
Transition Metal Chemistry p. 707 - 719 (2011)
Update date:2022-08-11
Topics:
 Manjari, Padma Sunitha
Manjari, Padma Sunitha
 Reddy, Cherkupally Sanjeeva
Reddy, Cherkupally Sanjeeva
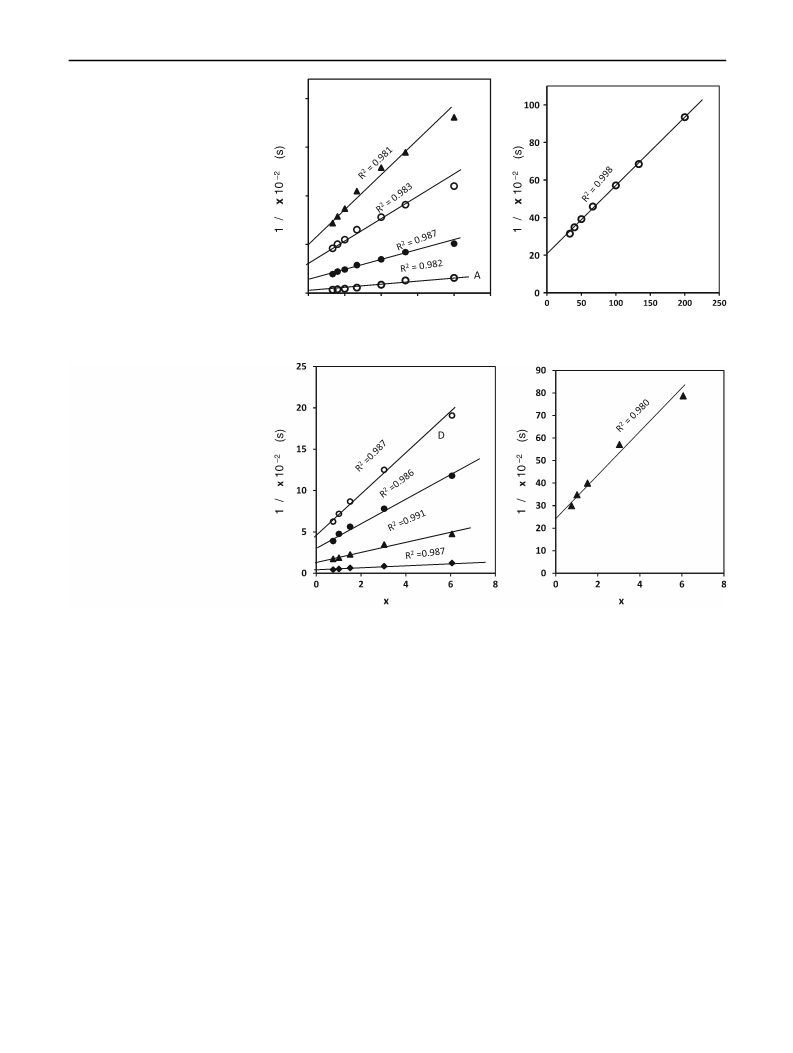
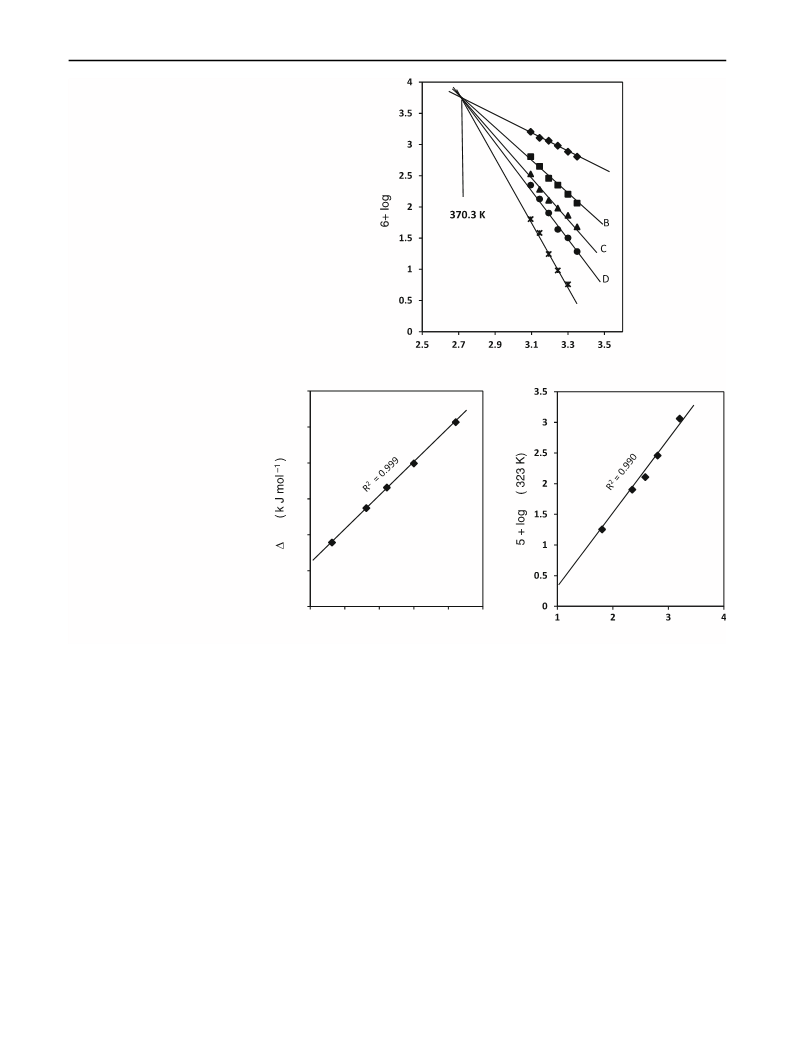
Ru(III) acts an efficient catalyst in the oxidation of substituted 4-oxo-4-arylbutanoic acids (4-oxo acids) by bromate in sulfuric acid medium, giving the corresponding benzoic acids in quantitative yields. The reaction shows first-order dependence in both [bromate] and [H2SO 4], and a non-linear dependence on both [oxo acid] and [catalyst]. Changing solvent from H2O to D2O increases the rate. The rate is not affected by ionic strength but decreases with increase in dielectric constant of the medium. Electron-releasing substituents in the phenyl ring of the substrate greatly accelerate the rate, whereas the retardation by electron-withdrawing substituents, though perceptible, is small. The linear free-energy relationship is characterized by smooth curves in Hammett plots of log k versus σ; however, linear plots are obtained with excellent correlation coefficients at all the studied temperatures, when Brown's σ+ values are used. The reaction constant is negative and decreases with increase in temperature. From the intersection of the lines in the Hammett and Arrhenius plots, the isokinetic relationship is evaluated. A mechanism involving a cyclic oxidant-substrate-catalyst ternary complex is proposed, in which both C-C bond-breaking and C-O bond formation are involved, and the oxidation state of Ru(III) remains unchanged. A rate law explaining all the kinetic results has been derived and verified. The reaction is an example of neighboring group participation in intramolecular catalysis and is potentially useful for the synthesis of substituted benzoic acids.
View More










Contact:+86-0512-88957371
Address:shanghai
Contact:+86-25-83719363
Address:106-7 Chunnan Rd, Chunxi Town, Gaochun, Nanjing, China
Contact:+86-10-83993285
Address:Rm.1708, Haobai Tower, Building 6, No.50, North Road, West Third Ring, Haidian District, Beijing, China
website:http://www.acrospharmatech.com
Contact:+1-3234804688
Address:Flat/RM 1502,Easey Commercial building 253-261 Hennessy Road,Wanchai,HongKong
lianyungang jinkang pharmaceutical technology co., ltd.
Contact:008651885445517
Address:Jinshan industrial park, Ganyu county, Lianyungang, Jiangsu Province, 222115, China
Doi:10.1021/jacs.8b01017
(2018)Doi:10.1016/j.dyepig.2018.04.016
(2018)Doi:10.1246/bcsj.55.3051
(1982)Doi:10.1021/ja409012q
(2013)Doi:10.1002/cphc.201901134
(2020)Doi:10.1039/c6dt04534k
(2017)