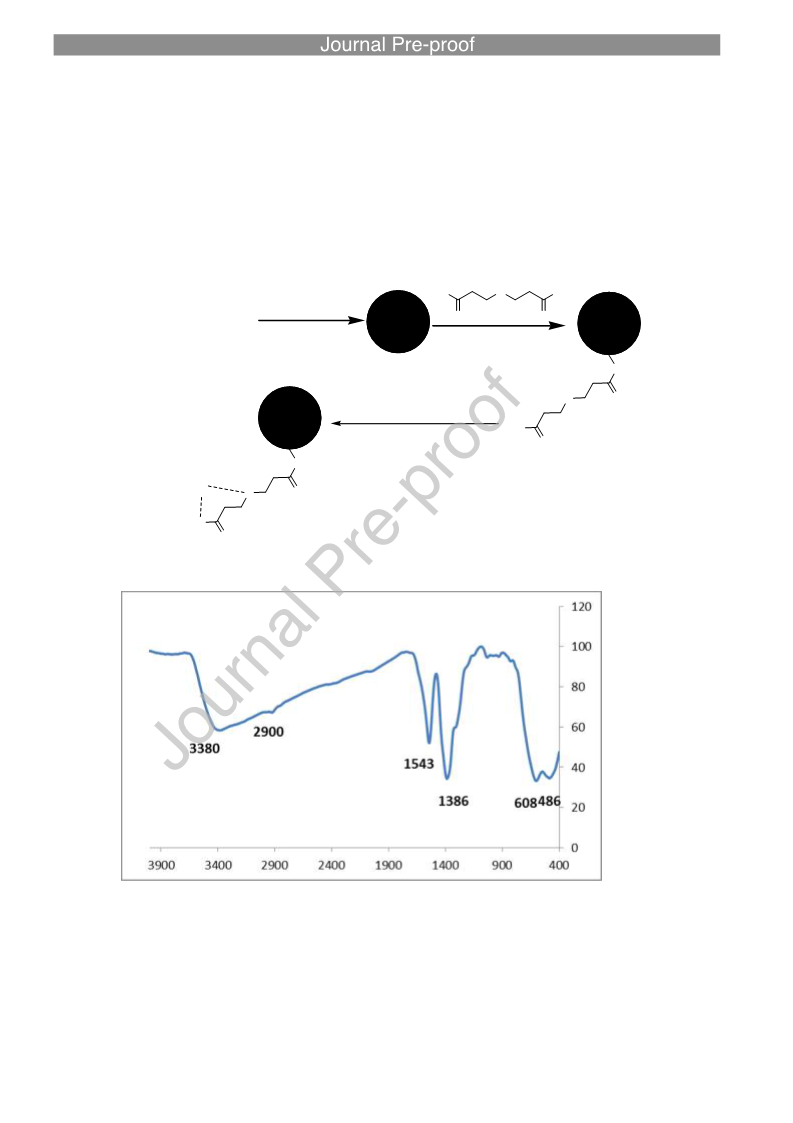
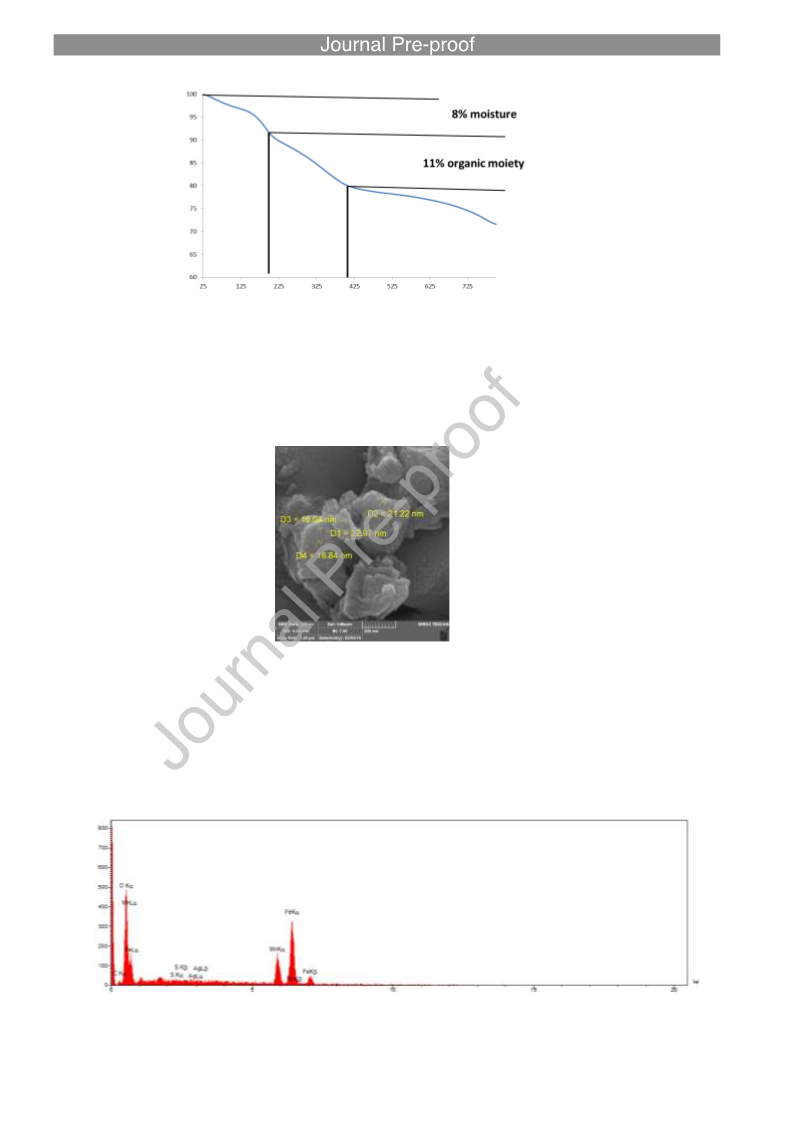
Direct C-C Cross‐Coupling of Secondary and Primary Alcohols Catalyzed by a γ‐
Alumina‐Supported Silver Subnanocluster, Angew. Chem. Int. Ed. 48(22) (2009) 3982-
3
986;f) K.i. Shimizu, K. Sugino, K. Sawabe, A. Satsuma, Oxidant‐Free
Dehydrogenation of Alcohols Heterogeneously Catalyzed by Cooperation of Silver
Clusters and Acid–Base Sites on Alumina, Chem.: Eur. J. 15(10) (2009) 2341-2351.
[
8] a) G.M. Whitesides, M. Hackett, R.L. Brainard, J.P.P. Lavalleye, A.F. Sowinski,
A.N. Izumi, S.S. Moore, D.W. Brown, E.M. Staudt, Suppression of unwanted
heterogeneous platinum (0)-catalyzed reactions by poisoning with mercury (0) in
systems involving competing homogeneous reactions of soluble organoplatinum
compounds: thermal decomposition of bis (triethylphosphine)-3, 3, 4, 4-
tetramethylplatinacyclopentane, Organometallics 4(10) (1985) 1819-1830
[
9] a) J.M. Pérez, R. Cano, M. Yus, D.J. Ramón, Straightforward synthesis of aromatic
imines from alcohols and amines or nitroarenes using an impregnated copper catalyst,
Eur. J. Org. Chem. 2012(24) (2012) 4548-4554; b) S. Pande, A. Saha, S. Jana, S Sarkar,
M. Basu, M. Pradhan, A.K. Sinha, S. Saha, A. Pal and T. Pal, Resin-immobilized CuO
and Cu nanocomposites for alcohol oxidation, Org. lett. 10(22) (2008) 5179-5181.
[
10] a) E.W. Dahl, T. Louis-Goff, N.K. Szymczak, Second sphere ligand modifications
enable a recyclable catalyst for oxidant-free alcohol oxidation to carboxylates, Chem
Commun. 53(14) (2017) 2287-2289. b) Z. Dai, Q. Luo, X. Meng, R. Li, J. Zhang, T.
Peng, Ru (II) complexes bearing 2, 6-bis (benzimidazole-2-yl) pyridine ligands: A new
class of catalysts for efficient dehydrogenation of primary alcohols to carboxylic acids
and H2 in the alcohol/CsOH system, Journal of Organometallic Chemistry 830 (2017)
1
1-18.
[
11] a) J.-H. Choi, L.E. Heim, M. Ahrens, M.H. Prechtl, Selective conversion of
alcohols in water to carboxylic acids by in situ generated ruthenium trans dihydrido
carbonyl PNP complexes, Dalton Trans.43(46) (2014) 17248-17254; b) C. Santilli, I.S.
Makarov, P. Fristrup, R. Madsen, Dehydrogenative synthesis of carboxylic acids from
primary alcohols and hydroxide catalyzed by a ruthenium N-heterocyclic carbene
complex, J. Organomet. Chem. 81(20) (2016) 9931-9938; c) D. Ventura-Espinosa, C.
Vicent, M. Baya, J.A. Mata, Ruthenium molecular complexes immobilized on graphene
as active catalysts for the synthesis of carboxylic acids from alcohol dehydrogenation,
Catal. Sci. Technol. 6(22) (2016) 8024-8035; d) L. Zhang, D.H. Nguyen, G. Raffa, X.
Trivelli, F. Capet, S. Desset, S. Paul, F. Dumeignil, R.M. Gauvin, Catalytic conversion
of alcohols into carboxylic acid salts in water: scope, recycling, and mechanistic
insights, ChemSusChem. 9(12) (2016) 1413-1423; e) J.-H. Choi, L.E. Heim, M. Ahrens,
M.H. Prechtl, Selective conversion of alcohols in water to carboxylic acids by in situ
generated ruthenium trans dihydrido carbonyl PNP complexes, Dalton Trans.43(46)
(
2014) 17248-17254; f) Z. Dai, Q. Luo, X. Meng, R. Li, J. Zhang, T. Peng, Ru (II)
complexes bearing 2, 6-bis (benzimidazole-2-yl) pyridine ligands: A new class of
catalysts for efficient dehydrogenation of primary alcohols to carboxylic acids and H
2
in
the alcohol/CsOH system, Journal of Organometallic Chemistry 830 (2017) 11-18;g) C.
Santilli, I.S. Makarov, P. Fristrup, R. Madsen, Dehydrogenative synthesis of carboxylic
acids from primary alcohols and hydroxide catalyzed by a ruthenium N-heterocyclic
carbene complex, J. Organomet. Chem. 81(20) (2016) 9931-9938; h) D. Ventura-
Espinosa, C. Vicent, M. Baya, J.A. Mata, Ruthenium molecular complexes immobilized
on graphene as active catalysts for the synthesis of carboxylic acids from alcohol
dehydrogenation, Catal. Sci. Technol. 6(22) (2016) 8024-8035;i) A. Sarbajna, I. Dutta,
P. Daw, S. Dinda, S.W. Rahaman, A. Sarkar, J.K. Bera, Catalytic conversion of
alcohols to carboxylic acid salts and hydrogen with alkaline water, ACS Catal. 7(4)
(
2017) 2786-2790;

 Yazdani, Elahe
Yazdani, Elahe
 Heydari, Akbar
Heydari, Akbar