Chemistry - A European Journal
10.1002/chem.201704389
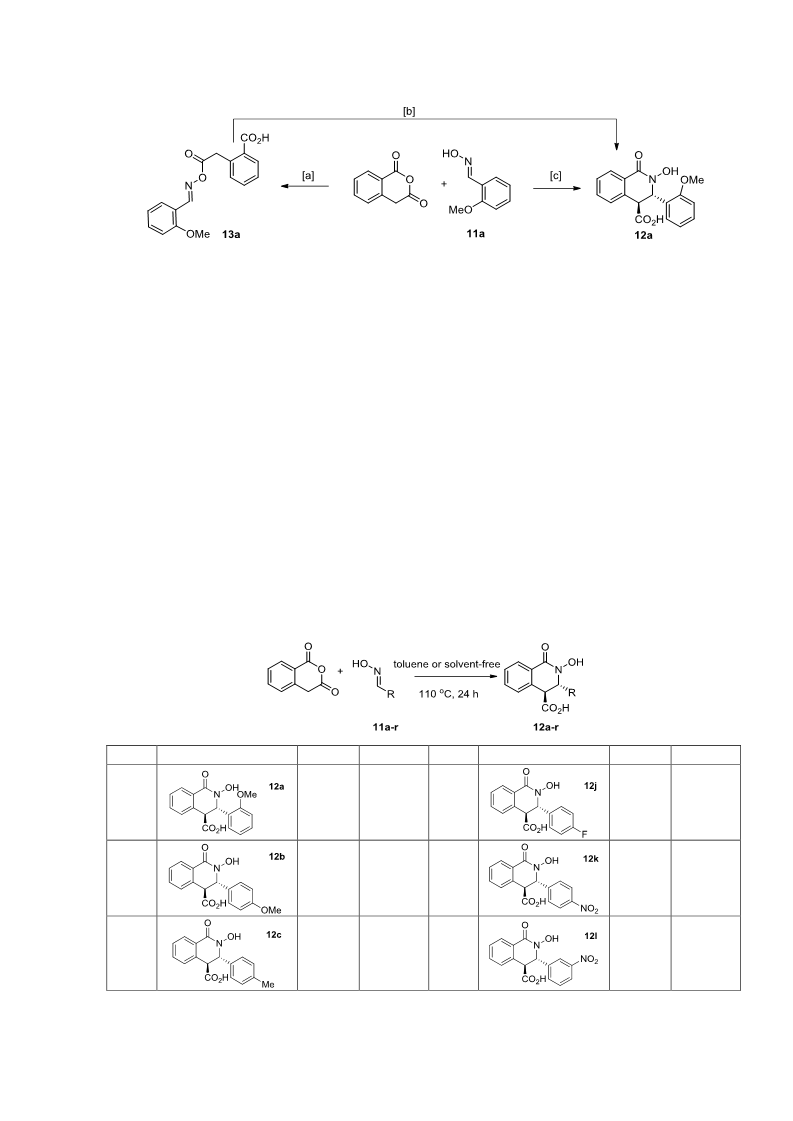
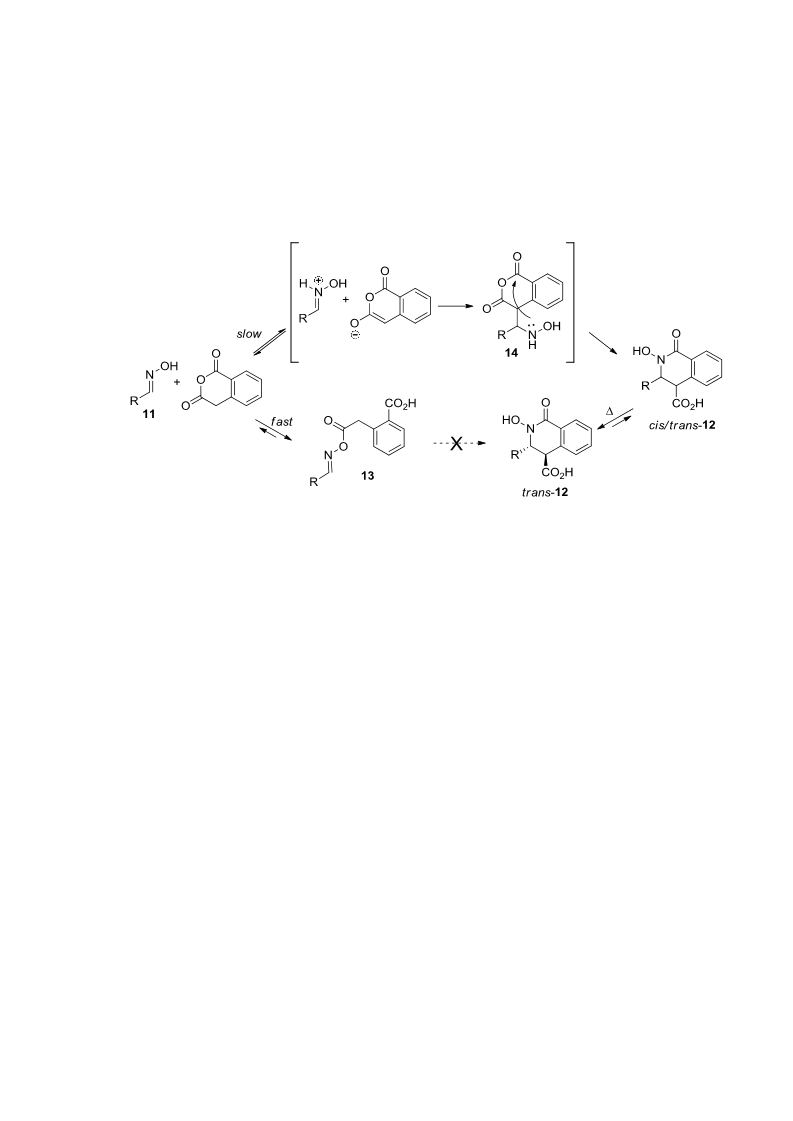
A mixture of homophthalic anhydride and corresponding oxime 11a-i, 16 or 19 (1.0 equiv.) was
suspended in dry toluene (2 mL/mmol) in a screw-cap vial and was placed in a pre-heated oil
o
bath at 110 C. After 24 h the reaction mixture was cooled to room temperature. Compounds
1
2a,b,d,e and 20 precipitated from the reaction mixture and were purified by filtration and
washing with small amount of cold diethyl ether. Compounds 12c,f-i and 17 were purified by
another procedure: the reaction mixture was concentrated, redissolved in DCM (5 mL/mmol) and
extracted with saturated aq. NaHCO (10 mL/mmol). The aqueous layer was separated and
3
washed with DCM (5 mL/mmol). The pH of aqueous phase was then adjusted to 1 with
concentrated aq. HCl at 0 °C. The formed precipitate was collected, washed with small amount
of water and dried in air to afford pure products.
General procedure 2. Neat protocol. Synthesis of compounds 12j-r.
A mixture of homophthalic anhydride and corresponding oxime 11j-r (1.0 equiv.) was placed in
a screw-cap vial and was thoroughly ground with a spatula. The vial was placed in a pre-heated
o
oil bath at 110 C. After 24 h the reaction mixture was cooled to room temperature. For
compounds 12l-r: a small amount of diethyl ether (5 mL/mmol) was added and the reaction
mixture was sonicated in ultrasonic bath for 15 minutes. The resulting suspension was cooled to
–
20 °C, filtered and dried in air to give pure title compounds. Compounds 12j,k were purified by
another procedure: the reaction mixture was dissolved in DCM (5 mL/mmol) under sonication
and was extracted with saturated aq. NaHCO (10 mL/mmol). The aqueous layer was separated
3
and washed with DCM (5 mL/mmol). The pH of aqueous phase was then adjusted to 1 with
concentrated aq. HCl at 0 °C. The formed precipitate was collected, washed with small amount
of water and dried in air to afford pure title compounds 12.
trans-2-Hydroxy-3-(4-methoxyphenyl)-1-oxo-1,2,3,4-tetrahydroisoquinoline-4-carboxylic
acid (12b) was prepared according to general procedure 1 from HPA (162 mg, 1 mmol) and 4-
methoxybenzaldehyde oxime 11b (151 mg, 1 mmol). Yield 275 mg, 88%. White solid, m.p.
1
2
1
2
1
24−226 °C. H NMR (400 MHz, DMSO-d6) δ 13.05 (s, 1H), 10.18 (s, 1H), 7.94 (d, J = 7.4 Hz,
H), 7.56 – 7.35 (m, 2H), 7.29 (d, J = 7.1 Hz, 1H), 7.04 (d, J = 8.6 Hz, 2H), 6.82 (d, J = 8.6 Hz,
1
3
H), 5.42 (s, 1H), 4.23 (s, 1H), 3.68 (s, 3H). C NMR (101 MHz, DMSO-d6) δ 172.4, 160.7,
59.1, 133.5, 132.2, 131.0, 130.2, 129.1, 128.3, 127.7, 127.0, 114.3, 65.1, 55.5, 52.0. UV/Vis
EtOH-H O): λ (logε) = 255 nm (3.89). HRMS (ESI), m/z calcd for C H NO [M+H]
max
+
(
2
17 16
5
3
14.1023, found 314.1012.
trans-3-(4-Trifluoromethylphenyl)-2-hydroxy-1-oxo-1,2,3,4-tetrahydroisoquinoline-4-
carboxylic acid (12n) was prepared according to general procedure 2 from HPA (162 mg, 1
mmol) and 4-trifluoromethylbenzaldehyde oxime 11n (189 mg, 1 mmol). Yield 225 mg, 64%.
1
White solid, m.p. 220−222 °C. H NMR (400 MHz, DMSO-d ) δ 13.16 (s, 1H), 10.37 (s, 1H),
6
7
2
.96 (dd, J = 7.5, 1.6 Hz, 1H), 7.67 (d, J = 8.3 Hz, 2H), 7.50 – 7.40 (m, 2H), 7.38 (d, J = 8.2 Hz,
H), 7.29 (dd, J = 7.1, 1.6 Hz, 1H), 5.60 (d, J = 1.8 Hz, 1H), 4.36 (d, J = 1.8 Hz, 1H). C NMR
1
3
(
101 MHz, DMSO-d6) δ 172.0, 160.9, 143.9 (q, J = 1.1 Hz), 133.1, 132.4, 130.2, 128.8, 128.7
СF
(
5
q, J = 31.6 Hz), 128.5, 127.5, 127.1, 125.9 (q, JСF = 3.5 Hz), 127.2 (q, JСF = 270.0 Hz), 65.3,
max
1.5. UV/Vis (EtOH-H O): λ (logε) = 255 nm (3.60). HRMS (ESI), m/z calcd for
2
+
C H F NO [M+H] 352.0791, found 352.0806.
17
13
3
4
1
3
This article is protected by copyright. All rights reserved.

 Bakulina, Olga
Bakulina, Olga
 Bannykh, Anton
Bannykh, Anton
 Dar'in, Dmitry
Dar'in, Dmitry
 Krasavin, Mikhail
Krasavin, Mikhail