X. Wang, et al.
JournalofPhotochemistry&PhotobiologyA:Chemistry400(2020)112695
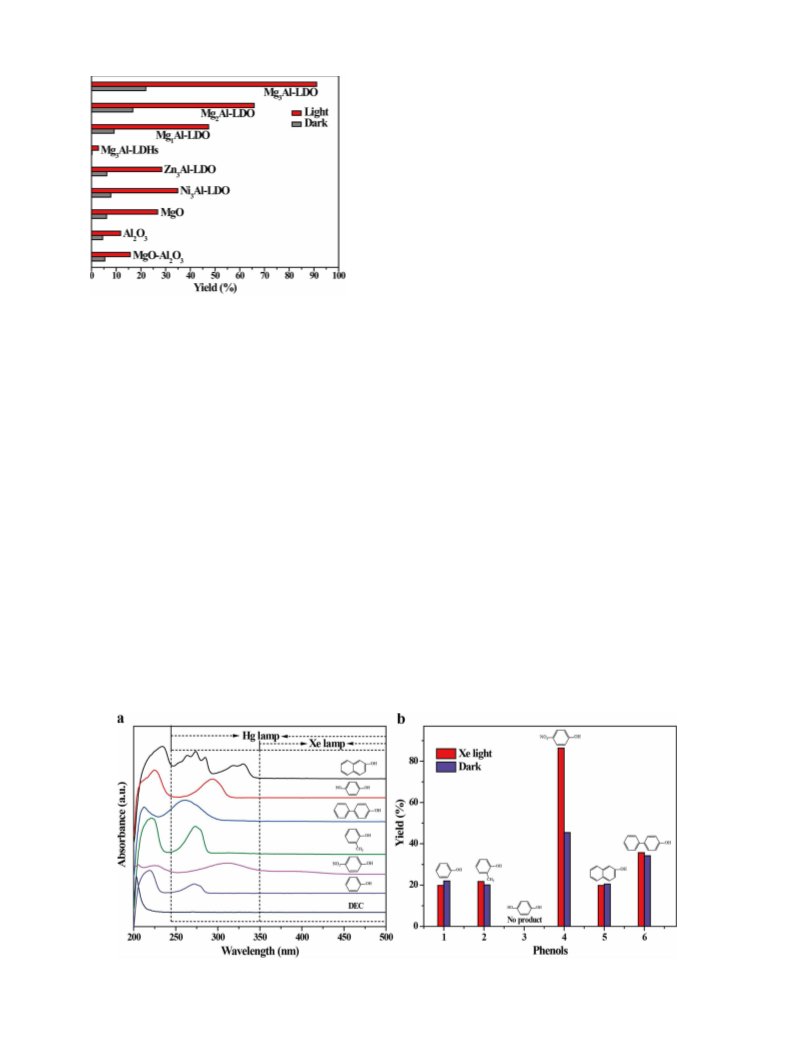
effect of light absorption of phenols and the acid–base pairs on the
catalyst simultaneously promoted the reaction.
indicated that the alkylation of phenols is difficult when using longer-
chain DEC as an alkylating agent compared with DMC. The ether yield
was 99.4 % when the ortho-position hydrogen atom of the phenolic
hydroxyl group was replaced by a methyl group (Table 1, entry 8).
When a substrate with an electron-withdrawing group in the para-po-
sition of the phenolic hydroxyl group was used, the yield of desired
ether can be up to 86.4 % at a longer reaction time of 24 h (Table 1,
entry 9) under the Xe lamp irradiation (the reason will be discussed in
detail below). Moreover, substrates containing two aromatic rings also
yield of corresponding ethers under Hg lamp irradiation. In addition,
the O-ethylation of phenol with DEC catalyzed by Ni3Al-LDO and
Zn3Al-LDO were also performed (Table S1) under Hg lamp irradiation
without the addition of a base, the phenetole yield are 34.9 % and 28.4
% respectively. It is noted that a good stoichiometric relation was
achieved between the ethanol and CO2 amounts, and the carbon mass
balance of the O-ethylation of phenol within an experimental error is
shown in Table S1. In summary, it is found that the excellent yield of
corresponding ethers can be achieved when using the long-chain al-
kylating agent of DEC with Mg3Al-LDO catalyst. In addition, all of the
O-alkylations of phenols display much higher catalytic activity in the
light than in the dark on the Mg3Al-LDO catalyst. The activation energy
is reduced by 83.3 kJ/mol under irradiation (Fig. S1), indicating that
the reaction can be promoted by the energy of light. In addition, the
hydroxyl groups in LDOs samples were basically removed as shown in
the FT-IR spectra of LDOs samples (Fig. S2b), and Mg3Al-LDO is highly
stable and can be reused several times (Fig. S4). Based on the high
efficiency of the catalytic system, the activation of phenol and DEC in
the alkylation process was investigated in detail below.
2. Experiment
2.1. Catalysts’ preparation
Layered double hydroxides (LDHs) were prepared as follows:
MgCl2·6H2O, Al(NO3)3·9H2O, and urea were dissolved in deionized
water. Then, the solution was refluxed at 365 K for 24 h. The resulting
white powder was filtered after cooling to room temperature naturally,
washed with deionized water for three times, and finally dried at 373 K
in air, designated as MgAl-LDHs. The LDHs with different molar ratios
of magnesium and aluminum can be prepared by varying the added
amounts of magnesium salt and aluminum salt. Mixed oxides were
achieved by calcining the LDHs at 723 K in air, denoted as MgAl-LDO.
Mg/Al atomic ratios of 1, 2 and 3 are denoted as Mg1Al-LDO, Mg2Al-
LDO, and Mg3Al-LDO, respectively.
2.2. Characterization
The surface area was calculated using the Brunauer–Emmett–Teller
(BET) method. Powder X-ray diffraction (XRD) spectroscopy was re-
corded with a Cu Kα radiation (λ =1.5405 Å) on a Bruker D8 Advance
diffractometer. Acidity and basicity measurements were carried out by
temperature programmed desorption (TPD) of NH3 and CO2.
Thermogravimetric/differential thermal analysis (TG/DTA) was carried
out on a Rigaku TG. Ultraviolet–visible diffuse reflectance spectroscopy
(UV–Vis DRS) was performed on a Shimadzu UV-3600 UV–vis–NIR
spectrophotometer. Fourier transform infrared spectroscopy (FT-IR)
was performed using a Nicolet Magna-IR 550-II spectrometer with KBr
pellets.
3.2. Monitoring the surface reaction
The activation of reactant molecules adsorbed on the Mg3Al-LDO
catalyst was investigated by in situ FT-IR spectra shown in Fig. 1 and
Fig. S5 (detailed explanation). The decreased intensity bands at
3374 cm–1 for the vibration bands of phenolic hydroxyl indicate the
dehydrogenation of hydroxyl in the catalytic process [22]. The dimin-
ished intensity and slight shift to lower wavenumbers of the deforma-
tion vibrations absorption band of CH2 (DEC) at 1367 cm–1 indicates
the nucleophilic attack at the methylene carbon of DEC by phenol
oxygen during the reaction. Significantly, the stretching vibration band
at 2937 cm–1 for the CeH in the phenol shifted to lower wavenumbers
compared with the neat phenol (3065 cm–1, 3047 cm–1) indicating that
the phenol molecules adsorbed on the basic sites at the catalyst surface
by hydroxyl hydrogen. In addition, the vibration band at 1722 cm–1 for
the carbonyl oxygen group in DEC molecules also shifted to lower
wavenumbers compared with the nmeat DEC (1780 cm–1), demon-
strating the carbonyl oxygen atom in the DEC molecules bonded to the
acidic sites at the surface of the catalyst.
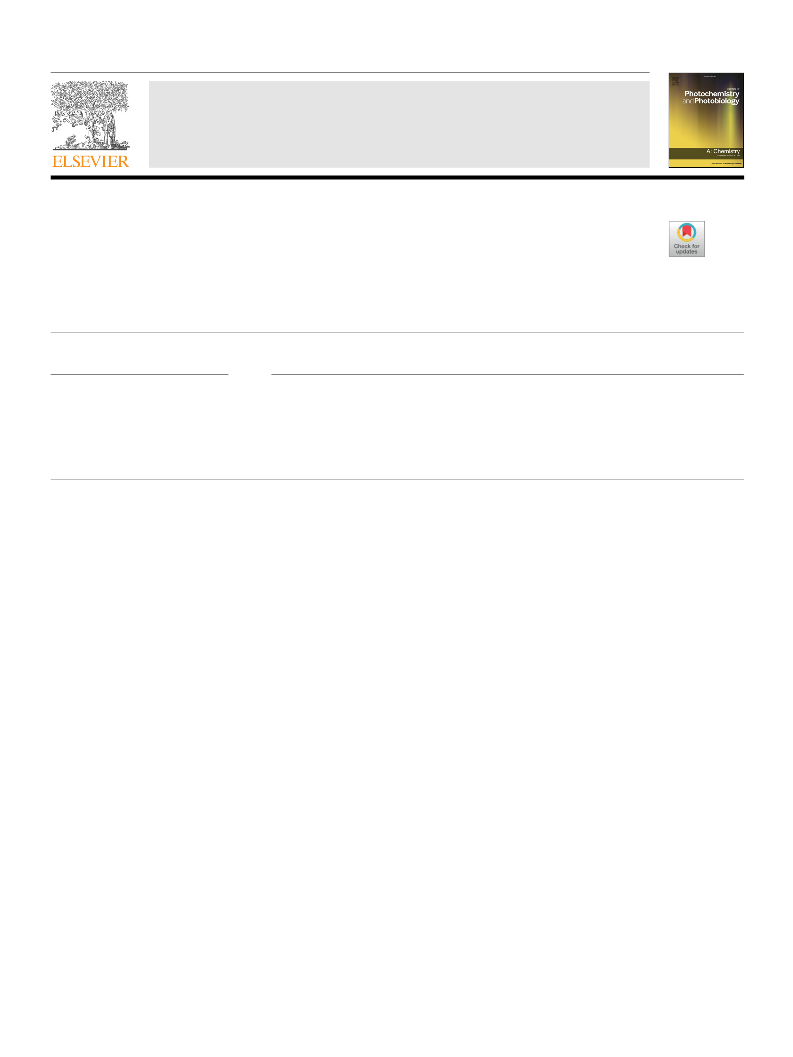
Besides, various catalysts were evaluated in the O-ethylation of
phenol with DEC to survey the influence of surface acidic and basic
surface sites shown in Fig. 2. MgAl-LDOs were used with different Mg/
Al atom ratios (Mg1Al-LDO, Mg2Al-LDO, Mg3Al-LDO). The total quan-
tity of acidic and basic sites in the Mg3Al-LDO were the most (Fig. S6),
simultaneously exhibiting the best catalytic activity with phenetole
yield of 91.0 % when irradiated by a Hg lamp. In addition, MgO and
Mg3Al-LDHs with basic sites and Al2O3 with acid sites all display low
catalytic conversion, whether under the condition of light irradiation or
no light irradiation, demonstrating that the separate basic sites or acidic
sites are not the dominating factors. Physical mixing of MgO and Al2O3
also exhibited lower catalytic activity than Mg3Al-LDO, which proved
the cooperative role of acid-base sites. It is obvious that the effect of the
specific surface area is excluded due to a smaller surface area, which
was displayed in the catalyst with the highest activity (Table S2).
Moreover, Zn3Al-LDO and Ni3Al-LDO catalysts produced phenetole
yields of 28.4 % and 34.9 %, respectively, which exhibit slightly better
2.3. Catalytic reactions
Alkylation reactions were carried out in a stainless steel autoclave
under an argon atmosphere, reactants and catalyst were added at the
same time, then stirred and heated to the required temperature. A
500 W high-pressure mercury lamp (Hg lamp) and a full-spectrum
xenon lamp (Xe lamp) were used as the light sources. The products were
analyzed on a Shimadzu 2014C GC equipped with a WondaCap 5
column. To make sure the comparison is meaningful, the similar tem-
peratures under irradiation and in the dark were maintained.
3. Results and discussion
3.1. Synthesis of phenyl alkyl ethers
Initially, O-ethylation of phenol with DEC catalyzed by Mg3Al-LDO
was performed (Table 1, entry 1) under the same condition of O-me-
thylation of phenol with DMC (Table 1, entry 5) in the presence and
absence of light at 423 K for 12 h. A phenetole yield of 91.0 % was
achieved under the high-pressure Hg lamp irradiation without the ad-
dition of a base, which exhibits better catalytic activity than in the dark
with the yield of 22.0 %. When taken hydroquinone as the reactant, the
main product was monoalkylated p-ethoxyphenol with a yield of 60.5 %
(Table 1, entry 2). However, the yield of the dialkylated product in-
creased to 47.7 % when the DMC as the alkylating agent (Table 1, entry
6). In addition, the catalytic activity of O-ethylation of hydroquinone
with DEC and O-methylation of hydroquinone with DMC at the reaction
time of 6 h were studied. When using DEC as the alkylating agent, the
yield of monoalkylated and dialkylated products were 29.4 % and 6.3
%, respectively (Table 1, entry 3). However, the yields of mono-
alkylated and dialkylated products were 37.9 % and 11.0 %, respec-
tively, when DMC was the alkylating agent, which are both higher than
that of the product of O-ethylation (Table 1, entry 7). It is clearly
2

 Wang, Ruiyi
Wang, Ruiyi
 Wang, Xiaoyu
Wang, Xiaoyu
 Zheng, Zhanfeng
Zheng, Zhanfeng