M. Soleiman-Beigi, F. Mohammadi
controlled by thin-layer chromatography. The solvent was
evaporated under vacuum. Then, crude reaction mixture
was purified by column chromatography (petroleum ether/
ethyl acetate 1:1) to give the secondary amines in good
yields.
transformations, vol 40a: Amines and Ammonium Salts. Georg
Thieme Verlag
4. McKetta JJ Jr (1993) Chemical Processing Handbook. CRC
Press, Florida
15. Castillo J-C, Orrego-Hern a´ ndez J, Portilla J (2016) Eur J Org
1
Chem 22:3824
6. Demir S, Damarhan Y, Ozdemir I (2015) J Mol Liq 204:210
7. Watson AJA, Maxwell AC, Williams JMJ (2011) J Org Chem
¨
1
1
Typical experimental procedure for tertiary amines
synthesis
7
6:2328
18. Nayal OS, Bhatt V, Sharma S, Kumar N (2015) J Org Chem
0:5912
8
1
9. Yagafarov NZ, Kolesnikov PN, Usanov DL, Novikov VV,
Nelyubina YV, Chusov D (2016) Chem Commun 52:1397
0. Liu S, Chen R, Deng GJ (2011) Chem Lett 40:489
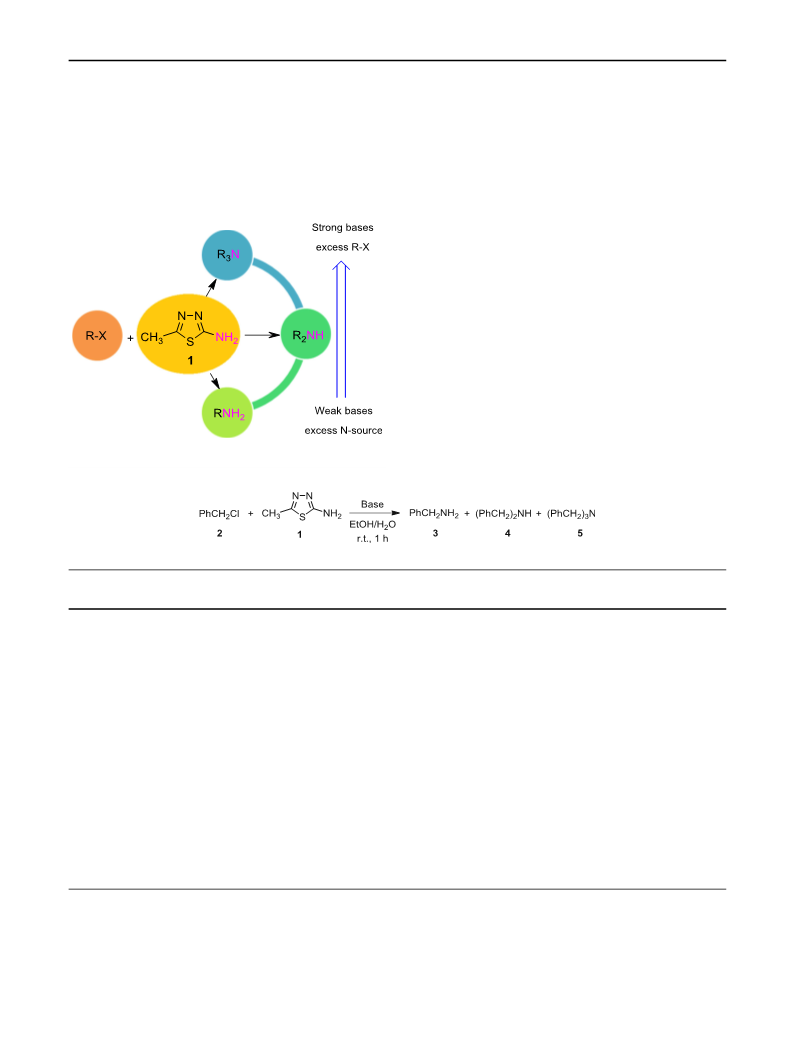
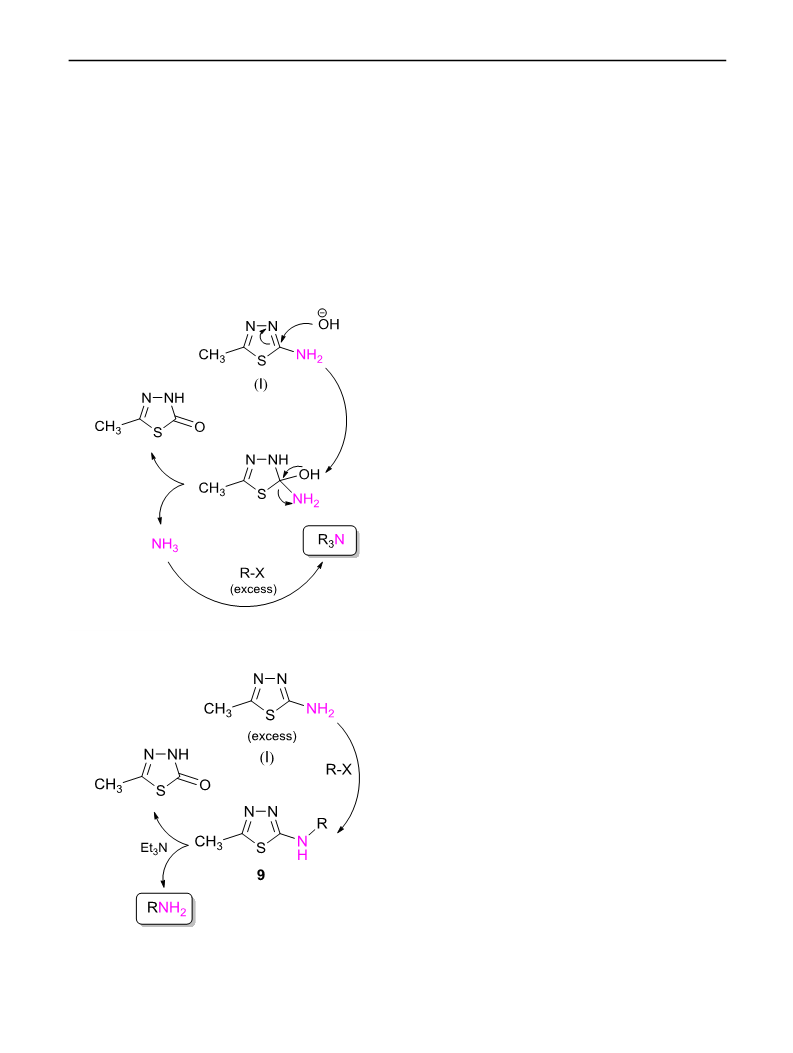
A mixture of alkyl halide (1.0 mmol) and 5-methyl-1,3,4-
thiadiazole-2-amine (1) (0.3 mmol) were added to a flask
3
containing 3 mmol KOH and 2 cm EtOH/H O (10:1). The
2
2
21. Artamkina GA, Sergeev AG, Stern MM, Beletskaya IP (2006)
Synlett 2006:235
2
reaction continued at room temperature under atmospheric
conditions until 1 h. The reaction progress was controlled
by thin-layer chromatography. The solvent was evaporated
under vacuum. Then, crude reaction mixture was purified
by column chromatography (petroleum ether/ethyl acetate
2. Younis FM, Krieck S, G o¨ rls H, Westerhausen M (2016) Dalton
Trans 45:6241
3. He L, Qian Y, Ding RS, Liu YM, He HY, Fan KN, Cao Y (2012)
Chemsuschem 5:621
2
24. Boche G, Mayer N, Bernheim M, Wagner K (1978) Angew Chem
lnt Ed 17:678
2
2
8
:1) to give the tertiary amines in good yields.
5. Berman AM, Johnson JS (2006) J Org Chem 71:219
6. Nyffeler PT, Liang CH, Koeller KM, Wong CH (2002) J Am
Chem Soc 124:10773
Acknowledgements We acknowledge the financial support of Ilam
University Research Council.
2
2
7. Chandrasekhar S, Prakash SY, Rao CL (2006) J Org Chem
7
8. Watson ID, Yu L, Yudin AK (2006) Acc Chem Rev 39:194
1:2196
References
29. Beak P, Selling GW (1989) J Org Chem 54:5574
3
0. Soleiman-Beigi M, Mohammadi F (2012) Tetrahedron Lett
3:7028
1. Soleiman-Beigi M, Mohammadi F (2015) Synlett 26:911
5
1
2
3
4
. Lawrence SA (ed) (2004) Amines: synthesis, properties and
application. Cambridge University Press, Cambridge
. Wittcoff HA, Reuben BG, Plotkin JS (2004) Industrial organic
chemicals, 2nd edn. Wiley, New York
. Hartwig JF (2002) Handbook of organopalladium chemistry for
organic synthesis. Wiley, New York, p 1051
3
3
2. Soleiman-Beigi M, Hemmati M (2013) Appl Organometal Chem
7:734
2
3
3
3. Soleiman-Beigi M, Arzehgar Z (2015) Sulfur Chem 36:395
4. B a¨ hn S, Imm S, Neubert L, Zhang M, Neumann H, Beller M
(
2011) Chem Eur J 17:4705
5. Szostak M, Sautier B, Spain M, Procter DJ (2014) Org Lett
6:1092
6. Miriyala B, Bhattacharyya S, Williamson JS (2004) Tetrahedron
0:1463
. Brown BR (1994) The organic chemistry of aliphatic nitrogen
compounds. Oxford University Press, New York
3
3
1
5
6
7
. Salvatore RN, Yoon CH, Jung KW (2001) Tetrahedron 57:7785
. Hoffmann AW (1850) Philos Trans 140:93
. Bahn S, Imm S, Neubert L, Zhang M, Neumann H, Beller M
6
3
3
7. Cui X, Dai X, Deng Y, Shi F (2013) Chem Eur J 19:3665
8. He J, Kim JW, Yamaguchi K, Mizuno N (2009) Angew Chem
(2011) ChemCatChem 3:1853
8
9
. Yang Q, Wang Q, Yu Z (2015) Chem Soc Rev 44:2305
. Hanada S, Ishida T, Motoyama Y, Nagashima H (2007) J Org
Chem 72:7551
1
21:10072
9. Koschabek R, Gleiter R, Rominger F (2006) Eur J Inorg Chem
006:609
0. Sachinvala N, Winsor DL, Maskos K, Grimm C, Hamed O, Vigo
TL, Bertoniere NR (2000) J Org Chem 65:9234
1. Peeters A, Claes L, Geukens I, Stassen I, De Vos D (2014) Appl
Catal A Gen 469:191
3
4
4
2
1
0. Das S, Wendt B, M o¨ ller K, Junge K, Beller M (2012) Angew
Chem 124:1694
1
1
1. Sorribes I, Jung K, Beller M (2014) J Am Chem Soc 136:14314
2. Katz DLV (1959) Handbook of natural gas engineering.
McGraw-Hill, New York
1
3. Azov V, Beller M, Butensch o¨ n H, B o¨ rner A, Couty FG (2014)
Science of Synthesis: Houben-Weyl methods of molecular
1
23

 Soleiman-Beigi, Mohammad
Soleiman-Beigi, Mohammad
 Mohammadi, Fariba
Mohammadi, Fariba