Y. Matsushita et al. / Tetrahedron Letters 45 (2004) 4723–4727
4727
8. Kobayashi, S.; Morikawa, M.; Hachiya, I. Tetrahedron
Lett. 1996, 37, 4183–4186.
9. Cui, D.-M.; Zhang, C.; Kawamura, M.; Shimada, S.
Tetrahedron Lett. 2004, 45, 1741–1745.
10. Kawamura, M.; Cui, D.-M.; Hayashi, T.; Shimada, S.
Tetrahedron Lett. 2003, 44, 7715–7717.
rated under the reduced pressure to give the almost pure
9 as colorless oil. Further purification was done by flash
column chromatography on silica gel (25 g) with hexane/
Et2O ¼ 20/1 to give 9 (1.03 g, 4.90 mmol) in 98% yield.
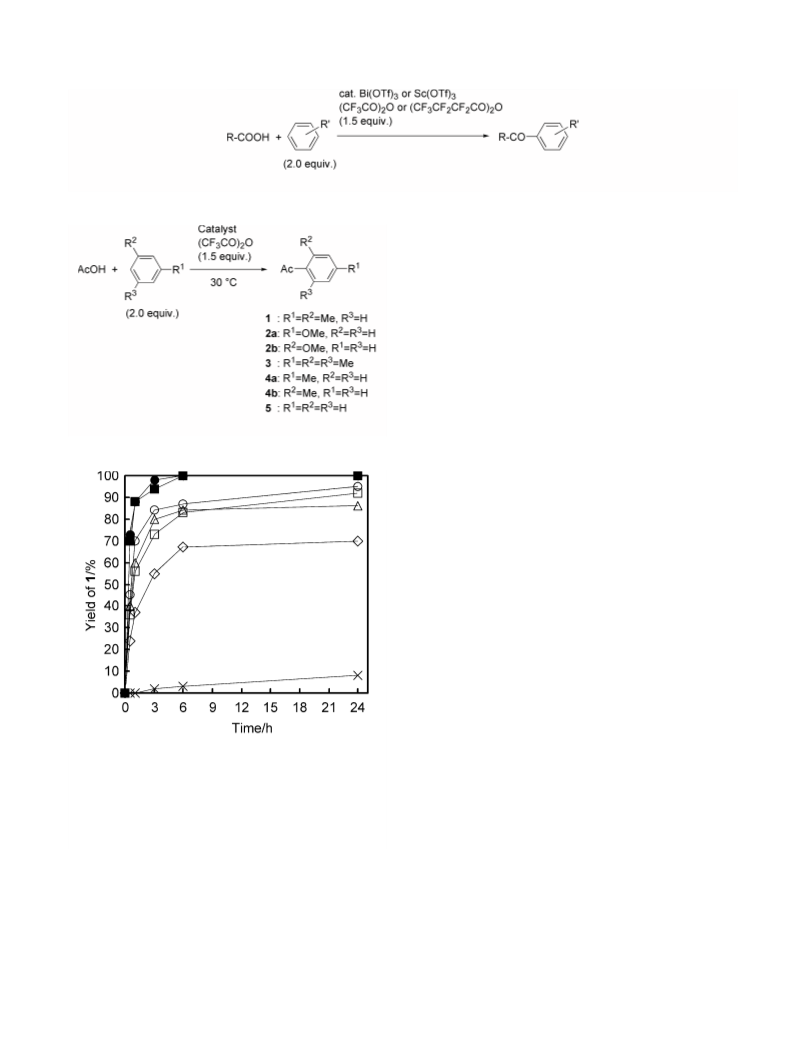
In conclusion, the Friedel–Crafts acylation of aromatic
compounds with carboxylic acids was successfully car-
ried out by using Bi(OTf)3 or Sc(OTf)3 as the catalyst in
the presence of perfluoroalkanoic anhydrides such as
TFAA and HFBA under solventless and mild condi-
tions, less reactive substrates such as benzene and
chlorobenzene could be converted into the correspond-
ing benzophenones. Further investigation to clarify the
scope and limitation of the present reaction and to apply
for synthesis of fine chemicals is now in progress.
11. Suzuki, K.; Kitagawa, H.; Mukaiyama, T. Bull. Chem.
Soc. Jpn. 1993, 66, 3729–3734.
12. Galli, C. Synthesis 1979, 303–304.
13. Smyth, T. P.; Corby, B. W. J. Org. Chem. 1998, 63, 8946–
8951.
14. Firouzabadi, H.; Iranpoor, N.; Nowrouzi, F. Tetrahedron
Lett. 2003, 44, 5343–5345.
15. For a review on Nafion-H catalysis in synthesis: Olah, G.
A.; Iyer, P. S.; Prakash, G. K. S. Synthesis 1986, 513–531.
16. For a review of super acids including TfOH: Effenberger,
F. Angew. Chem., Int. Ed. Engl. 1980, 19, 151–171.
17. For a review on bismuth chloride and bismuth triflate on
acylation reactions: Roux, C. L.; Dubac, J. Synlett 2002,
181–200.
References and notes
18. For a review on scandium triflate in organic syntheses:
Kobayashi, S. Eur. J. Org. Chem. 1999, 15–27.
1. House, H. O. Modern Synthetic Reactions, 2nd ed.; W.A.
Benjamin: California, 1972, pp 797–819.
ꢀ
19. (a) Desmurs, J. R.; Labrouillere, M.; Roux, C. L.;
Gaspard, H.; Laporterie, A.; Dubac, J. Tetrahedron Lett.
2. Olah, G. A. Friedel–Crafts Chemistry; John Wiley & Sons:
New York, 1973.
3. Premasagar, V.; Palaniswamy, V. A.; Eisenbraun, E. J. J.
Org. Chem. 1981, 46, 2974–2976.
ꢁ
1997, 38, 8871–8874; (b) Repichet, S.; Roux, C. L.; Dubac,
J.; Desmurs, J.-R. Eur. J. Org. Chem. 1998, 2743–2746.
20. Kobayashi, S.; Iwamoto, S. Tetrahedron Lett. 1998, 39,
4697–4700.
4. (a) Effenberger, F.; Epple, G. Angew. Chem., Int. Ed. Engl.
1972, 11, 299–300; (b) Effenberger, F.; Epple, G. Angew.
Chem., Int. Ed. Engl. 1972, 11, 300–301.
5. Yamato, T.; Hideshima, C.; Prakash, G. K. S.; Olah, G.
A. J. Org. Chem. 1991, 56, 3955–3957.
6. Chiche, B.; Finiels, A.; Gauthier, C.; Geneste, P.; Graille,
J.; Pioch, D. J. Org. Chem. 1986, 51, 2128–2130.
7. Kaur, J.; Kozhevnikov, I. V. Chem. Commun. 2002, 2508–
2509.
21. (a) Hachiya, I.; Moriwaki, M.; Kobayashi, S. Tetrahedron
Lett. 1995, 36, 409–412; (b) Hachiya, I.; Moriwaki, M.;
Kobayashi, S. Bull. Chem. Soc. Jpn. 1995, 68, 2053–2060.
22. Singh, R. P.; Kamble, R. M.; Chandra, K. L.; Saravanan,
C. P.; Singh, V. K. Tetrahedron 2001, 57, 241–247.
23. Olah, G. A.; Farooq, O.; Farnia, S. M. F.; Olah, J. A. J.
Am. Chem. Soc. 1988, 110, 2560–2565.
24. Matsuo, J.; Odashima, K.; Kobayashi, S. Synlett. 2000,
403–405.

 Matsushita, Yoh-Ichi
Matsushita, Yoh-Ichi
 Sugamoto, Kazuhiro
Sugamoto, Kazuhiro
 Matsui, Takanao
Matsui, Takanao