M. Brewer / Tetrahedron Letters 47 (2006) 7731–7733
7733
4. Pross, A.; Sternhell, S. Aust. J. Chem. 1970, 23, 989–
1003.
5. Barton, D.; Bashiardes, G.; Fourrey, J. Tetrahedron Lett.
1983, 24, 1605–1608.
6. Barton, D.; Bashiardes, G.; Fourrey, J. Tetrahedron 1988,
44, 147–162.
7. Rozen, S.; Zamir, D. J. Org. Chem. 1991, 56, 4695–4700.
8. Fry, A. J.; Cawse, J. N. J. Org. Chem. 1967, 32, 1677–
1679.
9. Todd, D. Org. React. 1948, 4, 378–422.
10. Mancuso, A. J.; Huang, S.-L.; Swern, D. J. Org. Chem.
1978, 43, 2480–2482.
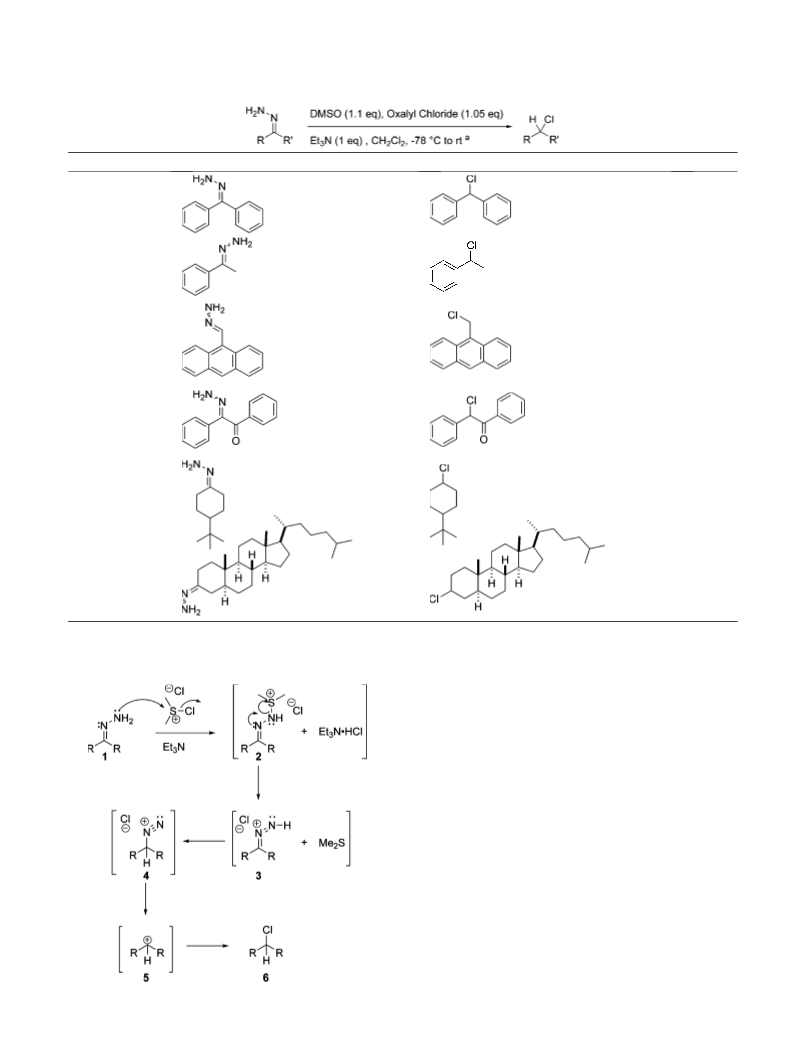
Scheme 2.
11. A typical experimental procedure follows: oxalyl chloride
(0.18 mL, 2.1 mmol, 1.05 equiv) was added to a À55 °C
solution of dimethyl sulfoxide (0.16 mL, 2.2 mmol,
1.10 equiv) in dichloromethane (15 mL) and the reaction
was maintained at this temperature until gas evolution
had ceased. The solution was then cooled to À78 °C and
a mixture of benzophenone hydrazone (0.39 g, 2.0 mmol,
1 equiv) and triethylamine (0.28 mL, 2.0 mmol, 1 equiv)
in dichloromethane (5 mL) was added in a dropwise
manner. The cooling bath was removed and the reaction
was allowed to warm to room temperature over which
time gas evolution was noted. The reaction was washed
with water (20 mL), the aqueous layer was extracted with
fresh dichloromethane (10 mL), the organics were com-
bined, washed with aqueous 10% HCl (10 mL), brine
(10 mL), dried (MgSO4), and concentrated to provide
0.38 g (93% yield) of chlorodiphenylmethane in greater
than 95% purity as determined by proton NMR and GC
analysis.
The proton NMR spectrum of this material revealed
that 93% of the protons on nitrogen had exchanged
for deuterium. Upon subjecting this material to the
above reaction conditions, a 92:8 mixture of chloro-
deutrodiphenylmethane and chlorodiphenylmethane
was isolated in a 90% yield. This ratio is consistent with
the proposed mechanism and unambiguously shows the
origin of the transferred proton. This also highlights the
utility of this methodology as a facile means to incorpo-
rate deuterium into organic substrates.
We are currently working to develop these reaction con-
ditions into a method for the preparation and isolation
of diazo compounds.
Acknowledgements
12. All products display characterization data identical to
literature values.
13. An alternative, but similar, mechanism can be written in
which the internal nitrogen of the hydrazone acts as the
initial site of reactivity.
14. Pross and Sternhell reported the formation of 2-chloro-1-
phenylpropane in a 9% yield upon treatment of the
hydrazone of benzyl methyl ketone with chlorine. [Pross,
A.; Sternhell, S. Aust. J. Chem. 1971, 24, 1437–1447.] They
propose a mechanism in which an electrophilic addition of
chlorine to the carbon–nitrogen double bond leads to a
diimide derivative that reacts further to provide the alkyl
chloride. I would like to thank a reviewer for noting that
in the present work an alternative mechanism (similar to
that proposed by Pross and Sternhell) could be written in
which the dimethylchlorosulphonium ion adds chlorine to
the carbon–nitrogen double bond.
The financial support from the University of Vermont is
gratefully acknowledged. The support from the Ver-
mont Experimental Program to Stimulate Competitive
Research (Grant # EPS0236976) is gratefully acknowl-
edged. Amgen is gratefully acknowledged for the finan-
cial support of this research in the form of an Amgen
´
new faculty award. I thank Jose Madalengoitia for his
helpful discussions about this work.
References and notes
1. Holton, T. L.; Shechter, H. J. Org. Chem. 1995, 60, 4725–
4729.
2. Regitz, M.; Mass, G. Diazo Compounds Properties and
Synthesis; Academic Press: Orlando, 1986.
3. Bo¨shar, M.; Fink, J.; Heydt, H.; Wagner, O.; Regitz, M.
In Methoden Der Organischen Chemie (Ho¨uben-Weyl);
Klaman, D., Hagemann, H., Eds.; Georg Thieme: New
York, 1990; Vol. E14B, pp 996–1010.
15. Maskill, H.; Whiting, M. C. J. Chem. Soc., Perkin Trans. 2
1976, 13, 1462–1470.
16. Debono, M.; Molloy, R. M. J. Org. Chem. 1969, 34, 1454–
1455.

 Brewer, Matthias
Brewer, Matthias