
Russian Chemical Bulletin p. 2110 - 2116 (1998)
Update date:2022-08-11
Topics:
 Denisov
Denisov
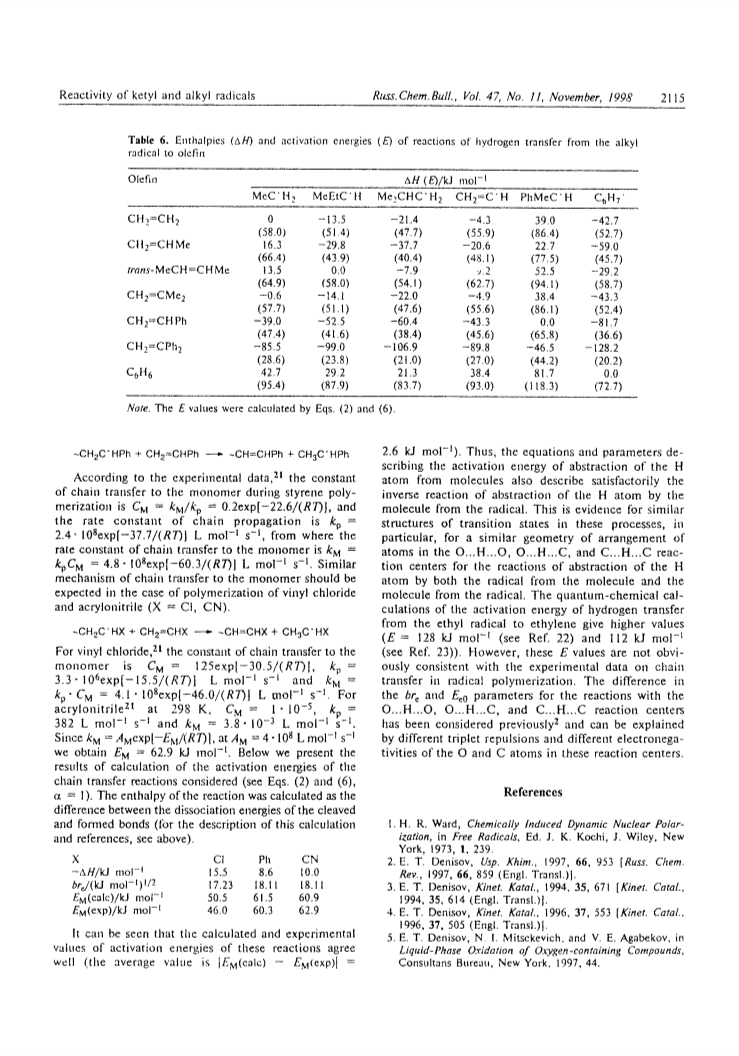
A parabolic model of bimolecular radical reactions was used for analysis of the hydrogen transfer reactions of ketyl radicals: >C+OH + R1COR2 → >C=O + R1R2C+OH. The parameters describing the reactivity of the reagents were calculated from the experimental data. The parameters that characterize the reactions of ketyl and alkyl radicals as hydrogen donors with olefins and with carbonyl compounds were obtained: >C+OH + R1CH=CH2 → >C=O + R1C+ HCH3; >R1CH=CH2 + R2C+HCH2R3 → R2C+HCH3 + R2CH=CHR3. These parameters were used to calculate the activation energies of these transformations. The kinetic parameters of reactions of hydrogen abstraction by free radicals and molecules (aldehydes, ketones, and quinones) from the C-H and O-H bonds were compared.
View More





He Bei Shun Er Chemical Co., LTD.
Contact:86-0311-86996932/86860168
Address:No 18,North street
Hangzhou Hysen Pharma co.,Ltd.
website:http://www.hysenpharma.cn/
Contact:0086-571-88298791
Address:#701,Gudun Road Hangzhou
Contact:+86-13666670345
Address:Agricultural Development Zone, Haining, Jiaxing, Zhejiang
Contact:.+86-579-85566777
Address:12222222222dsadsdsdeeeee
Contact:+86-570-4336358
Address:No.87 Building,Tianqian,Sidu Town
Doi:10.1515/znb-1968-0502
(1968)Doi:10.1002/adsc.200800501
(2009)Doi:10.1002/chem.200500918
(2006)Doi:10.1080/00304948.2016.1206424
(2016)Doi:10.1021/jo972183m
(1998)Doi:10.1016/j.bmcl.2014.05.033
(2014)