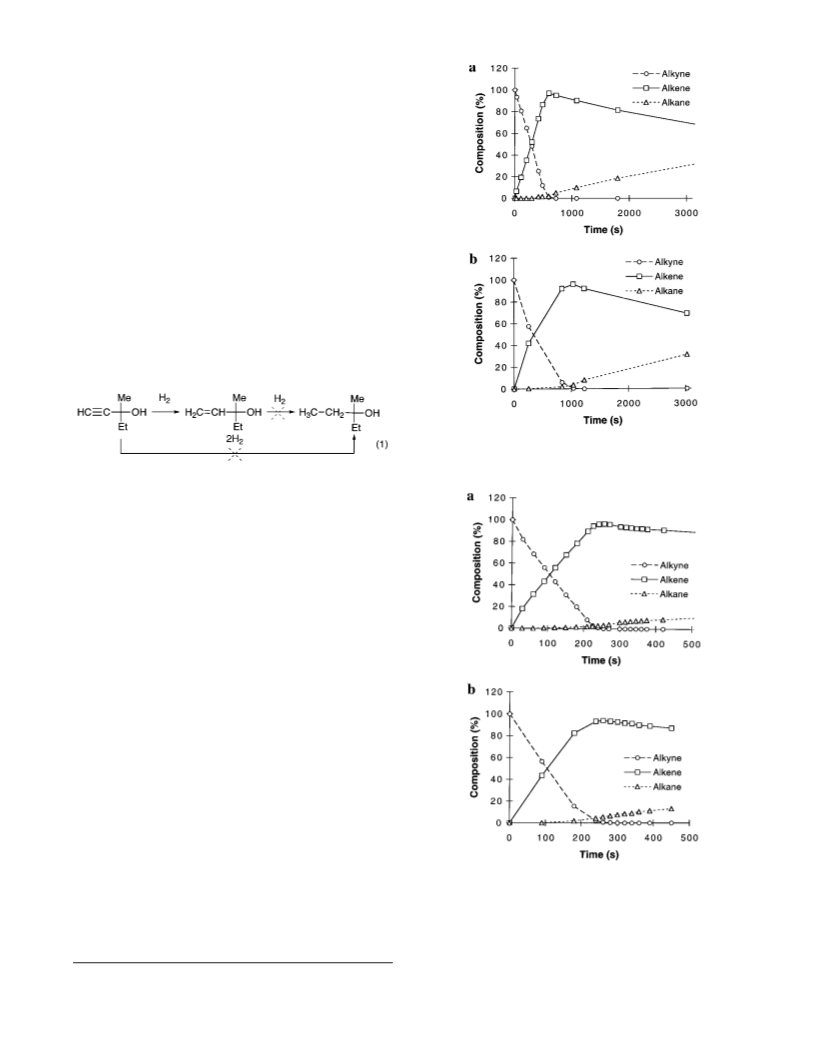
1
648 J . Org. Chem., Vol. 66, No. 5, 2001
Spee et al.
Furthermore, palladium can be alloyed with copper,
silver, platinum, and other metals to improve the
Sch em e 1. P r ep a r a tion of Lith iu m
Di(4-tolyl)cu p r a te a n d P d Cu /SiO
2
selectivity.2
9,36-40
Recently, Nosova et al. reported on a
41
bimetallic palladium-copper catalyst supported on alu-
mina with which they were able to selectively semihy-
drogenate vinyl acetylene to butadiene in the gas-phase
at low temperatures. Analogous results were published
by another group.42
In contrast to gas-phase reactions, which are less
sensitive to mass-transport limitations, liquid-phase
reactions with supported catalysts tend to exhibit mass-
transport limitation due to the smaller diffusion coef-
ficients in liquids.43 The support bodies for liquid-phase
catalysts should have dimensions within the range of 3
to about 50 µm, not only to avoid transport limitations,
but also to minimize problems with separation of the
catalyst from the liquid. With support bodies of this size,
the high-temperature hydrogen reduction of the metal
salt precursor(s) to catalysts may present problems such
as blow-out of the solid or channelling. Furthermore, such
reduction does not guarantee the formation of alloy
phases.4 Therefore, catalysts for liquid-phase reactions
are mainly restricted to supported metals of which the
salts are readily reduced at ambient temperature or to
unsupported metals in highly disperse form like Raney
metals. To avoid high-temperature reduction, it is also
possible to prepare metal particles by decomposing
4,45
of a lithium cuprate and a palladium(II) salt to give a
bimetallic-organometallic intermediate, in which copper
and palladium are linked by bridging organic ligands.
Subsequent decomposition of this intermediate gives an
Pd-Cu alloy that is deposited. This deposition proceeds
at rt and does not require heating. The atomic mixing of
the two metals present in the precursor prior to deposi-
tion leads to a homogeneous combination of the metals
in the metallic phase, and consequently to more active
and/or selective catalysts.
organometallic complexes, chemically, thermally, or by
reduction.4
6-53
For example, Cocco et al. prepared sup-
ported palladium-platinum alloys by reaction of a mix-
ture of palladium and platinum allyl compounds in
pentane with the surface hydroxyl groups of the support,
5
4
followed by reduction with hydrogen.
2
The PdCu/SiO catalysts prepared according to an
We here report a fast and facile preparation of sup-
ported bimetallic catalysts, including those of less noble
metals, which is based on the reduction of organometallic
compounds on the surface of a support in solution at room
temperature (rt). This method circumvents high-temper-
ature activation, and the catalyst can be used directly
optimized protocol have been used in the liquid-phase
semihydrogenation of several multifunctionalized acety-
lenes and propargylic alcohols.
Resu lts a n d Discu ssion
after the deposition of the metal particles. On the basis
Catalyst P r epar ation . On the basis of earlier work,55,56
silica-supported palladium-copper catalysts were pre-
pared using lithium di(4-tolyl)cuprate (see Scheme 1).
Addition of a toluene solution of the cuprate 2 to a
solution of palladium(II) acetate in toluene at rt most
probably leads to the in situ formation of a thermally
unstable organocopper-palladium complex in which the
lithium atoms of 2 have been replaced by the more
electronegative precious metal. Evidence for the forma-
tion of such a thermally labile mixed metal intermediate
can be found in the isolation and characterization of
of our earlier work,5
5,56
silica-supported bimetallic pal-
ladium-copper catalysts have been prepared by reaction
(
(
(
36) Carturan, G.; Cocco G. J . Mol. Catal. 1984, 26, 375.
37) Leviness, S.; Nair, V.; Weiss, A. H. J . Mol. Catal. 1984, 25, 131.
38) Aduriz, R. H.; Bodnariuk, P.; Coq, B.; Figueras, F. J . Catal.
1
991, 129, 47.
(39) Frevel, L. K.; Kresley, L. J . U.S. Patent 2 802 889, 1957.
(40) Boitiaux, J . P.; Cosyns, J . E.P. Patent 0 089 252, 1983.
(41) Nosova, L. V.; Zaikovskii, V. I.; Kalkinkin, A. V.; Talzi, E. P.;
Paukstitis, E. A.; Ryndin, Xu. A. Kinet. Catal. 1995, 36, 328.
42) Renouprez, A. J .; Lebas, K.; Bergeret, G.; Rousset, J . L.;
Delich e` re, P. Stud. Surf. Sci. Catal. 1996, 101, 1105.
43) Cussler, E. L. Diffusion: Mass transfer in fluid systems;
Cambridge University Press: Cambridge, 1984; Chapter 2 and 9.
44) Ruiz-Vizcaya, M. E.; Novaro, O.; Ferreira, J . M.; Gomez, R. J .
Catal. 1978, 51, 108.
(
copper bis([(dimethylamino)methyl]phenyl)aurate, [Au
2
-
(
Cu (C CH NMe -2) ], prepared by the transmetalation
2
6
H
4
2
2
4
(
66
of the lithium aurate with copper(I) bromide. The
(
(
(
(
45) Grill, M.; Gonzales, R. D. J . Catal. 1980, 64, 487.
46) Cymbaluk, T. H. U.S. Patent 4 659 687, 1987.
47) Schwartz, J . J . Mol. Catal. 1992, 72, 373.
(55) Cohen, M. S.; Noltes, J . G.; van Koten, G. U.S. Patent 4 152
303, 1979.
(56) Cohen, M. S.; Noltes, J . G.; van Koten, G. U.S. Patent 4 222
898, 1980.
(57) J ohnson, E. L.; McEwen, J . J . Am. Chem. Soc. 1926, 48, 475.
(58) Smith, L. I.; Hoehn, H. H. J . Am. Chem. Soc. 1941, 63, 1176.
(59) Pittman, C.; Olah, G. J . Am. Chem. Soc. 1965, 87, 5632.
(60) Brandsma, L.; Verkruijsse, H. D. Synthesis of Acetylenes, Allenes
and Cumulenes; Elsevier Scientific Publishing Company: Amsterdam,
1981.
(61) Brandsma, L.; Verkruijsse, H. D. Preparative Acetylenic Chem-
istry, 2nd ed.; Elsevier: Amsterdam, 1988.
(62) Fleming, I.; Takai, K.; Thomas, A. P. J . Chem. Soc., Perkin
Trans. 1 1987, 2269.
(63) Steenwinkel, P.; J ames, S. L.; Grove, D. M.; Veldman, N.; Spek,
A. L.; van Koten, G. Chem. Eur. J . 1996, 2, 1440.
48) Didillon, B.; Candy, J . P.; Le Peletier, F.; Ferretti, O. A.; Basset,
J . M. Stud. Surf. Sci. Catal. 1993, 78, 147.
49) Usui, K.; Nishiyama, Y.; Arai, M. J . Chem. Soc., Chem.
Commun. 1993, 1853.
(
(
(
50) Nogin, Y. N. Catal. Lett. 1994, 23, 79.
51) N e´ dez, C.; Lefebvre, F.; Choplin, A.; Niccolai, G. P.; Basset, J .
M.; Benazzi, E. J . Am. Chem. Soc. 1994, 116, 8638.
52) Matsuda, T.; Shizuta, M.; Yoshizawa, J .; Kikuchi, E. Appl.
Catal. 1995, 125, 293.
53) Abis, L.; Dell′Amico, D.; Calderazzo, F.; Caminiti, R.; Garbassi,
F.; Ianelli, S.; Pelizzi, G.; Robino, P.; Tomei, A. J . Mol. Catal. 1996,
(
(
1
08, L113.
54) Cocco, G.; Carturan, G.; Enzo, S.; Schiffini, L. J . Catal. 1984,
5, 405.
(
8

 Spee
Spee
 Boersma
Boersma
 Meijer
Meijer
 Slagt
Slagt
 Van Koten
Van Koten
 Geus
Geus