
Chemistry - A European Journal p. 9546 - 9550 (2016)
Update date:2022-08-28
Topics:
 Chen, Wenxin
Chen, Wenxin
 Tao, Huachen
Tao, Huachen
 Huang, Wenhao
Huang, Wenhao
 Wang, Guoqiang
Wang, Guoqiang
 Li, Shuhua
Li, Shuhua
 Cheng, Xu
Cheng, Xu
 Li, Guigen
Li, Guigen
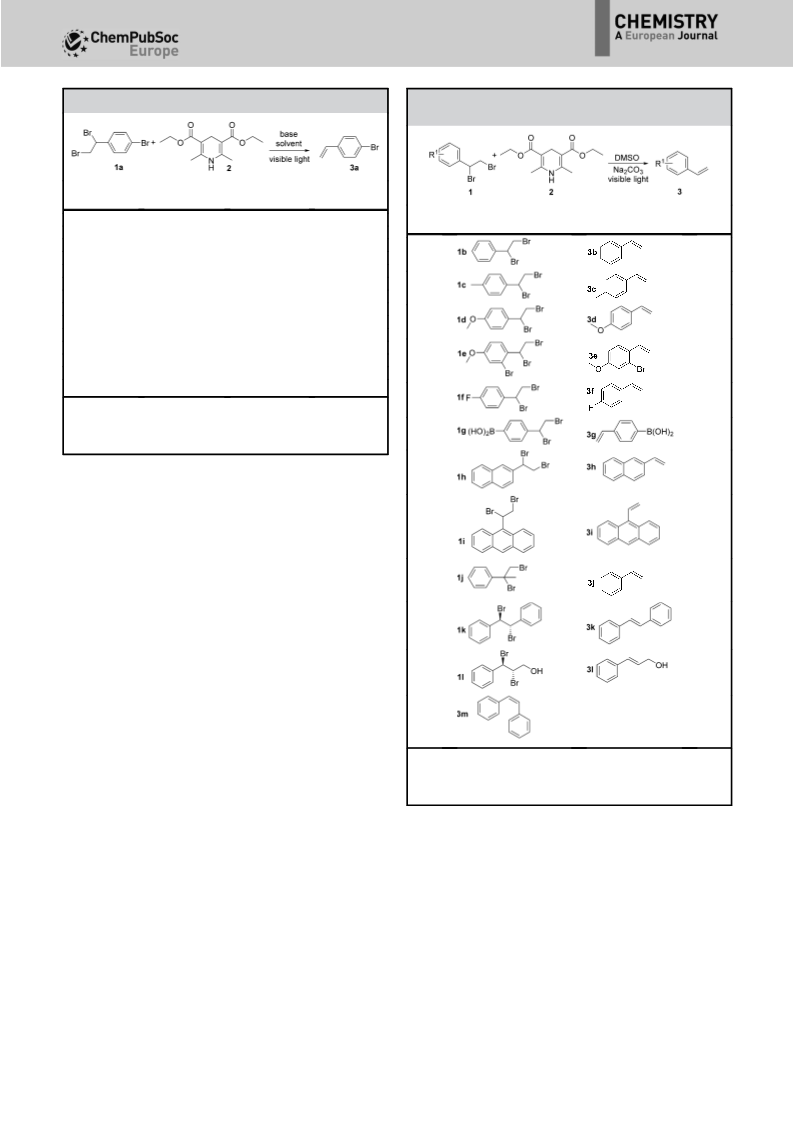
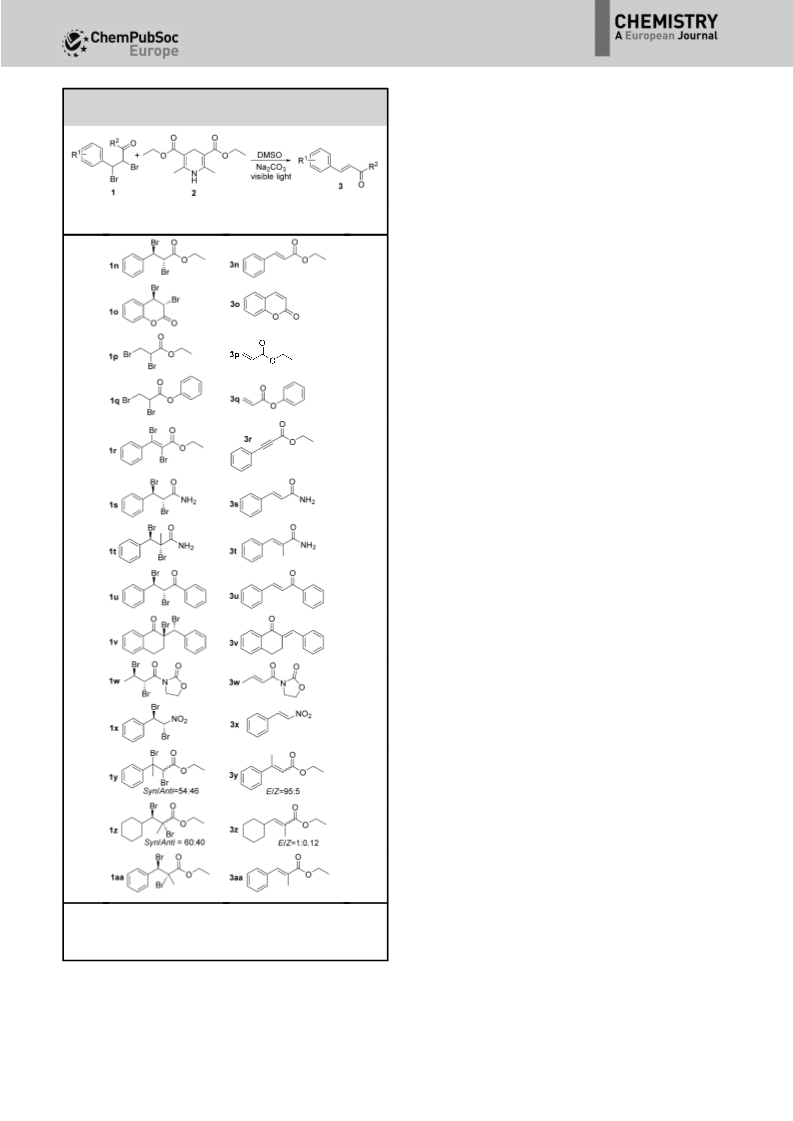
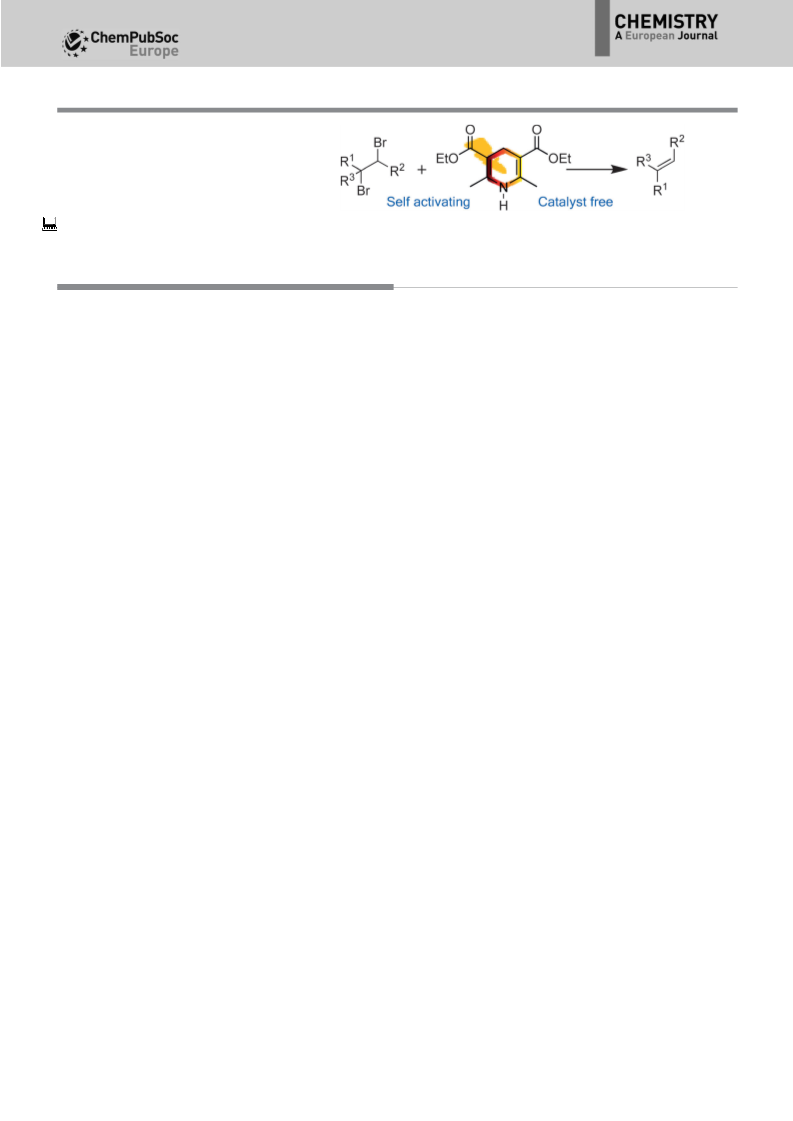
The debromination of vicinal dibromo compounds to generate alkenes usually requires harsh reaction conditions and the addition of catalysts. Just recently the visible-light-induced debromination of vicinal dibromo compounds emerged as a possible alternative to commonly used methods, but the substrate scope of this reaction is limited and a photocatalyst is necessary for the successful conversion of the starting compounds. A catalyst-free visible-light-induced debromination of vicinal dibromo compounds with a base-activated Hantzsch ester as photosensitizer is reported. The method has a wide substrate scope and a broad functional-group compatibility.
View More


Jinan Chenghuishuangda Chemical CO.,Ltd
website:http://www.jnchsd.com
Contact:+86-531-58897082
Address:No.2350, Kaituo Road, High-tech Development District, Jinan City, Shandong, China
Shijiazhuang Sdyano Fine Chemical Co., Ltd
Contact:+86-311-89830448
Address:NO.48 Ta Nan Road,Yuhua District,Shijiazhuang,Hebei,China
Kaiping Genuine Biochemical Pharmaceutical Co.,Ltd.
Contact:+86-750-2881198
Address:No.1, Xinke Road, Shatang Town, Kaiping, Guangdong Province, P.R.China
Hubei Onward Bio-Development Co., Ltd.
Contact:+86-718-8417012
Address:No.517,Shizhou Avenue,Enshi City,Hubei Province,China,445002
Contact:+86-570-4336358
Address:No.87 Building,Tianqian,Sidu Town
Doi:10.1016/j.molcatb.2015.04.005
(2015)Doi:10.1016/S0040-4039(00)85110-2
(1986)Doi:10.1016/S0040-4039(97)00350-X
(1997)Doi:10.3390/molecules22091523
(2017)Doi:10.1016/0039-128X(80)90048-3
(1980)Doi:10.1016/j.tetasy.2003.09.016
(2003)