10.1002/cctc.201700517
ChemCatChem
COMMUNICATION
Lindhardt, T. Skrydstrup, Acc. Chem. Res. 2016, 49, 594-605; k) Y. Bai,
D. C. Davis, M. Dai, J. Org. Chem. 2017, 82, 2319−2328.
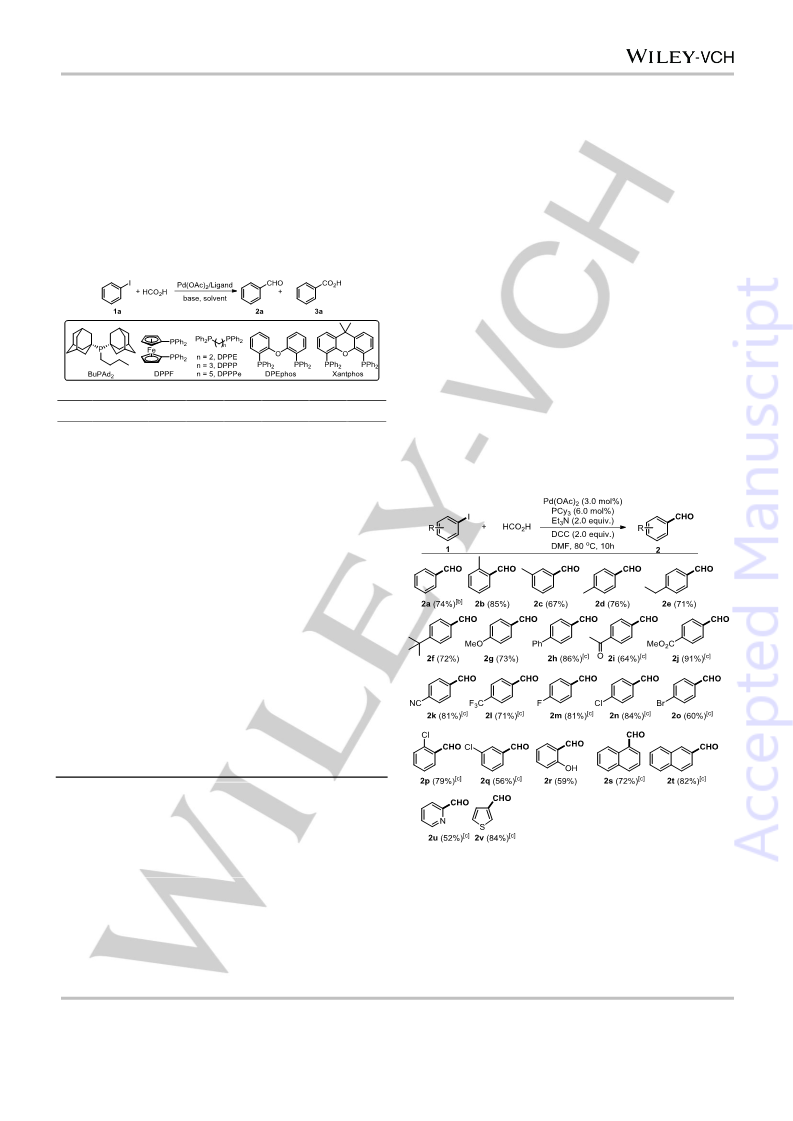
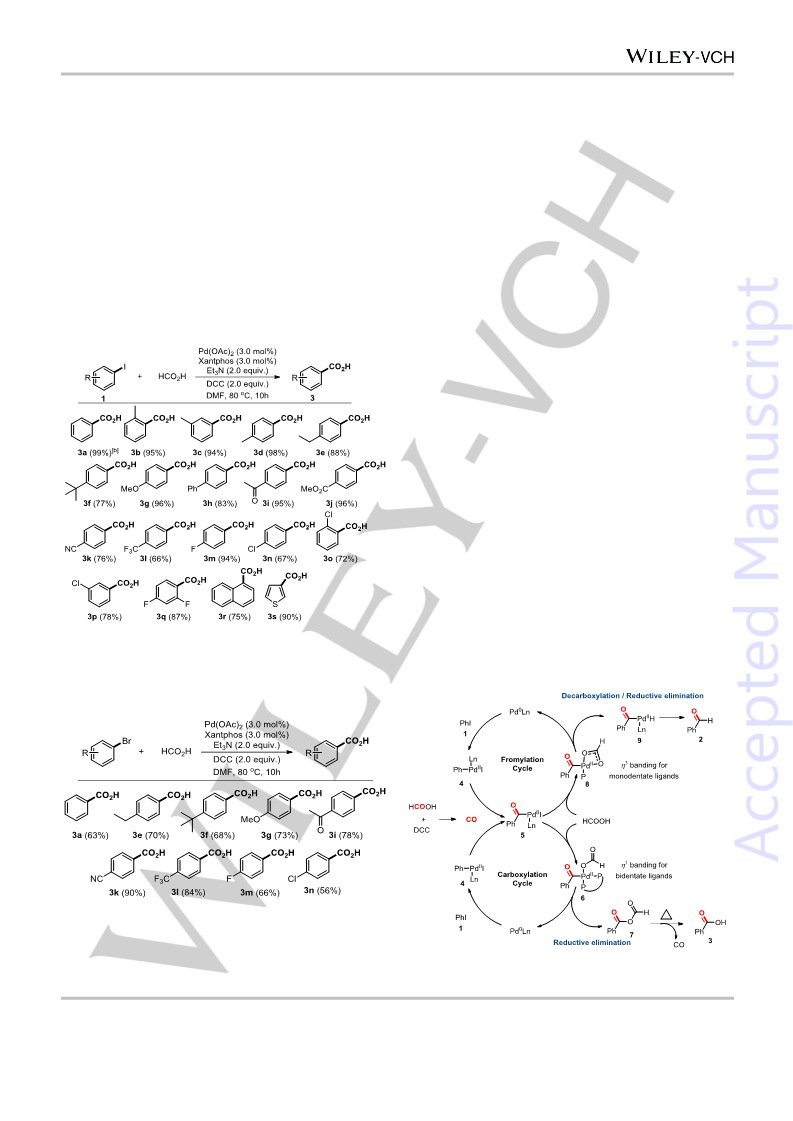
In conclusion, a strategy for ligand controlled divergent synthesis
of aldehyde and acid from aryl halides had been established.
With formic acid as the carbonyl source, a wide range of
substituted aryl halides were transformed into the corresponding
aldehydes or acid derivatives selectively in moderate to
excellent yields. Generally, small sterically hindered
monodentate alkylphosphine facilitate the formation of
aldehydes, while wide bite angle bidentate ligand promote the
formation of acid. This strategy provides a general method for
the divergent synthesis of aldehydes and carboxylic acids from
easily available aryl halides under identical reaction conditions.
[3]
a) P. W. N. M. van Leeuwen, P. C. J. Kamer, J. N. H. Reek, P. Dierkes,
Chem. Rev. 2000, 100, 2741-2769; b) P. Kamer, P. W. N. M. van
Leeuwen, J. Reek, Acc. Chem. Res. 2001, 34, 895-904; c) Z. Freixa, P.
W. N. M. van Leeuwen, Dalton Trans. 2003, 1890-1901; d) M.-N.
Birkholz (née Gensow), Z. Freixab, P. W. N. M. van Leeuwen, Chem.
Soc. Rev. 2009, 38, 1099-1118; e) F. Agbossou, J.-F. Carpentier, A.
Mortreux, Chem. Rev. 1995, 95, 2485–2506; f) P. Eilbracht, L.
Bꢀrfacker, C. Buss, C. Hollmann, B. E. Kitsos-Rzychon, C. L.
Kranemann, T. Rische, R. Roggenbuck, A. Schmidt, Chem. Rev. 1999,
99, 3329–3366; g) J. Klosin, C. R. Landis, Acc. Chem. Res. 2007, 40,
1251–1259; h) F. Hebrard, P. Kalck, Chem. Rev. 2009, 109, 4272–
4282; i) R. Franke, D. Selent, A. Bꢁrner, Chem. Rev. 2012, 112, 5675–
5732.
Experimental Section
[4]
[5]
M. Feng, B. Tang, N. Wang, H.-X. Xu, X. Jiang, Angew. Chem. Int. Ed.
2015, 54, 14960-14964.
a) T. Morimoto, K. Kakiuchi, Angew. Chem. Int. Ed. 2004, 43, 5580-
5588; b) L. Wu, Q. Liu, R. Jackstell, M. Beller, Angew. Chem. Int. Ed.
2014, 53, 6310-6320; c) P. Gautam, B. M. Bhanage, Catal. Sci.
Technol. 2015, 5, 4663-4702.
Pd(OAc)2 (3 mol %), ligand (6 mol% for monodentate ligands and 3 mol%
for bidentate ligand) were transferred into an oven-dried tube which was
filled with nitrogen. DMF (2.0 mL), formic acid (7.0 mmol), and aryl halide
(1.0 mmol) were added to the reaction tube. After DCC (2.0 mmol) and
Et3N (2.0 mmol) were added, the tube was sealed and the mixture was
stirred at 80 °C for 10 h. After the reaction was completed, the reaction
mixture was filtered and concentrated under vacuum. The crude product
was purified by column chromatography on silica gel to afford the
corresponding product.
[6]
a) S. Cacchi, G. Fabrizi, A. Goggiamani, Org. Lett. 2003, 5, 4269–4272;
b) S. Cacchi, G. Fabrizi, A. Goggiamani, J. Comb. Chem. 2004, 6, 692–
694; c) S. Korsager, R. H. Taaning, T. Skrydstrup, J. Am. Chem. Soc.
2013, 135, 2891–2894; d) X. Qi, L.-B. Jiang, C.-L. Li, R. Li, X.-F. Wu,
Chem. Asian J. 2015, 10, 1870–1873; e) X. Qi, L.-B. Jiang, H.-P. Li, X.-
F. Wu, Chem. Eur. J. 2015, 21, 17650–17656; f) X. Qi, H.-P. Li, X.-F.
Wu, Chem. Asian J. 2016, 11, 2453–2457; g) X. Qi, C.-L. Li, L.-B. Jiang,
W.-Q. Zhang, X.-F. Wu, Catal. Sci. Technol. 2016, 6, 3099–3107; h) L.-
B. Jiang, R. Li, H.-P. Li, X. Qi, X.-F. Wu, ChemCatChem 2016, 8,
1788–1791; i) X. Qi, C.-L. Li, X.-F. Wu, Chem. Eur. J. 2016, 22, 5835–
5838 j) X. Qi, R. Li, X.-F. Wu, RSC Adv. 2016, 6, 62810–62813; k) L.-B.
Jiang, X. Qi, X.-F. Wu, Tetrahedron Lett. 2016, 57, 3368-3370; l) H.-P.
Li, H.-J. Ai, X. Qi, J.-B. Peng, X.-F. Wu, Org. Biomol. Chem. 2017, 15,
1343-1345.
Acknowledgements
The authors thank the financial supports from NSFC (21472174,
21602201, 21602204) and Zhejiang Natural Science Fund for
Distinguished Young Scholars (LR16B020002).
Keywords: palladium catalyst • coupling reaction • formic acid •
reductive carbonylation • carboxylation
[7]
[8]
[1]
[2]
J. F. Hartwig, Organotransition Metal Chemistry: From Bonding to
Catalysis: From Bonding to Catalysis, 2009, University Science
Books,U.S.
When the reaction was conducted with 2.0 equivalent of formic acid
and 2.0 equivalent of DCC in 1,4-Dioxane at 60 oC, since formic acid
was completely dehydrated to release CO, the acyl-palladium
intermediate was captured by benzoic ion, and moderate yield of
benzoic anhydride was obtained.
a) M. Queirolo, A. Vezzani, R. Mancuso, B. Gabriele, M. Costa, N. D.
Ca’, J. Mol. Catal. A: Chem. 2015, 398, 115-126; b) G. P. Chiusoli, M.
Costa, L. Cucchia, B. Gabriele, G. Salerno, L. Veltri, J. Mol. Catal. A:
Chem. 2003, 204-205, 133-142.
For selected reviews, see: a) L. Kollár in Modern Carbonylation
Methods, Wiley-VCH, Weinheim (Germany), 2008; b) C. F. J. Barnard,
Organometallics 2008, 27, 5402–5422; c) A. Brennführer, H. Neumann,
M. Beller, Angew. Chem. Int. Ed. 2009, 48, 4114–4133; d) Q. Liu, H.
Zhang, A. Lei, Angew. Chem. Int. Ed. 2011, 50, 10788–10799; e) B.
Gabriele, R. Mancuso, G. Salerno, Eur. J. Org. Chem. 2012, 6825–
6839; f) S. Sumino, A. Fusano, T. Fukuyama, I. Ryu, Acc. Chem. Res.
2014, 47, 1563-1574; g) J.-B. Peng, X. Qi, X.-F. Wu, Synlett 2017, 28,
175-194; h) X.-F. Wu, RSC Adv. 2016, 6, 83831-83837; i) J.-B. Peng, X.
Qi, X.-F. Wu, ChemSusChem 2016, 9, 2279-2283; j) S. D. Friis, A. T.
This article is protected by copyright. All rights reserved.

 Wu, Fu-Peng
Wu, Fu-Peng
 Peng, Jin-Bao
Peng, Jin-Bao
 Meng, Ling-Shen
Meng, Ling-Shen
 Qi, Xinxin
Qi, Xinxin
 Wu, Xiao-Feng
Wu, Xiao-Feng