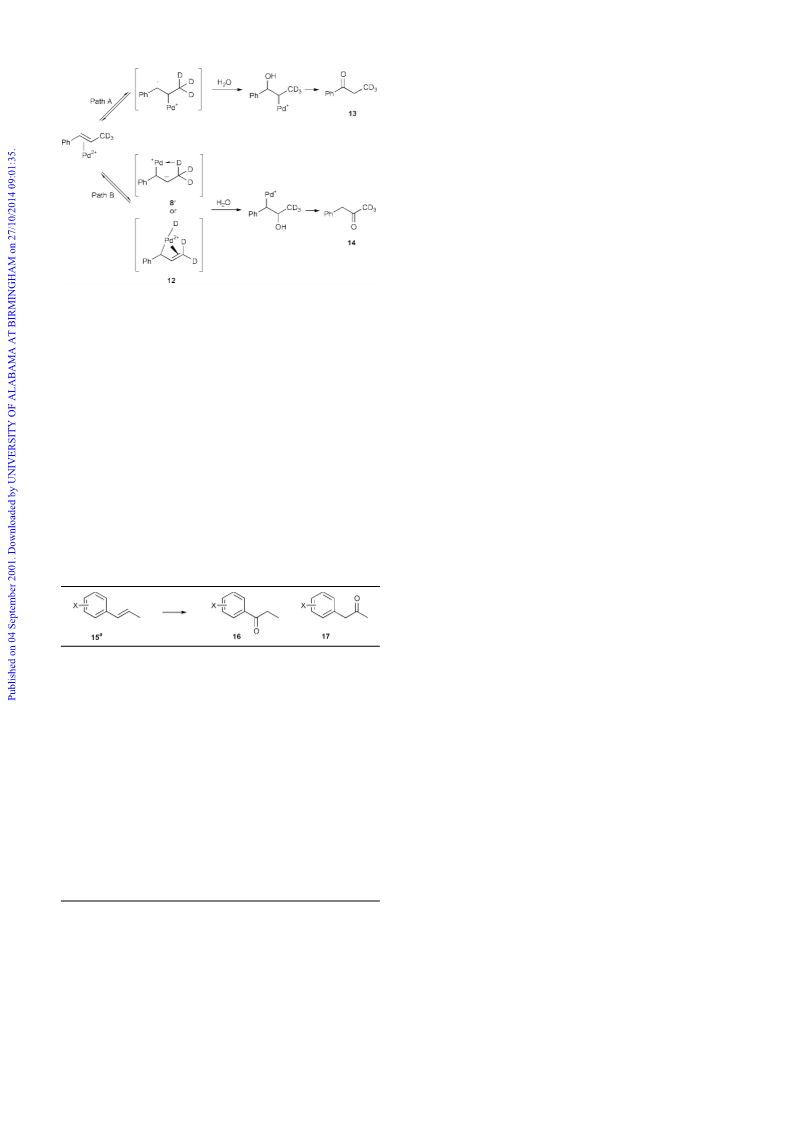
To investigate if the balance between path A (Markovnikov)
and B (anti-Markovnikov) can be influenced by altering the
electronic properties of the aromatic ring, a series of analogues
of b-methylstyrene, 15a–f, were oxidised under standard
Wacker conditions.
The results clearly show that as the electron density of the
aromatic system is increased, path A (Scheme 3) is enhanced,
which is consistent with the greater stabilisation of the
carbocation at the benzylic position (Table 3). The dramatic
influence on the regioselectivity of the oxidation by involve-
ment of the allylic hydrogens is clearly illustrated since path A
only becomes dominant when the aromatic ring is substituted
with three methoxy groups, 15f.
This study provides evidence that the regioselectivity of the
Wacker oxidation can be greatly influenced by the availability
of an allylic hydrogen. The allylic hydrogen might participate in
the Wacker oxidation by being involved in either an agostic C–
H or enyl (s+p) complex. The involvement of these complexes
could account for preferential formation of the anti-Markovni-
kov product when the substrate bears an allylic hydrogen.
We thank The Wellcome Trust (M. J. G.), St. John’s College,
Cambridge (J.-Q. Y) and the Royal Society (J. B. S.) for
funding.
Scheme 3
intermediate since deuterium should either be lost from the
methyl position or partially transferred to the benzylic posi-
tion.10 This also confirms that isomerisation to the terminal
alkene followed by oxidation, which would require the loss of a
deuterium atom from the methyl group, is not the mechanism
that can account for the preferential formation of the anti-
Markovnikov product. It has been shown previously that enyl
Notes and references
1
1
(
s+p) intermediates are precursors for p-allylic complexes,
1
J. Tsuji, in Comprehensive Organic Synthesis, Eds. B. M. Trost and I.
Fleming, Pergamon Press, New York, 1991, Vol. 7, pp. 449–467.
and therefore may be formed in the Wacker reaction. While the
p-allylic complex 11 is probably too stable to react with water,
it is possible that an enyl (s+p) complex 12 with the proton still
attached to the palladium could be intramolecularly reproton-
ated allowing this intermediate to react with water (Scheme 3).
The labelling studies are therefore consistent with either a
mechanism involving the stabilisation of the anti-Markovnikov
intermediate occurring through an agostic C–H 8 or a
mechanism where the intermediate is stabilised by an enyl
2 O. Hamed, C. Thompson and P. M. Henry, J. Org. Chem., 1997, 62,
7082; O. Hamed and P. M. Henry, Organometallics, 1997, 16, 4903; K.
Zaw and P. M. Henry, Organometallics, 1992, 11, 2008; B. Akermark,
B. C. Soderberg and S. S. Hall, Organometallics, 1987, 6, 2608; J. E.
Bäckvall, B. Akermark and S. O. Ljunggren, J. Am. Chem. Soc., 1979,
1
01, 2411.
3
4
5
B. L. Feringa, J. Chem. Soc., Chem. Commun., 1986, 909; H. Ogawa, S.
Miyamoto, T. Mandai, S. Wakabayashi and J. Tsuji, Tetrahedron Lett.,
1
988, 29, 5181; H. Pellissier, P. Y. Michellys and M. Santelli,
(
s+p) complex 12 that can be intramolecularly reprotonated.
Tetrahedron, 1997, 53, 7577.
E. Keinan, K. K. Seth and R. Lamed, J. Am. Chem. Soc., 1986, 108,
3474; J. Tsuji, in Comprehensive Organic Synthesis, Eds. B. M. Trost
and I. Fleming, Pergamon Press, New York, 1991, Vol. 7, pp
Table 3 Probing electronic factors in the oxidation of b-methylstyrenes
4
65–466.
The reaction was monitored by GC which determined that approx-
imately 1% of the terminal double bond was formed by double bond
shift during the reaction course. This eliminates the possibility that the
major pathway for the formation of the anti-Markovnikov product is
from oxidation of the terminal alkene (also see the deuterium labeling
studies with b-methylstyrene later in the text).
a
b
c
X = 4-CF
X = 4-H
X = 4-CH
X = 2-OCH
X = 4-OCH
X = 2,4-OCH
X = 2,4,6-OCH
3
1
1
1
1
1
1.2
2.3
> 19
7.5
3
3.8
2.7
2.0
1
6 M. Brookhart and M. L. H. Green, J. Organometal. Chem., 1983, 250,
395.
d
3
3
b
7 R. H. Crabtree, Angew. Chem., Int. Ed. Engl., 1993, 32, 799.
8 B. M. Trost and P. J. Metzner, J. Am. Chem. Soc., 1980, 102, 3572; H.
Grennberg, V. Simon and J. E. Bäckvall, J. Chem. Soc., Chem.
Commun., 1994, 265; R. G. Brown, R. V. Chaudhari and J. M.
Davidson, J. Chem. Soc., Dalton Trans., 1977, 176.
e
f
3
3
1
a
Representative procedure for the Wacker Oxidations of methylstyrenes
5a–f. A flask containing a suspension of palladium(II) chloride (35.4 mg,
.2 mmol) and copper(I) chloride (198 mg, 2 mmol) in N,N-dimethylforma-
mide (1 ml) and water (1 ml) was stirred under an oxygen atmosphere for
h. Alkene, 1b, (296 mg, 2 mmol) in N,N-dimethylformamide (0.5 ml) and
water (0.5 ml) was added and the reaction mixture was stirred at 50 °C for
4 h. The crude reaction mixture was applied directly to a pad of silica (ethyl
1
0
9
A. D. Ketley and J. Braatz, J. Chem. Soc., Chem. Commun., 1968, 169;
F. Conti, M. Donati, G. F. Pregaglia and R. Ugo, J. Organomet. Chem.,
971, 30, 421.
1
1
1
0 When compound 4b, labelled with deuterium at position 2, was
oxidised, 90% of the deuterium was found to be located at position 1 and
10% at 3. Although the presence of 10% deuterium at the position 3
suggests the involvement of a p-allylic complex as a minor pathway,
GC analysis of the reaction mixture showed the formation of small
amount (1%) of terminal alkene that if oxidised could give this result.
1 D. N. Lawson, J. A. Osborn and G. Wilkinson, J. Chem. Soc. A, 1966,
2
1
acetate–hexane; 1+4) and the concentrated filtrate was analysed by
H
NMR. Purification by flash silica gel chromatography afforded ketones 2b
and 3b (in the ratio 1+2.0) as a pale yellow oil (195 mg, 59%). b It is
interesting to note that the temperature of the reaction (compare to the result
at 100 °C, Table 1) can affect the regioselectivity of the reaction. At higher
temperatures the ‘anti-Markovnikov’ product is further favoured.
1
1
5
733; P. A. Evans and J. D. Nelson, J. Am. Chem. Soc., 1998, 120,
581.
Chem. Commun., 2001, 1844–1845
1845

 Gaunt
Gaunt
 Yu
Yu
 Spencer
Spencer