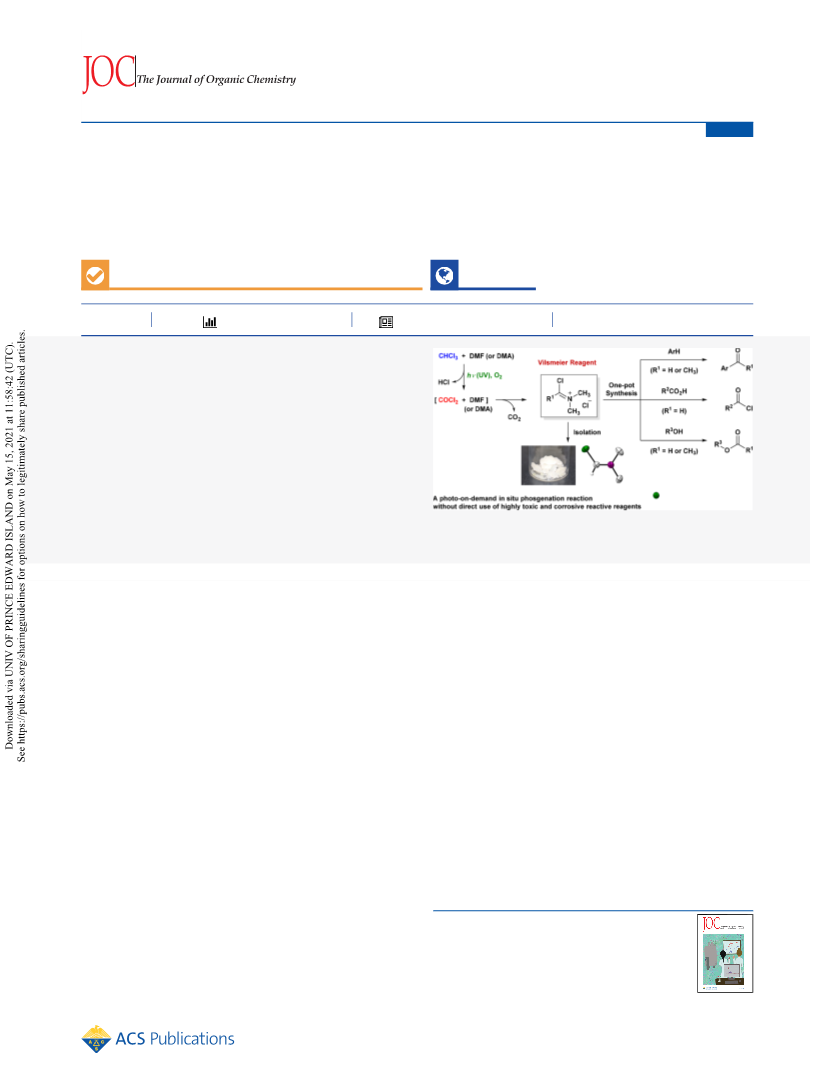
The Journal of Organic Chemistry
Article
1
a colorless liquid in 50% yield (0.73 g, 5 mmol). H and 13C NMR
spectra are in agreement with those recorded in the Spectral Database
for Organic Compounds (SDBS).40 1H NMR (400 MHz, CDCl3, 293
K): δ 7.99 (dd, J = 4.0, 1.2 Hz, 1H), 7.84 (dd, J = 5.2, 1.6 Hz, 1H),
7.21 (dd, J = 4.8, 4.0 Hz, 1H). 13C{1H} NMR (100 MHz, CDCl3, 293
K): δ 159.7, 138.0, 137.7, 137.4, 128.7.
mL, 10 mmol) were mixed in a cylindrical flask. The sample solution
was vigorously stirred upon bubbling with O2 (25 mL/min) under
exposure to the light at 30 °C for 3.5 h. The lamp was turned off, and
the sample solution was stirred at 30 °C for 2 h. Aniline (4.6 mL, 50
mmol) was added slowly at 0 °C, and then, stirred at room
temperature for 1 h. The sample solution was washed with water (30
mL) and extracted with CH2Cl2 (20 mL × 3). The combined organic
extracts were dried over anhydrous Na2SO4 and evaporated to
dryness. 1H NMR spectroscopy revealed that the residue contains 5b
in 93% yield. It was then subjected to silica gel column
chromatography (CH2Cl21) to afford 5b as a yellow solid in 67%
yield (1.36 g, 6.7 mmol). H and 13C NMR spectra are in agreement
with those reported in the literature.44 1H NMR (400 MHz, CDCl3,
293 K): δ 8.09 (brs, 1H), 7.57 (d, J = 7.6 Hz, 2H), 7.39 (t, J = 7.2 Hz,
2H), 7.21 (t, J = 7.6 Hz, 1H), 6.04 (s, 1H). 13C{1H} NMR (100
MHz, CDCl3, 293 K): δ 161.7, 136.2, 129.2, 125.7, 120.2, 66.9.
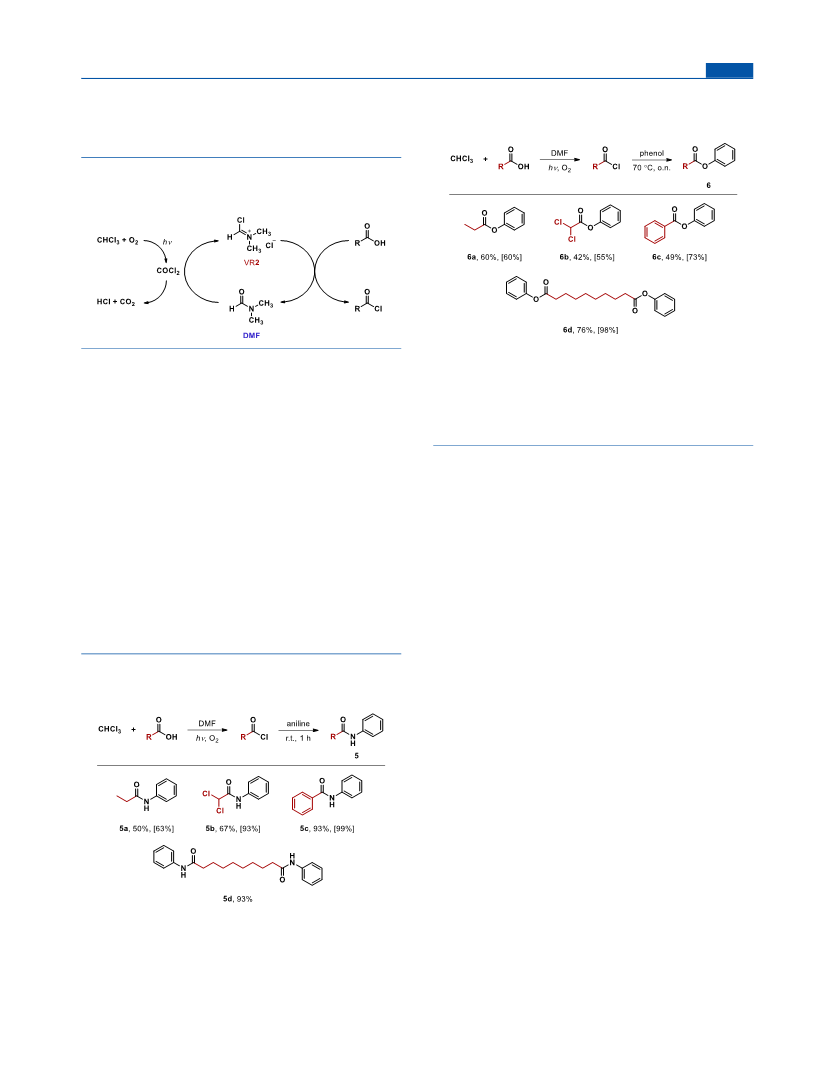
N-Phenylbenzamide (5c). Chloroform (20 mL, 250 mmol), DMF
(0.4 mL, 5 mmol), and benzoic acid (1.28 g, 10 mmol) were mixed in
a cylindrical flask. The sample solution was vigorously stirred upon
bubbling with O2 (25 mL/min) under exposure to the light at 30 °C
for 3 h. The lamp was turned off, and the sample solution was stirred
at 50 °C for 2 h. Aniline (4.6 mL, 50 mmol) was added slowly at 0 °C,
and then, stirred at room temperature for 1 h. The sample solution
was washed with water (30 mL) and extracted with CH2Cl2 (20 mL ×
3). The combined organic extracts were dried over anhydrous
4-Fluorobenzoyl Chloride (4f). Chloroform (30 mL, 250 mmol),
DMF (0.78 mL, 10 mmol), and 4-fluorobenzoic acid (1.4 g, 10
mmol) were mixed in a cylindrical flask. The sample solution was
vigorously stirred upon bubbling with O2 (25 mL/min) under
exposure to the light at 30 °C for 3.5 h. The lamp was turned off, and
the sample solution was stirred at 50 °C for 2 h. 1H NMR
spectroscopy revealed that the resulting sample solution contains 4f in
99% yield. Vacuum distillation of the sample solution afforded 4f as a
1
colorless liquid in 58% yield (0.92 g, 5.8 mmol). H and 13C NMR
spectra are in agreement with those reported in the literature.39 1H
NMR (400 MHz, CDCl3, 293 K): δ 8.19−8.14 (m, 2H), 7.22−7.17
(m, 2H). 13C{1H} NMR (100 MHz, CDCl3, 293 K): δ 168.4 (C−F,
1JC−F = 256.6 Hz), 167.0, 165.9 (C−F, 1JC−F = 256.6 Hz), 134.3 (C−
F, 3JC−F = 10.2 Hz), 134.2 (C−F, 3JC−F = 10.2 Hz), 129.6 (C−F, 4JC−F
4
2
= 2.9 Hz), 129.5 (C−F, JC−F = 2.9 Hz), 116.5 (C−F, JC−F = 21.9
Hz), 116.2 (C−F, JC−F = 21.9 Hz). 19F NMR (376 MHz, CDCl3,
2
293 K): δ −100.82, −100.80, −100.79, −100.78, −100.77, −100.76,
−100.74.
Sebacoyl Chloride (4g). Chloroform (20 mL, 250 mmol), DMF
(0.78 mL, 10 mmol), and sebacic acid (2.02 g, 10 mmol) were mixed
in a cylindrical flask. The sample solution was vigorously stirred upon
bubbling with O2 (25 mL/min) under exposure to the light at 30 °C
for 4.5 h. The lamp was turned off, and the sample solution was
1
Na2SO4. H NMR spectroscopy revealed that the sample solution
contains 5c in 99% yield. It was then evaporated to leave an orange
solid. The residue was washed with a 20:1 mixture solution of n-
hexane and CH2Cl2 to afford 5c as a yellow solid in 93% yield (1.84 g,
1
9.3 mmol). H and 13C NMR spectra are in agreement with those
1
stirred at 50 °C for 2 h. H NMR spectroscopy revealed that the
recorded in the Sigma-Aldrich FT-NMR Library (ver. 4.0.10). 1H
NMR (400 MHz, CDCl3, 293 K): δ 7.89 (d, J = 7.2 Hz, 2H), 7.79
(brs, 1H), 7.66 (d, J = 7.6 Hz, 2H), 7.58−7.49 (m, 3H), 7.39 (t, J =
7.2 Hz, 2H), 7.16 (t, J = 7.6 Hz, 2H). 13C{1H} NMR (100 MHz,
CDCl3, 293 K): δ 165.7, 137.9, 135.0, 131.9, 129.1, 128.8, 127.0,
124.6, 120.2.
resulting sample solution contains 4g in 99% yield. Vacuum
distillation of the sample solution afforded 4g as a colorless liquid
in 44% yield (1.05 g, 4.4 mmol). H and 13C NMR spectra are in
1
agreement with those recorded in the Spectral Database for Organic
Compounds (SDBS).41 1H NMR (400 MHz, CDCl3, 293 K): δ 2.89
(t, J = 7.6 Hz, 4H), 1.74−1.67 (m, 4H), 1.37−1.30 (m, 8H). 13C{1H}
NMR (100 MHz, CDCl3, 293 K): δ 173.8, 47.0, 28.8, 28.3, 25.0.
Terephthaloyl Dichloride (4h). Chloroform (30 mL, 250 mmol),
DMF (1.56 mL, 20 mmol), and terephthalic acid (1.66 g, 10 mmol)
were mixed in a cylindrical flask. The sample solution was vigorously
stirred upon bubbling with O2 (25 mL/min) under exposure to the
light at 30 °C for 6 h. The lamp was turned off, and the sample
solution was stirred at 50 °C for 2 h. 1H NMR spectroscopy revealed
that the resulting sample solution contains 4h in 82% yield. Vacuum
distillation of the sample solution afforded 4h as a colorless liquid in
79% yield (1.60 g, 7.9 mmol). 1H and 13C NMR spectra are in
agreement with those reported in the literature.42 1H NMR (400
MHz, CDCl3, 293 K): δ 8.26 (s, 4H). 13C{1H} NMR (100 MHz,
CDCl3, 293 K): δ 167.5, 138.3, 131.4.
N1,N10-Diphenyldecanediamide (5d). Chloroform (20 mL, 250
mmol), DMF (0.78 mL, 10 mmol), and sebacic acid (2.02 g, 10
mmol) were mixed in a cylindrical flask. The sample solution was
vigorously stirred upon bubbling with O2 (25 mL/min) under
exposure to the light at 30 °C for 4.5 h. The lamp was turned off, and
the sample solution was stirred at 50 °C for 2 h. Aniline (4.6 mL, 50
mmol) was added slowly at 0 °C and stirred at room temperature for
1 h to give a precipitate. It was then collected through filtration and
washed with methanol to give 5d as a white solid in 93% yield (3.29 g,
1
9.3 mmol). mp 199−202 °C. H NMR (400 MHz, DMSO-d6, 293
K): δ 9.83 (brs, 2H), 7.59 (d, J = 7.6 Hz, 4H), 7.27 (t, J = 7.2 Hz,
4H), 7.01 (t, J = 7.6 Hz, 2H), 2.86 (t, J = 7.2 Hz, 4H), 1.60−1.57 (m,
4H), 1.30 (m, 8H). 13C{1H} NMR (100 MHz, DMSO-d6, 293 K): δ
171.1, 139.2, 128.5, 122.8, 118.9, 36.3, 28.6, 28.6, 25.0. IR (ATR) ν
(cm−1): 3310, 2928, 2851, 1655, 1597, 1534, 1442, 755, 712, 687.
HRMS (ESI) m/z: [M + H]+ Calcd for C22H28N2O2 353.2224;
Found: 353.2216.
N-Phenylpropionamide (5a). Chloroform (20 mL, 250 mmol),
DMF (0.4 mL, 5 mmol), and propionic acid (0.67 mL, 10 mmol)
were mixed in a cylindrical flask. The sample solution was vigorously
stirred upon bubbling with O2 (25 mL/min) under exposure to the
light at 30 °C for 3 h. The lamp was turned off, and the sample
solution was stirred at 30 °C for 2 h. Aniline (4.6 mL, 50 mmol) was
added slowly at 0 °C, and then, stirred at room temperature for 1 h.
The sample solution was washed with water (30 mL) and extracted
with CH2Cl2 (20 mL × 3). The combined organic extracts were dried
over anhydrous Na2SO4 and evaporated to dryness. 1H NMR
spectroscopy revealed that the residue contains 5a in 63% yield. It
was then subjected to silica gel column chromatography (n-hexane/
CH2Cl2, v/v = 1:2) to afford 5a as a yellow solid in 50% yield (0.75 g,
Phenyl Propionate (6a). Chloroform (20 mL, 250 mmol), DMF
(0.4 mL, 5 mmol), and propionic acid (0.67 mL, 10 mmol) were
mixed in a cylindrical flask. The sample solution was vigorously stirred
upon bubbling with O2 (25 mL/min) under exposure to the light at
30 °C for 3 h. The lamp was turned off, and the sample solution was
stirred at 30 °C for 2 h. Phenol (1.88 g, 20 mmol) disssolved in 2 mL
of CHCl3 was added slowly at 0 °C, and then, refluxed for overnight.
1H NMR spectroscopy revealed that the sample solution contains 6a
in 60% yield. It was then evaporated to dryness and washed with hot
water (>65 °C, 30 mL × 7). The organic layer was dried over
anhydrous Na2SO4 and evaporated to dryness. The residue was
subjected to silica gel column chromatography (CH2Cl2) to afford 6a
5.0 mmol). H and 13C NMR spectra are in agreement with those
1
reported in the literature.43 1H NMR (400 MHz, CDCl3, 293 K): δ
7.52 (d, J = 8.0 Hz, 2H), 7.32 (t, J = 7.6 Hz, 2H), 7.10 (t, J = 7.2 Hz,
1H), 2.43 (q, J = 7.6 Hz, 2H), 1.26 (t, J = 7.6 Hz, 3H). 13C{1H}
NMR (100 MHz, CDCl3, 293 K): δ 171.9, 137.9, 129.0, 124.2, 119.7,
30.8, 9.7.
1
as a colorless liquid in 60% yield (0.91 g, 6.0 mmol). H and 13C
NMR spectra are in agreement with those recorded in the Spectral
Database for Organic Compounds (SDBS).45 1H NMR (400 MHz,
CDCl3, 293 K): δ 7.37 (t, J = 7.6 Hz, 2H), 7.22 (t, J = 7.2 Hz, 1H),
7.09 (d, J = 8.0 Hz, 2H), 2.62 (q, J = 7.2 Hz, 2H), 1.27 (t, J = 7.6 Hz,
2,2-Dichloro-N-phenylacetamide (5b). Chloroform (20 mL, 250
mmol), DMF (0.4 mL, 5 mmol), and 2,2-dichloroacetic acid (0.82
6514
J. Org. Chem. 2021, 86, 6504−6517

 Liang, Fengying
Liang, Fengying
 Eda, Kazuo
Eda, Kazuo
 Okazoe, Takashi
Okazoe, Takashi
 Wada, Akihiro
Wada, Akihiro
 Mori, Nobuaki
Mori, Nobuaki
 Konishi, Katsuhiko
Konishi, Katsuhiko
 Tsuda, Akihiko
Tsuda, Akihiko