Organic Letters
Letter
deuterated alcohol 61a, in 92% yield. Although the oxidation
of aldehydes obviously included a hydroxylation process with
sodium hydroxide, we still performed experiments with 18O-
NaOH. As expected, the 18O-containing acid was isolated as
Information). The proposed oxidation reaction pathway is
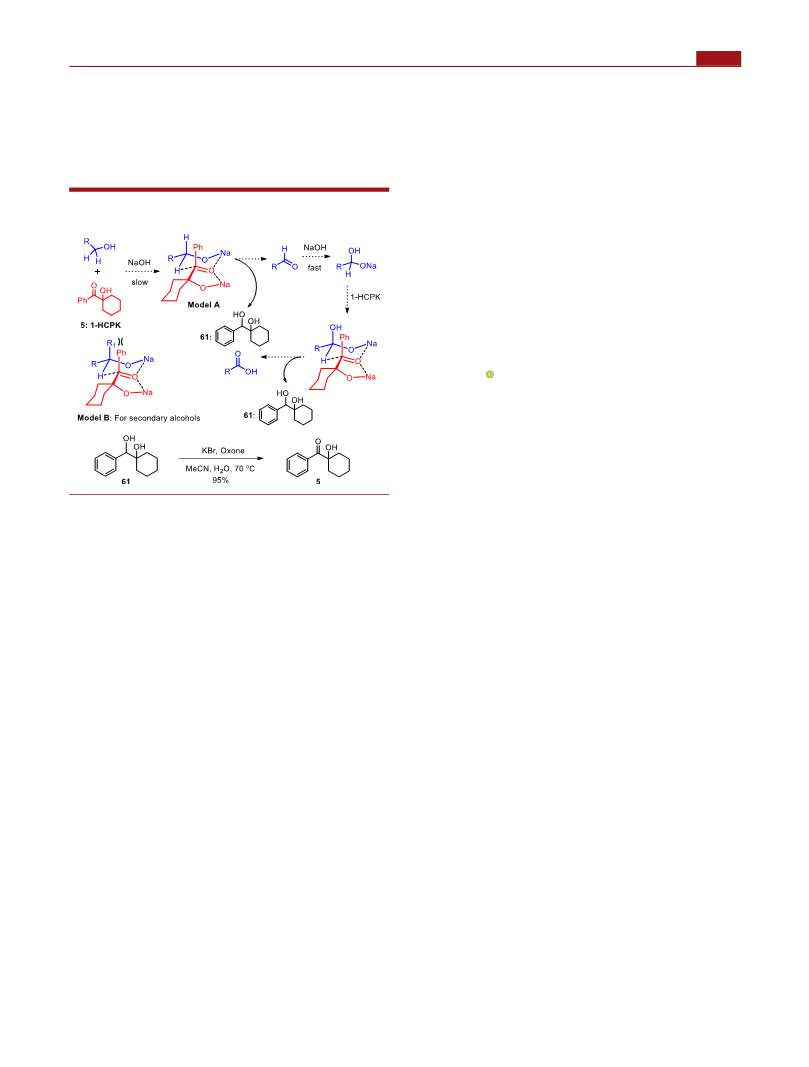
shown in Scheme 5. Although the reaction follows the
Experimental procedures, characterization data, and
spectra of all new compounds (PDF)
Accession Codes
CCDC 2092153 contains the supplementary crystallographic
data for this paper. These data can be obtained free of charge
bridge Crystallographic Data Centre, 12 Union Road,
Cambridge CB2 1EZ, UK; fax: +44 1223 336033.
Scheme 5. Proposed Pathyway for 1-HCPK-Mediated
Oxidations and Recycling of 1-HCPK
AUTHOR INFORMATION
■
Corresponding Author
Hongbin Zhang − Key Laboratory of Medicinal Chemistry for
Natural Resource, Ministry of Education, Yunnan Provincial
Center for Research and Development of Natural Products,
School of Chemical Science and Technology, Yunnan
University, Kunming, Yunnan 650091, People’s Republic of
Authors
Wen-Yun Tan − Key Laboratory of Medicinal Chemistry for
Natural Resource, Ministry of Education, Yunnan Provincial
Center for Research and Development of Natural Products,
School of Chemical Science and Technology, Yunnan
University, Kunming, Yunnan 650091, People’s Republic of
China
Yi Lu − Key Laboratory of Medicinal Chemistry for Natural
Resource, Ministry of Education, Yunnan Provincial Center
for Research and Development of Natural Products, School of
Chemical Science and Technology, Yunnan University,
Kunming, Yunnan 650091, People’s Republic of China
Jing-Feng Zhao − Key Laboratory of Medicinal Chemistry for
Natural Resource, Ministry of Education, Yunnan Provincial
Center for Research and Development of Natural Products,
School of Chemical Science and Technology, Yunnan
University, Kunming, Yunnan 650091, People’s Republic of
China
Wen Chen − Key Laboratory of Medicinal Chemistry for
Natural Resource, Ministry of Education, Yunnan Provincial
Center for Research and Development of Natural Products,
School of Chemical Science and Technology, Yunnan
University, Kunming, Yunnan 650091, People’s Republic of
China
Oppenauer oxidation pathway,21 the hydroxyl group present
in the oxidant might form sodium alkoxide and further activate
the adjacent carbonyl via coordination (for primary alcohols:
model A, Scheme 5). The selective oxidation of primary
alcohols (model A) rather than secondary alcohols might result
from the steric interactions between the R1-substituted group
(present in secondary alcohols) and the benzene group
(present in the oxidant), as indicated in Scheme 5 (model B).
Finally, recycling of 1-hydroxycyclohexyl phenyl ketone was
conducted (Scheme 5). Although 1-HCPK is quite cheap, it is
still desirable to recycle this organic compound. Thus, diol 61
produced from the reaction process (around 90−97% yields)
was collected and subjected to oxidation under the conditions
of KBr and oxone,24 and 1-hydroxycyclohexyl phenyl ketone
was obtained in 95% yield.
In conclusion, we have developed a new process for the
chemoselective oxidation of primary alcohols and aldehydes to
the corresponding carboxylic acids. The mechanism was also
elaborated by isotope experiments. Features of this oxidation
are a new hydrogen transfer acceptor, namely the commercially
available and cheap material 1-hydroxycyclohexyl phenyl
ketone, tolerance of a wide range of functional groups,
including vulnerable tert-butanesulfinimades and secondary
alcohols, an easy to handle procedure, and good to excellent
yields. The oxidation process is practical and can be carried out
on a gram scale. On the basis of our experience, this selective
oxidation should find application in organic synthesis.
Complete contact information is available at:
Notes
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
■
This work was supported by grants from the Natural Science
Foundation of China (U1702286, 21572197, 21762047, and
21901224), the Program for Changjiang Scholars and
Innovative Research Team in University (IRT17R94), the
Ling-Jun Scholars of Yunnan Province (202005AB160003),
and the Talent Plan of Yunnan Province (YNWR-QNBJ-2018-
025). Dr. Xiaonian Li at the Kunming Institute of Botany is
gratefully acknowledged for X-ray crystallographic analysis.
ASSOCIATED CONTENT
* Supporting Information
■
sı
The Supporting Information is available free of charge at
D
Org. Lett. XXXX, XXX, XXX−XXX

 Tan, Wen-Yun
Tan, Wen-Yun
 Lu, Yi
Lu, Yi
 Zhao, Jing-Feng
Zhao, Jing-Feng
 Chen, Wen
Chen, Wen
 Zhang, Hongbin
Zhang, Hongbin