Mar-Apr 2007
Deoxygenation of Pyridine N-Oxides With Dimethylthiocarbamoyl Chloride
489
[3] A. R. Katritzky, Quart. Rev. 10, 395 (1956).
[4] S. Ochai, J. Org. Chem., 18, 534 (1953).
[5] A. Albini and S. Pietra, Heterocyclic N-Oxides; CRC: Boca
Raton, 1991; pp 120-134;
[6] A. R. Katritzky, J. M. Lagowski, Chemistry of the
Heterocyclic N-Oxides, Academic Press, London and New York, 1971,
pp 166-226.
[7] Some recent deoxygenation methods are mentioned in ref
[12] below.
[8] Y. Urushibara, S. Nishimura and H. Vehara, Bull. Chem. Soc.
Japan, 28, 446 (1955).
[9] E. Hayashi, H. Yamanaka and K. Shimizu, Chem. Pharm.
Bull., 6, 323 (1958).
[10] R. Balicki, Synthesis, 645 (1989).
[11] R. Balicki and L. Kaczmarek, Gazz. Chim. Ital., 124, 385
(1994).
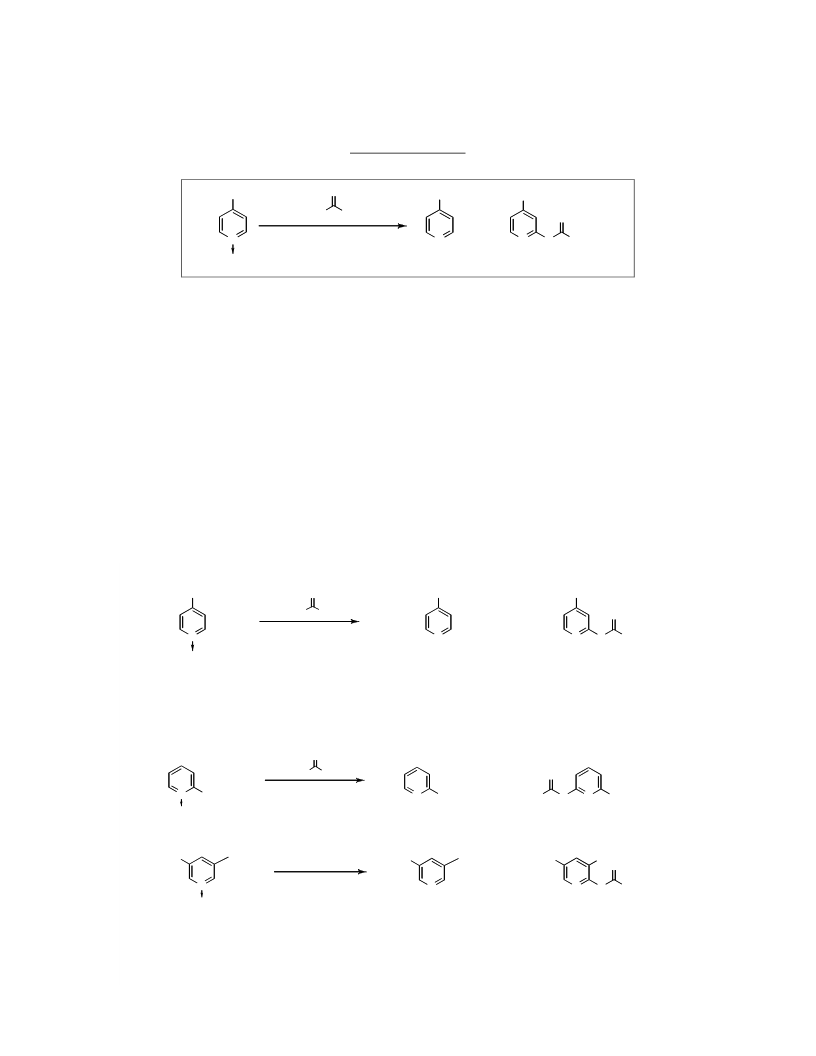
Addition of 2 equivalents of iodide ion to the reaction
mixture leads to the production of iodine but does not
increase the yields of pyridines nor does it accelerate the
rate of disappearance of pyridine N-oxides. [28] One mole
of DMTCC per mole of pyridine oxide is necessary, and
dimethylcarbamyl chloride is ineffective at causing
deoxygenation of 1, with or without iodide ion. 3,5-
Lutidine N-oxide 19 and 2,6-lutidine N-oxide 39 are
reduced at comparable rates and in comparable yield,
suggesting that species analogous to 45 (whose formation
in the case of 39 would be sterically disfavored) are not
involved in the deoxygenation process [19]. Finally we
note that small amounts of tetramethyl thiourea are
produced in all reductions
[12] S. Chandrasekhar, C. R. Reddy, R. J. Rao and J. M. Rao,
Synlett, 349 (2002).
[13] E. V. Brown, J. Am. Chem. Soc., 79, 3565 (1957).
[14] T. R. Emerson and C. W. Rees, J. Chem. Soc., 2319 (1964).
[15] W. C. Ross, J. Chem. Soc. (C),1816 (1966).
EXPERIMENTAL
[16] Y. Aoyagi, T. Abe and A. Ohta, Synthesis, 891 (1997).
[17] R. Balicki, M. Cybulski and G. Maciejewski, Synth.
Commun., 33, 4137 (2003).
[18] The N-oxides 4, 31, 35, and 39 are commercially available.
Other N-oxides, 1, 7, 10, 13, 16, 19, 22, 25, 28 and 42 were prepared by
treating the corresponding pyridines with 30% aqueous hydrogen
peroxide in boiling acetic acid for 5 hours [1] and their properties were
compared with the reported data.
[19] A. A. Ponaras and Ö. Zaim, In The Encyclopedia of Reagents
for Organic Synthesis, John Wiley; New York: 1993.
[20] All yields given here refer to isolated yields, sometimes by
distillation, more often by column chromatography.
[21] All compounds were characterized by IR, NMR and MS.
[22] Authentic samples of 3 and 6 were prepared via Newman-
Kwart reactions [23,24] on 4-t-butyl-2-pyridone and 4-phenyl-2-
pyridone, prepared by heating 1 and 4 with trifluoroacetic anhydride at
100 °C in a sealed tube.
[23] M. S. Newman and H. A. Karnes, J. Org. Chem., 31, 3980
(1966).
[24] H. Kwart and E. R. Evans, J. Org. Chem., 31, 410 (1966).
[25] Small amounts (<5%) of substances presumed to be the 6-
dimethylcarbamoylthio isomers are also present.
[26] We have prepared crystalline N-(dimethylthiocarbamoyloxy)
pyridinium chlorides from 1, 4, 19 and 39 by mixing the pyridine N-
oxides and DMTCC at room temperature in methylene chloride and then
concentrating the solvent. These salts go on to products when heated in
acetonitrile. The salt from pyridine N-oxide and DMTCC has been
prepared and characterized but, apparently, has not been subjected to any
further transformation: A. N. Pudovic, V. Y. Kovtun, V. K. Khairullin
and M. A. Vasyanina, Zh. Obshch Khim., 62, 269 (1992).
[27] For a discussion of some analogous processes which occur
when pyridine N-oxides are treated with acylating agents such as acetic
General procedure. A 1.48-g (12 mmol) portion of DMTCC
is added to a magnetically-stirred solution of 10 mmol pyridine
N-oxide in 20 mL of reagent-grade acetonitrile, and the solution
is heated at reflux for 4 to 14 hrs until GC shows no further
increase in the desired pyridine. Because of the high water-
solubility of the products, the following non-aqueous workup is
used. The mixture is cooled and treated with 12 mL of a 1 M
solution of HCl in ether. Solvent is removed on the rotary
evaporator and then at the vacuum pump and the residue is
triturated with two 20-mL portions of ether; the ether-soluble
phase, containing non-basic by-products such as bis(dimethyl-
carbamyl) disulfide, is set aside. The ether-insoluble phase is
dissolved in hot methylene chloride and treated with 2 mL (18
mmol) N,N-dimethylethaneamine and the solvent is removed on
the rotary evaporator and then at the vacuum pump. The residue
is triturated several times with 20-mL portions of ether and the
ether-insoluble solid (N,N-dimethylethaneamine hydrochloride
salt) is discarded. The ether-soluble phase is concentrated and
distilled or chromatographed on 100 g of silica gel (Davisil
grade 643, 200-425 mesh) packed in hexane/ethyl acetate.
This procedure was altered slightly for acid-sensitive
compounds by not treating product mixture with acid solution.
After removal of solvent on the rotary evaporator and then at the
vacuum pump the residue is dissolved in the minimum amount
of hot methylene chloride and passed through a short column
containing 10 g of basic alumina (CAMAG 5016-A-1, 150
mesh) eluting with hexane/methylene chloride. The eluant is
concentrated and the residue is distilled or chromatographed as
above.
anhydride, see: [a] V. J. Traynelis,
Mechanisms of Molecular
Migrations; B. S. Thyagaragan, Ed.; 1969; Vol. 2, p 1; [b] S. Tamagaki,
S. Kozuka and S. Oae, Tetrahedron Lett., 4765 (1968) and references
cited therein.
Acknowledgements. We thank the NSF and the ACS-PRF
for their financial support.
[28] Our initial work, attempting to exploit a similarity between
diosphenols and azaarene N-oxides (they are both subject to nucleophilic
addition alpha to the oxy group), employed a combination of DMTCC
and iodide ion (see ref. 9). It was later found that iodide ion is
unnecessary. The deoxygenation of diosphenols with dimethylthio-
carbamoyl chloride requires 2-equivalents of iodide ion: A. A. Ponaras,;
Ö. Zaim,; Y. Pazo and L. Ohannesian, J. Org. Chem., 53, 1110 (1988)
[29] We had previously suggested such intermediates (ref [19]).
REFERENCES AND NOTES
[1] A. Albini and S. Pietra, Heterocyclic N-Oxides, CRC: Boca
Raton, 1991; p 120.
[2] D. E. Young, Heterocyclic Chemistry, Longman, London,
1975, pp 72-80.

 Ponaras, Anthony A.
Ponaras, Anthony A.
 Zaim, Oemer
Zaim, Oemer