10.1002/anie.201707006
Angewandte Chemie International Edition
COMMUNICATION
Z. Yu, Y. Fu, ACS Catal. 2015, 5, 1414–1423; e) S. Nakaoka, Y.
Murakami, Y. Kataoka, Y. Ura, Chem. Commun. 2016, 335–338.
a) B. Morandi, Z. K. Wickens, R. H. Grubbs, Angew. Chem. Int. Ed.
2013, 52, 9751–9754; Angew. Chem. 2013, 125, 9933–9936; b) B.
Morandi, Z. K. Wickens, R. H. Grubbs, Angew. Chem. Int. Ed. 2013, 52,
2944–2948; Angew. Chem. 2013, 125, 3016–3020; c) T. Mitsudome, S.
Yoshida, T. Mizugaki, K. Jitsukawa, K. Kaneda, Angew. Chem. Int. Ed.
2013, 52, 5961–5964; Angew. Chem. 2013, 125, 6077–6080; d) T.
Mitsudome, K. Mizumoto, T. Mizugaki, K. Jitsukawa, K. Kaneda, Angew.
Chem. Int. Ed. 2010, 49, 1238–1240; Angew. Chem. 2010, 122, 1260–
1262; e) R. J. DeLuca, J. L. Edwards, L. D. Steffens, B. W. Michel, X.
Qiao, C. Zhu, S. P. Cook, M. S. Sigman, J. Org. Chem. 2013, 78,
1682−1686.
Lett. 2014, 16, 1610–1613; d) C. N. Cornell, M. S. Sigman, Org. Lett.
2006, 8, 4117–4120; e) G.-J. ten Brink, I. W. C. E. Arends, G.
Papadogianakis, R. A. Sheldon, Chem. Commun. 1998, 2359–2360.
[16] a) P. T. Anastas, J. C. Warner, Green Chemistry Theory and Practice;
Oxford University Press, New York, 1998; b) R. A. Sheldon, Chem. Soc.
Rev. 2012, 41, 1437–1451.
[5]
[17] G. T. Zhang, X. Hu, C.-W. Chiang, H. Yi, P. K. Pei, A. K. Singh, A. W.
Lei, J. Am. Chem. Soc. 2016, 138, 12037−12040.
[18] a) B. Plietker, Iron Catalysis in Organic Chemistry, Wiley-VCH,
Weinheim, 2008; b) C. Bolm, J. Legros, J. L. Paith, L. Zani, Chem. Rev.
2004, 104, 6217–6254; c) S. Enthaler, K. Junge, M. Beller, Angew.
Chem.Int. Ed. 2008, 47, 3317–3321; Angew. Chem. 2008, 120, 3363–
3367; d) I. Bauer, H. J. Knölker, Chem. Rev. 2015, 115, 3170–3387; e)
C. L. Sun, B. J. Li, Z. J. Shi, Chem. Rev, 2011, 111, 1293–1314.
[19] a) For few examples of Fe-catalyzed tandem epoxidation–isomerization
of terminal alkenes to aldehydes, see: a) Y.-D. Du, C.-W. Tse, Z.-J. Xu,
Y. G. Liu, C.-M. Che, Chem. Commun. 2014, 50, 12669–12672; b) A. D.
Chowdhury, R. Ray, G. K. Lahiri, Chem. Commun. 2012, 48, 5497–
5499; c) For an example of iron-catalyzed aerobic C=C cleavage, see:
A. Gonzalez-de-Castro, J. L. Xiao, J. Am. Chem. Soc. 2015, 137,
8206–8218.
[6]
a) B.W. Michel, L. D. Steffens, M. S. Sigman, J. Am. Chem. Soc. 2011,
133, 8317–8325; b) B. W. Michel, J. R. McCombs, A. Winkler, M. S.
Sigman, Angew. Chem. Int. Ed. 2010, 49, 7312−7315; Angew. Chem.
2010, 122, 7470–7473; c) B. W. Michel, A. M. Camelio, C. N. Cornell,
M. S. Sigman, J. Am. Chem. Soc. 2009, 131, 6076−6077; d) C. N.
Cornell, M. S. Sigman, J. Am. Chem. Soc. 2005, 127, 2796–2797; e) A.
Naik, L. Meina, M. Zabel, O. Reiser, Chem. Eur. J. 2010, 16, 1624–
1628; f) J. R. McCombs, B. W. Michel, M. S. Sigman, J. Org. Chem.
2011, 76, 3609–3613.
[20] a) P. R. Ortiz de Montellano, “Cytochrome P-450. Structure,
Mechanism, and Biochemistry,” Plenum Press, New York, 1986; b) D.
Mansuy, Pure Appl.Chem. 1987, 59, 759−770; c) B. Meunier, S. P. De
Visser, S. Shaik, Chem. Rev. 2004, 104, 3947–3980.
[7]
a) A. Stirling, N. N. Nair, A. Lledós, G. Ujaque, Chem. Soc. Rev. 2014,
43, 4940–4952; b) J. A. Keith, P. M. Henry, Angew. Chem. Int. Ed.
2009, 48, 9038–9049; Angew. Chem. 2009, 121, 9200–9212; c) P.
Kocovsky, J.-E. Bäckvall, Chem. Eur. J. 2015, 21, 36–56; d) J.-E.
Bäckvall, B. Akermark, S. O. Ljunggren, J. Am. Chem. Soc. 1979, 101,
2411–2416; e) G. Kovács, A. Stirling, A. Lledós, G. Ujaque, Chem. Eur.
J. 2012, 18, 5612–5619.
[21] I. Tabushi, M. Kodera, M. Yokoyama, J. Am. Chem. Soc. 1985, 107,
4466–4473.
[22] a) J. Gui, C.-M. Pan, Y. Jin, T. Qin, J. C. Lo, B. J. Lee, S. H. Spergel, M.
E. Mertzman, W. J. Pitts, T. E. La Cruz, M. A. Schmidt, N. Darvatkar,
S.R. Natarajan, P. S. Baran, Science 2015, 348, 886–891; b) J. C. Lo, J.
Gui, Y. Yabe, C.-M. Pan, P. S. Baran, Nature 2014, 516, 343–348; c) J.
C. Lo, D. Y. Kim, C.-M. Pan, J. T. Edwards, Y. Yabe, J. H. Gui, T. Qin,
S. Gutiérrez, J. Giacoboni, M. W. Smith, P. L. Holland, P. S. Baran, J.
Am. Chem. Soc. 2017, 139, 2484–2503; d) J. C. Lo, Y. Yabe, P. S.
Baran, J. Am. Chem. Soc. 2014, 136, 1304–1307; e) H. T. Dao, C. Li,
Q. Michaudel, B. D. Maxwell, P. S. Baran, J. Am. Chem. Soc. 2015,
137, 8046–8049.
[8]
[9]
a) S. Donck, E. Gravel, N. Shah, D. V. Jawale, E. Doris, I. N. N.
Namboothiri, ChemCatChem 2015, 7, 2318–2322; b) V. V. Namboodiri,
R. S. Varma, E. Sahle-Demessie, U. R. Pillai, Green Chem. 2002, 4,
170–173; c) M. Sommovigo, H. Alper, J. Mol. Catal. 1994, 88, 151–158.
D. A. Chaudhari, R. A. Fernandes, J. Org. Chem. 2016, 81, 2113–2121.
[10] a) P. Teo, Z. K. Wickens, G. Dong, R. H. Grubbs, Org. Lett. 2012, 14,
3237–3239; b) G. Zhang, X. Xie, Y. Wang, X. Wen, Y. Zhao, C. Ding,
Org. Biomol. Chem. 2013, 11, 2947–2950.
[11] a) Q. Cao, D. S. Bailie, R. Fu, M. J. Muldoon, Green Chem. 2015, 17,
2750–2757; b) X. Wang, N. S. Venkataramanan, H. Kawanami, Y.
Ikushima, Green Chem. 2007, 9, 1352–1355; c) V. V. Namboodiri, R. S.
Varma, E. Shale-Demessie, U. R. Pillai, Green Chem. 2002, 4, 170–
173.
[23] a) S. W. M. Crossley, C. Obradors, R. M. Martinez, R. A. Shenvi, Chem.
Rev. 2016, 116, 8912−9000; b) T. Hashimoto, D. Hirose, T. Taniguchi,
Angew. Chem. Int. Ed. 2014, 53, 2730–2734; Angew. Chem. 2014, 126,
2768–2772; c) E. K. Leggans, T. J. Barker, K. K. Duncan, D. L. Boger,
Org. Lett. 2012, 14, 1428–1431; d) T. Taniguchi, N. Goto, A. Nishibata,
H. Ishibashi, Org. Lett. 2010, 12, 112–115; e) T. Sugimori, S.-I. Horike,
S. Tsumura, M. Handa, K. Kasuga, Inorg. Chim. Acta 1998, 283, 275–
278; f) M. Takeuchi, M. Kodera, K. Kano, Z.Yoshida, J. Mol. Catal. A-
Chem. 1996, 113, 51–57.
[12] a) J. Ettedgui, R. Neumann, J. Am. Chem. Soc. 2009, 131, 4–5; b) S.
Uchiumi, K. Ataka, T. Matsuzaki, J. Organomet. Chem. 1999, 576,
279–289.
[13] a) R. A. Fernandes, D. A. Chaudhari, J. Org. Chem. 2014, 79, 5787–
5793; b) R. A. Fernandes, V. Bethi, Tetrahedron 2014, 70, 4760–4767;
c) R. S. Phatake, C. V. Ramana, Tetrahedron Lett. 2015, 56, 3868–
3871.
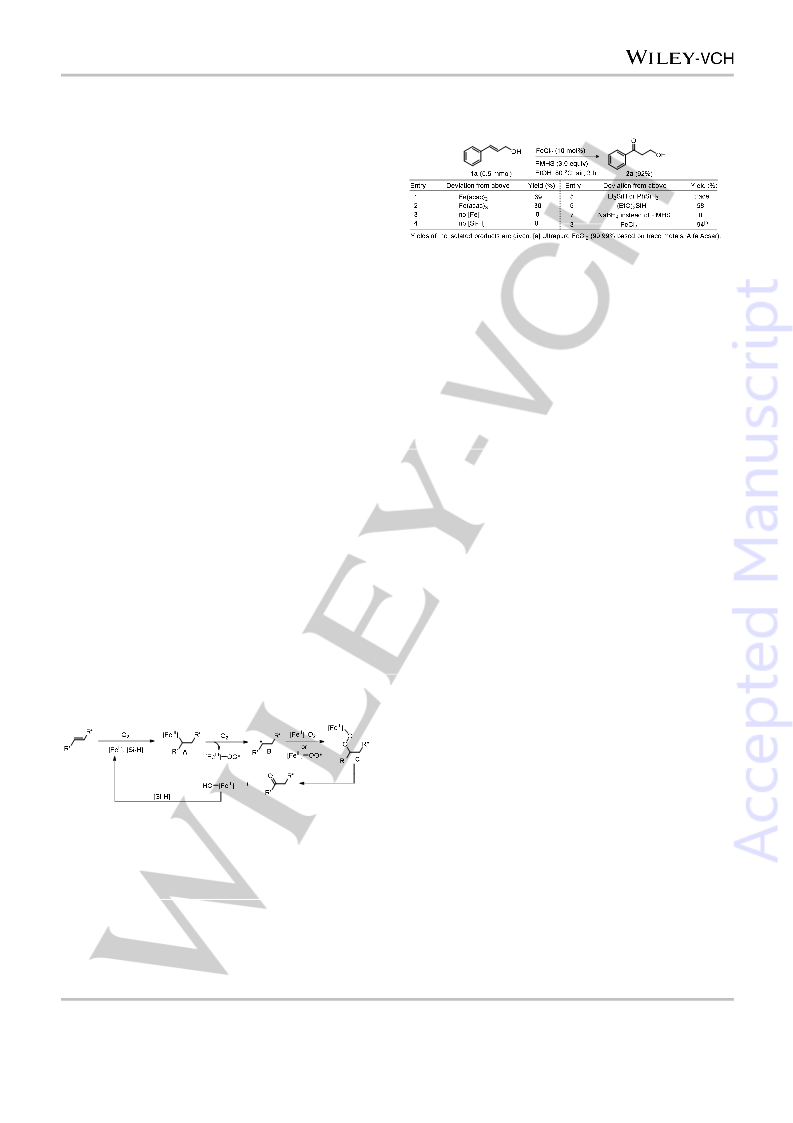
[24] See the Supporting Information for details.
[25] R. D. Arasasingham, A. L. Balch, C. R. Cornman, L. Latos-Grazynski, J.
Am. Chem. Soc. 1989, 111, 4357–4363.
[14] a) S. S. Stahl, Science 2005, 309, 1824–1826; b) J. Piera, J.-E.
Bäckvall, Angew. Chem. Int. Ed. 2008, 47, 3506–3523; Angew. Chem.
2008, 120, 3558–3576; c) Z. Shi, C. Zhang, C. Tang, N. Jiao, Chem.
Soc. Rev. 2012, 41, 3381–3430; d) T. Punniyamurthy, S. Velusamy, J.
Iqbal, Chem. Rev. 2005, 105, 2329–2364.
[26] H. Song, B. Kang, S. H. Hong, ACS Catal. 2014, 4, 2889–2895.
[27] J. Meinwald, S. S. Labana, M. S. Chadha, J. Am. Chem. Soc.1963, 85,
582−585.
[28] a) J.-J. Chen, P.-H. Chen, C.-H. Liao, S.-Y. Huang, I.-S. Chen, J. Nat.
Prod. 2007, 70, 1444−1448.
[15] a) B. J. Anderson, J. A. Keith, M. S. Sigman, J. Am. Chem. Soc. 2010,
132, 11872–11874; b) T. Mitsudome, T. Umetani, N. Nosaka, K. Mori, T.
Mizugaki, K. Ebitani, K. Kaneda, Angew. Chem. Int. Ed. 2006, 45, 481–
485; Angew. Chem. 2006, 118, 495–499; c) Y.-F. Wang, Y.-R. Gao, S.
Mao, Y.-L. Zhang, D.-D. Guo, Z.-L. Yan, S.-H. Guo, Y.-Q. Wang, Org.
[29] For a report on the catalytic Wacker-type oxidation of electron-deficient
aliphatic olefins, see: ref. [5c].
[30] E. Kim, D. M. Gordon, W. Schmid, G. M. Whitesides, J. Org. Chem.
1993, 58, 5500–5507.
This article is protected by copyright. All rights reserved.

 Liu, Binbin
Liu, Binbin
 Jin, Fengli
Jin, Fengli
 Wang, Tianjiao
Wang, Tianjiao
 Yuan, Xiaorong
Yuan, Xiaorong
 Han, Wei
Han, Wei