10.1002/chem.201604904
Chemistry - A European Journal
COMMUNICATION
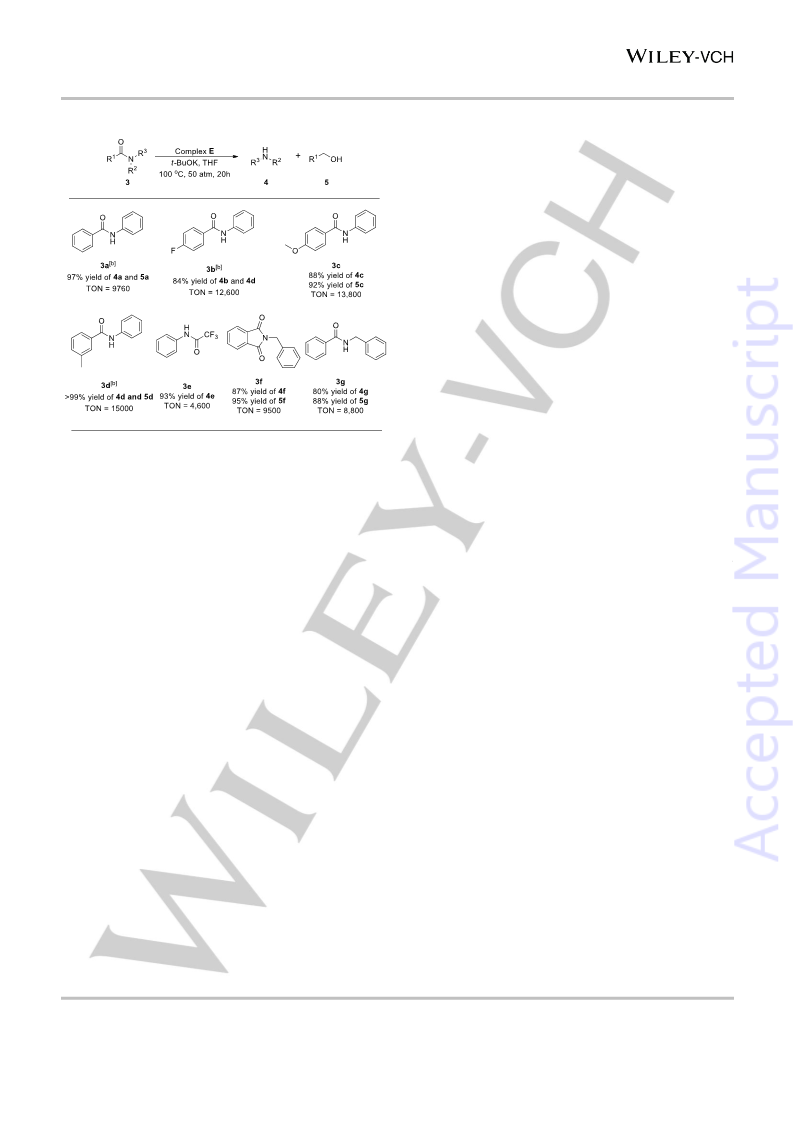
Table 3. Substrate scope using Complex E as the catalytic precursor.[a]
[1]
[2]
P. A. Dub, T. Ikariya, ACS Catal. 2012, 2, 1718-1741.
J. Seyden-Penne, Reductions by the Alumino and Borohydrides in
Organic Synthesis, 2nd ed., Wiley-VCH, Weinheim, 1997.
[3]
Examples for heterogeneous catalysts: a) B. Wojcik, H. Adkins, J. Am.
Chem. Soc. 1934, 56, 2419; b) R. M. King, US Pat. 4,448,998, May 15th,
1984; c) C. Hirosawa, N. Wakasa, T. Fuchikami, Tetrahedron Lett. 1996,
37, 6749; d) G. Beamson, A. J. Papworth, C. Philipps, A. M. Smith, R. J.
Whyman, J. Catal. 2011, 278, 228; e) G. Beamson, A. J. Papworth, C.
Philipps, A. M. Smith, R. J. Whyman, Adv. Synth. Catal. 2010, 352, 869;
f) G. Beamson, A. J. Papworth, C. Philipps, A. M. Smith, R. J. Whyman,
J. Catal. 2010, 296, 93. g) J. Coetzee, H. G. Manyar, C. Hardacre, D. J.
Cole-Hamilton, ChemCatChem, 2013, 5, 2843–2847.
[4]
[5]
[6]
S. Werkmeister, K. Junge, M. Beller, Org. Process Res. Dev. 2014, 18,
289-302.
M. Kilner, D. V. Tyers, S. P. Crabtree, M. A. Wood, PCT Int. Pat. Appl.
WO 03/093208A1, Nov 13th, 2003.
Examples for homogeneous catalysts: a) A. A. Nunez Magro, G. R.
Eastham, D. J. Cole-Hamilton, Chem. Commun. 2007, 3154-3156; b) M.
A. A. Nunez, G. R. Eastham, D. J. Cole-Hamilton, Chem Commun. 2012,
48, 12249-12250; c) K. Beydoun, T. vom Stein, J. Klankermayer, W.
Leitner, Angew. Chem. 2013, 125, 9733−9736; Angew. Chem., Int. Ed.
2013, 52, 9554-9557; d) J. Coetzee, D. L. Dodds, J. Klankermayer, S.
Brosinski, W. Leitner, A. M. Z. Slawin, D. J. Cole-Hamilton, Chem. - Eur.
J. 2013, 19, 11039-11050; e) Y. Li, I. Sorribes, T. Yan, K. Junge, M. Beller,
Angew. Chem. 2013, 125, 12378−12382; Angew. Chem., Int. Ed. 2013,
52, 12156-12160; f) Y. Kita, T. Higuchi, K. Mashima, Chem. Commun.
2014, 50, 11211-11213; (g) M. L. Yuan, J. H. Xie, S. F. Zhu and Q.-L.
Zhou, ACS Catal., 2016, 6, 3665-3669.
[a] The reaction was conducted in 4 mL of THF, Complex E was used as
catalytic precursor, Complex E 0.0005mmol, t-BuOK (1%-2% of substrates),
100 oC, 50 bar of H2, 20 h, unless otherwise noted. The reaction scale: 5 mmol
of 3a, 7.5 mmol of 3b-3d, 2.5 mmol of 3e, 5 mmol of 3f and 3g. Isolated yields.
[b] GC yields.
[7] a) M. Ito, A. Sakaguchi, C. Kobayashi, T. Ikariya, J. Am. Chem. Soc. 2007,
129, 290-291. b) M. Ito, L. W. Koo, A. Himizu, C. Kobayashi, A.
Sakaguchi, T. Ikariya, Angew. Chem. 2009, 121, 1350; Angew. Chem.,
Int. Ed. 2009, 48, 1324-1327. c) M. Ito, C. Kobayashi, A. Himizu, T.
Ikariya, J. Am. Chem. Soc. 2010, 132, 11414-11415. d) M. Ito, T.
Ootsuka, R. Watari, A. Shiibashi, A. Himizu, T. Ikariya, J. Am. Chem. Soc.
2011, 133, 4240-4242.
In summary, we have developed a practical strategy for the
highly selective convertion of simple amides to amines and
alcohols. This reaction features high reaction activity (TONs up to
19,600), wide substrate scope, mild reaction conditions, and
simple operation. It can be served as an outstanding and general
method to remove amide protecting group and should have broad
interest for organic communities. Studies on the substrate scope
of this system, catalyst variations, and the mechanism are
undergoing in our laboratory.
[8]
E. Balaraman, B. Gnanaprakasam, L. J. W. Shimon, D. Milstein, J. Am.
Chem. Soc. 2010, 132, 16756-16758.
[9]
a) J. M. John, S. H. Bergens, Angew. Chem. 2011, 123, 10561; Angew.
Chem., Int. Ed. 2011, 50, 10377-10380; b) J. M. John, R. Loorthuraja, E.
Antoniuk, S. H. Bergens, Catal. Sci. Technol. 2015, 5, 1181-1186.
[10] J. R. Cabrero-Antonino, E. Alberico, H.-J. Drexler, W. Baumann, K. Junge,
H. Junge and M. Beller, ACS Catal., 2016, 6, 47-54.
[11] a) J. A. Garg, S. Chakraborty, Y. Ben-David and D. Milstein, Chem.
Commun. 2016, 52, 5285-5288; b) F. Schneck, M. Assmann, M. Balmer,
K. Harms and R. Langer, Organometallics, 2016, 35, 1931-1943.
[12] X. Tan, Y. Wang, Y. Liu, F. Wang, L. Shi, K.-H. Lee, Z. Lin, H. Lv, X.
Zhang, Org. Lett. 2015, 17, 454-457.
Experimental Section
In an argon-filled glove box, a 5 mL vial equipped with a magnetic stirring
bar, was added the required amount of complex D (0.023 mg/mL in THF),
substrate (10 mmol) and base successively. Additional THF was added to
bring the total reaction volume to 4 mL. The vials were subsequently
transferred into an autoclave which was charged with hydrogen (50 bar).
[13] a) K. Abdur-Rashid, S. E. Clapham, A. Hadzovic, J. N. Harvey, A. J.
Lough, R. H. Morris, J. Am. Chem. Soc. 2002, 124, 15104; b) C. A.
Sandoval, T. Ohkuma, K. Muniz, R. J. Noyori, Am. Chem. Soc. 2003,
̃
125, 13490; c) R. Abbel, K. Abdur-Rashid, M. Faatz, A. Hadzovic, A. J.
Lough, R. H. Morris, J. Am. Chem. Soc. 2005, 127, 1870; (d) S.
Takebayashi, S. H. Bergens, Organometallics 2009, 28, 2349; (e) J. M.
John, S. Takebayashi, N. Dabral, M. Miskolzie, S. H. Bergens, J. Am.
Chem. Soc. 2013, 135, 8578; (f) P. A Dub, N. J. Henson, R. L. Martin, J.
C. Gordon, J. Am. Chem. Soc. 2014, 136, 3505.
o
The reaction was then stirred at 100 C for 20 h. The hydrogen gas was
released slowly and carefully. The solution was passed through a short
column of silica gel (eluent: EtOAc) to remove the metal complex. The
yields of compounds 1 and 3 were then determined by GC analysis.
[14] R. H. Crabtree, Chem Rev 2014.
Keywords: Amides • Hydrogenation • Chemoselectivity •
Amines • Alcohols
This article is protected by copyright. All rights reserved.

 Shi, Liyang
Shi, Liyang
 Tan, Xuefeng
Tan, Xuefeng
 Long, Jiao
Long, Jiao
 Xiong, Xiong
Xiong, Xiong
 Yang, Song
Yang, Song
 Xue, Peng
Xue, Peng
 Lv, Hui
Lv, Hui
 Zhang, Xumu
Zhang, Xumu