Inorganic Chemistry
Forum Article
Svelle, S.; Lillerud, K. P.; Kolboe, S. Catal. Today 2005, 106, 108−111.
(e) Haw, J. F.; Song, W. G.; Marcus, D. M.; Nicholas, J. B. Acc. Chem.
Res. 2003, 36, 317−326.
selective amine N-monoalkylation. MW heating proved highly
beneficial in the TH and N-alkylation reactions. The value of
our catalysts thus extends well beyond the dehydrogenation of
glycerol and other polyols.
(3) Mythili, R.; Venkatachalam, P.; Subramanian, P.; Uma, D. Int. J.
Energy Res. 2014, 38, 1233−1259.
(4) Thomas, C. M.; Suss-Fink, G. Coord. Chem. Rev. 2003, 243, 125−
142.
EXPERIMENTAL SECTION
■
General Procedures. Reactions were carried out under a N2
atmosphere using degassed solvents unless otherwise noted. NMR
spectra were recorded using Agilent DD2-400 and -500 or Bruker
AMX-500 spectrometers. Reactions under MW irradiation were
carried out in a Biotage initiator microwave synthesizer with a
Robot Eight automated sampler.
(5) (a) Olah, G. A.; Goeppert, A.; Prakash, S. G. K. Beyond Oil and
Gas: The Methanol Economy; Wiley-VCH: Weinheim, Germany, 2006.
(b) Olah, G. A. Angew. Chem., Int. Ed. 2013, 52, 104−107. (c) Olah, G.
A. Angew. Chem., Int. Ed. 2005, 44, 2636−2639.
(6) Dobereiner, G. E.; Crabtree, R. H. Chem. Rev. 2009, 110, 681−
703.
General Procedure for Acceptorless Methanol Dehydrogen-
ation. A Schlenk tube attached to a condenser and equipped with a
stir bar or a reactor vessel (Radleys Carousel 12 Place Reaction
Station, RR98030) was charged with iridium catalyst (0.001−0.004
mol %), potassium hydroxide (85% by mass, 1.34 g, 20 mmol), and
degassed MeOH (3 mL) under a N2 atmosphere. The resulting
solution was heated at 115 °C for 15−40 h. Upon cooling, deuterium
oxide (2 mL) was added, along with sodium acetate (34.5 mg, 0.420
mmol) as an internal standard for NMR. Methanol dehydrogenation
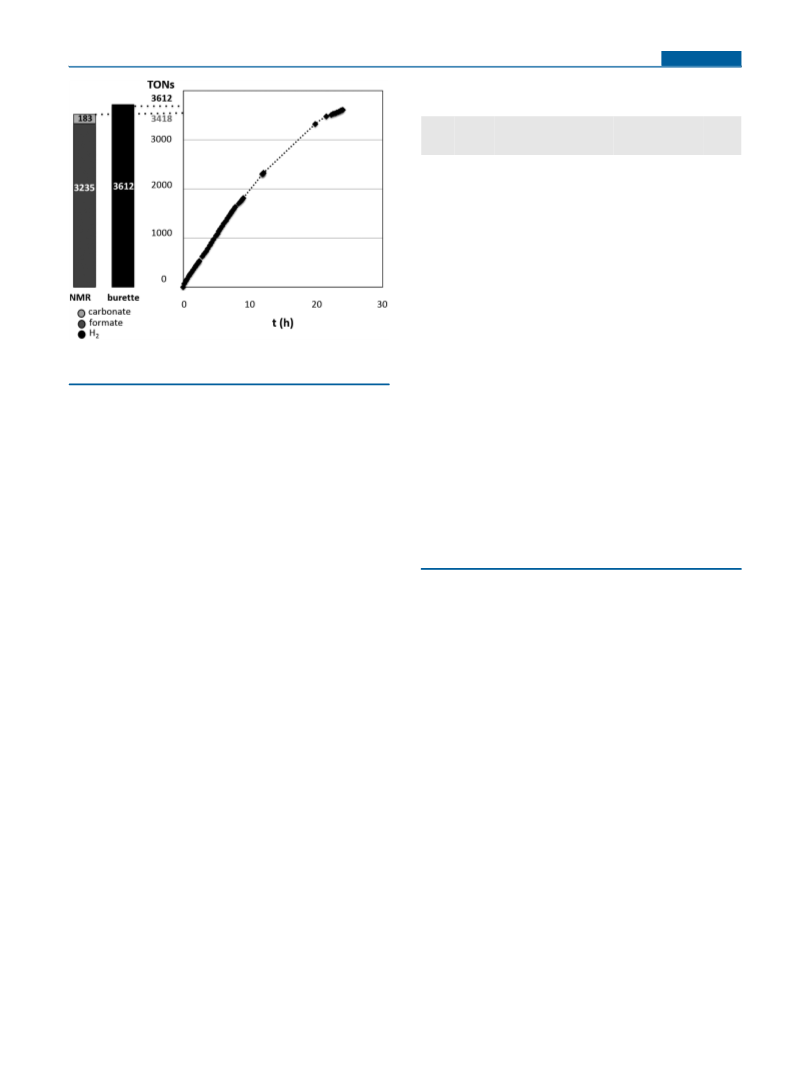
was quantified by integration of formate and carbonate signals
obtained by 1H and 13C{1H} NMR spectroscopy (see the SI for
details). TON was calculated based on the amount of H2 produced (2
times the amount of formate and 3 times the amount of carbonate).
General Procedure for TH and N-Methylation Reactions. To
a Biotage microwave vial (size 2−5 mL) were added iridium catalyst
(1.5−6.5 μmmol, 1−5 mol %) and potassium hydroxide (0.043−0.65
mmol, 0.33−5 equiv vs substrate). The vial was purged with N2, then
MeOH (0.5 mL) and substrate (0.13 mmol) were added, and the vial
was sealed and heated under MW irradiation (120 °C, 5 h). After
cooling to room temperature, CD2Cl2 (0.5 mL) and trimethox-
ybenzene (5 mg, 0.030 mmol, NMR internal standard) were added.
(7) Gunanathan, C.; Milstein, D. Science 2013, 341, DOI: 10.1126/
science.1229712.
(8) Trincado, M.; Banerjee, D.; Grutzmacher, H. Energy Environ. Sci.
̈
2014, 7, 2464−2503.
(9) (a) Watson, A. J. A.; Williams, J. M. J. Science 2010, 329, 635−
636. (b) Nixon, T. D.; Whittlesey, M. K.; Williams, J. M. J. Dalton
Trans. 2009, 753−762. (c) Guillena, G.; Ramon
́
Chem., Int. Ed. 2007, 46, 2358−2364. (d) Guillena, G.; Ramon
́
Yus, M. Chem. Rev. 2010, 110, 1611−1641. (e) Marr, A. C. Catal. Sci.
Technol. 2012, 2, 279−287.
(10) (a) Grigg, R.; Mitchell, T. R. B.; Sutthivaiyakit, S.; Tongpenyai,
N. J. Chem. Soc., Chem. Commun. 1981, 611−612. (b) Arcelli, A.; Khai,
B.-T.; Porzi, G. J. Organomet. Chem. 1982, 235, 93. (c) Kawahara, R.;
Yamamoto, K.; Yamaguchi, R. Org. Lett. 2002, 16, 2691−2694. (d) Del
Zotto, A.; Baratta, W.; Sandri, M.; Verardo, G.; Rigo, P. Eur. J. Inorg.
́
Chem. 2004, 524−529. (e) Prades, A.; Corberan, R.; Poyatos, M.;
Peris, E. Chem.Eur. J. 2008, 14, 11474−11479. (f) Fujita, K.-I.;
Enoki, Y.; Yamaguchi, R. Tetrahedron 2008, 64, 1943−1954. Examples
of amine N-alkylation using other alcohols: (g) Fujita, K.-I.;
Yamaguchi, R. Synlett 2005, 4, 560−571. (h) Hollmann, D.; Tillack,
A.; Michalik, D.; Jackstell, R.; Beller, M. Chem.Asian J. 2007, 2,
403−410. (i) Haniti, M.; Hamid, S. A.; Williams, J. M. J. Chem.
Commun. 2007, 725−727. (j) Gunathan, C.; Milstein, D. Angew.
1
The products were identified by a comparison of H NMR spectra
with those of authentic samples (all products were either commercially
1
available or previously reported). Yields were quantified by H NMR
spectroscopy.
́
Chem., Int. Ed. 2008, 47, 8661−8664. (k) Prades, A.; Corberan, R.;
ASSOCIATED CONTENT
* Supporting Information
Poyatos, M.; Peris, E. Chem.Eur. J. 2008, 14, 11474−11479.
■
(11) (a) Klomp, D.; Hanefeld, U.; Peters, J. A. In The Handbook of
Homogeneous Hydrogenation; de Vries, J. G., Elsevier, C. J., Eds.; Wiley-
S
Details of the quantitation of carbonate, H2, and CO, as well as
homogeneity studies, other control reactions, and NMR data.
This material is available free of charge via the Internet at
VCH: Weinheim, Germany, 2007. (b) Samec, J. S. M.; Backvall, J. E.;
̈
Andersson, P. G.; Brandt, P. Chem. Soc. Rev. 2006, 35, 237−248.
(12) Johnson, T. C.; Morris, D. J.; Wills, M. Chem. Soc. Rev. 2010, 39,
81−88.
(13) (a) van den Berg, A.; Areon, C. O. Chem. Commun. 2008, 668.
(b) Sartbaeva, A.; Kuznetsov, V. L.; Wells, S. A.; Edwards, P. P. Energy
Environ. Sci. 2008, 1, 79−85. (c) Armaroli, N.; Balzani, V. Angew.
Chem., Int. Ed. 2007, 46, 52−66.
AUTHOR INFORMATION
Corresponding Author
■
Author Contributions
(14) Navarro, R. M.; Pena, M. A.; Fierro, J. L. G. Chem. Rev. 2007,
107, 3952−3991.
†These authors contributed equally to this work.
(15) Dalebrook, A. F.; Gan, W.; Grasemann, S. M.; Laurenczy, G.
Chem. Commun. 2013, 49, 8735−8751.
Notes
The authors declare no competing financial interest.
(16) Crabtree, R. H. Energy Environ. Sci. 2008, 1, 134−138.
(17) Kusche, M.; Enzenberger, F.; Bajus, S.; Niedermeyer, H.;
ACKNOWLEDGMENTS
■
Bcş mann, A.; Kaftan, A.; Laurin, M.; Libuda, J.; Wasserscheid, P.
Angew. Chem., Int. Ed. 2013, 52, 5028−5032.
This material is based on work supported by a U.S. Department
of Energy, Office of Science, Office of Basic Energy Sciences,
catalysis award (Award DE-FG02-84ER13297 to J.C., L.S.S.,
M.G.M., and R.H.C.).
(18) (a) Shinoda, S.; Itagaki, H.; Saito, Y. J. Chem. Soc., Chem.
Commun. 1985, 860−861. (b) Smith, T. A.; Aplin, R. P.; Maitlis, P. M.
J. Organomet. Chem. 1985, 291, C13−C14. (c) Itagaki, H.; Saito, Y.;
Shinoda, S. J. Mol. Catal. 1987, 41, 209−220. (d) Makita, K.; Nomura,
K.; Saito, Y. J. Mol. Catal. 1994, 89, 143−150. (e) Yang, L. C.; Ishida,
T.; Yamakawa, T.; Shinoda, S. J. Mol. Catal. A: Chem. 1996, 108, 87−
93.
REFERENCES
■
(1) Palo, D. R.; Dagle, R. A.; Holladay, J. D. Chem. Rev. 2007, 107,
3992−4021.
(2) (a) Stocker, M. M. Microporous Mesoporous Mater. 1999, 29, 3−
48. (b) Keil, F. J. Microporous Mesoporous Mater. 1999, 29, 49−66.
(c) Liu, Z. M.; Sun, C. L.; Wang, G. W.; Wang, Q. X.; Cai, G. Y. Fuel
Process. Technol. 2000, 62, 161−172. (d) Olsbye, U.; Bjorgen, M.;
(19) Nielsen, M.; Alberico, E.; Baumann, W.; Drexler, H.-J.; Junge,
H.; Gladiali, S.; Beller, M. Nature 2013, 495, 85−89.
(20) Rodríguez-Lugo, R. E.; Trincado, M.; Vogt, M.; Tewes, F.;
Santiso-Quinones, G.; Grutzmacher, H. Nat. Chem. 2013, 5, 342−347.
̈
E
Inorg. Chem. XXXX, XXX, XXX−XXX

 Campos, Jesús
Campos, Jesús
 Sharninghausen, Liam S.
Sharninghausen, Liam S.
 Manas, Michael G.
Manas, Michael G.
 Crabtree, Robert H.
Crabtree, Robert H.