Journal of the American Chemical Society
Communication
structure and external substrate oxidizing behavior of a Ni(II)−
mCPBA complex.
Our strategy for the isolation of a Ni(II)−mCPBA complex
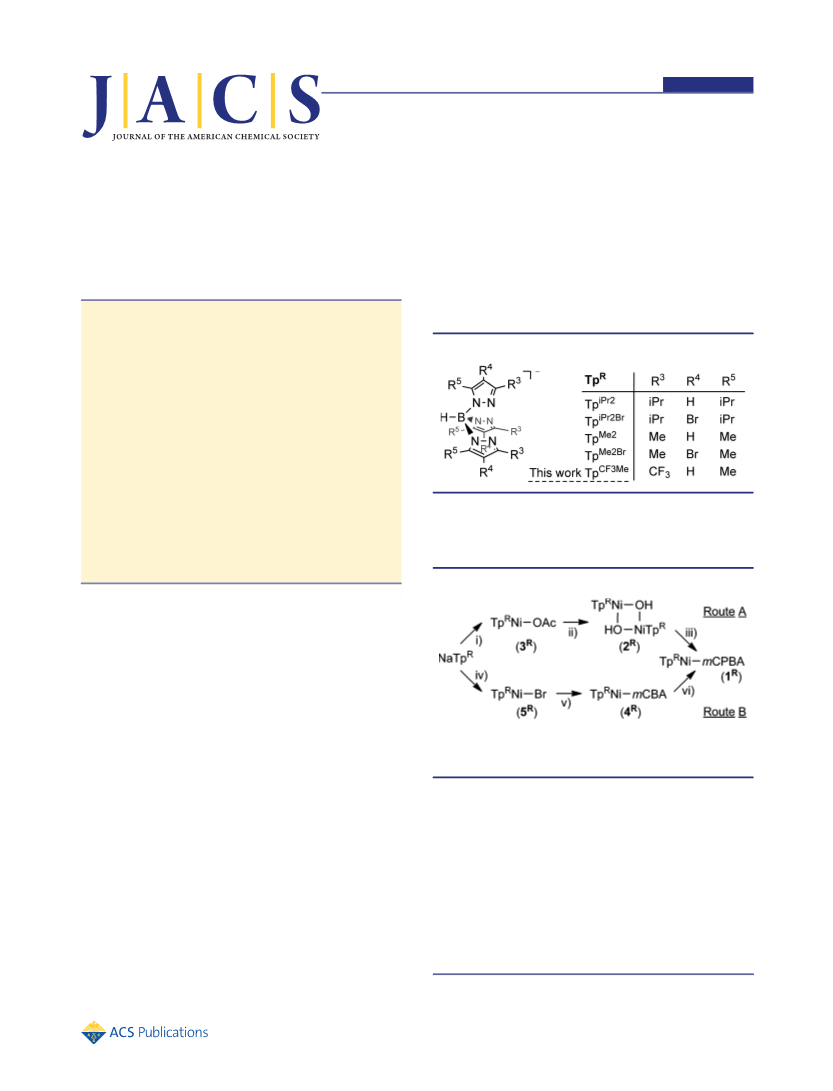
involved controlling the properties of the Ni-supporting TpR
ligand. As mentioned above, the incorporation of electron-
withdrawing groups (EWGs) on the pyrazolyl rings of TpR
enhances the stability of 1. In addition, steric hindrance and
chemical stability (i.e., resistance to oxidation) of the
substituent groups on TpR are critical. Bulky alkyl groups
(e.g., isopropyl groups) at the R3 position of the pyrazolyl rings
wrap the O−O moiety of the metal-bound acylperoxide,
stabilizing 1 and hindering the reaction with external substrates.
Moreover, C−H bonds of the R3 substituents are oxidized by a
proximal oxidant (i.e., metal-bound acylperoxide or further
activated species generated via O−O breaking) through
intramolecular reaction. Therefore, in this study hydrotris(3-
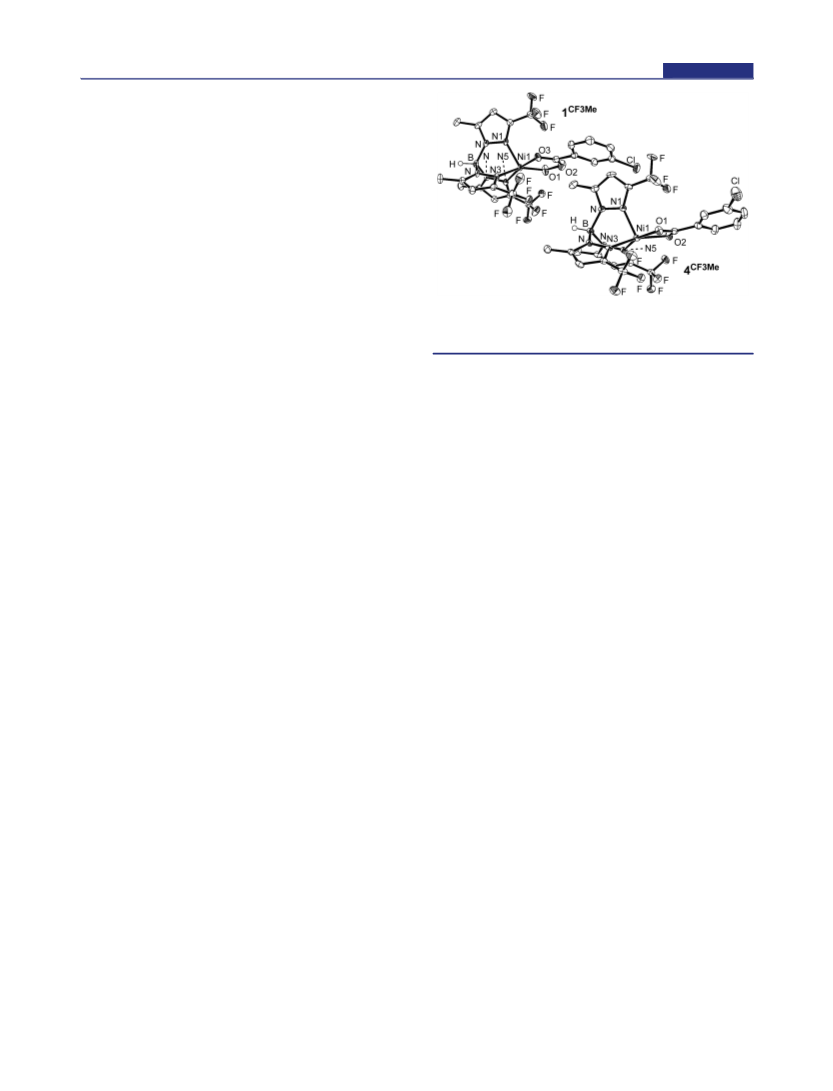
Figure 1. ORTEP diagrams of the complexes 1CF Me and 4CF Me with
50% thermal ellipsoids. H atoms on the carbon backbones have been
omitted for clarity.
3
3
trifluoromethyl-5-methylpyrazolyl)borate (TpCF Me) was em-
3
ployed as the Ni-supporting ligand because the proximal
trifluoromethyl groups exhibit an electron-withdrawing nature,
high oxidation resistance, and moderate steric hindrance. The
O3C16 = 1.914(2), 2.040(2), and 1.223(3) Å, respectively].
versatility of TpCF Me for the stabilization of metal−dioxygen
3
The Ni−N(Tp) lengths in 1CF Me [Ni1−N1, Ni1−N3, and
3
species was revealed by the Cu2(μ-η2:η2-O2) complex reported
by Gorun and co-workers.7
Ni1−N5 = 2.037(2), 2.041(2), and 2.058(2) Å, respectively]
are comparable to those in 4CF Me, where the Ni center is
3
supported by the tridentate TpCF Me ligand and the κ2-binding
3
Our first attempt to synthesize an acylperoxo complex with
TpCF Me (1CF Me) in a manner similar to that used to form the
reported 1R (i.e., dehydrative condensation between the
Ni(II)−hydroxo species and HmCPBA; route A in Scheme
1)6 failed because of the insolubility of the putative Ni(II)−
3
3
mCBA ligand. The geometry of the nickel center of 1CF Me is
3
slightly distorted from square-pyramidal (τ = 0) toward
trigonal-bipyramidal (τ = 1), as indicated by its τ value of
0.26. The Ni−mCPBA site is fully surrounded by CF3 groups.
These structural characteristics are consistent with the
hydroxo complex 2CF Me formed by the reaction of the Ni(II)−
3
acetato complex [NiII(TpCF Me)(OAc)] (3CF Me) with aqueous
NaOH. Therefore, we selected an m-chlorobenzoate (mCBA)
following observed spectroscopic properties:8 Paramagnetically
3
3
1
shifted H NMR signals of 1CF Me assignable to the protons of
3
complex, [NiII(TpCF Me)(mCBA)] (4CF Me), as a precursor to
3
3
the TpCF Me and mCPBA ligands support the high-spin
3
1CF Me because exchange of the mCBA ligand with mCPBA on
3
configuration (S = 1) of the distorted five-coordinate Ni(II)
center (Figures S5 and S6). Coordination of the carbonyl
moiety of the acylperoxo ligand leads to a red shift of the νCO
vibration to 1644 cm−1 in the IR spectrum, as found in the
previously reported TpR derivatives (i.e., for 1Me2 in CH2Cl2
solution at −40 °C, νCO = 1646 cm−1) (Figures S7−S9).4,6,11
The oxidation reactivity of the isolated Ni(II)−acylperoxo
the Ni(II) center might occur during the turnover process in
catalytic alkane oxidation by Ni/mCPBA systems (route B in
5
Scheme 1). 4CF Me was prepared from bromido complex 5CF Me
by ligand exchange with KmCBA.8 As we expected, the reaction
3
3
of 4CF Me with a small excess (∼2 equiv) of HmCPBA in
3
CH2Cl2 at −40 °C allowed a perfect conversion to 1CF Me
.
3
complex 1CF Me for external substrates in aprotic solvents
3
Monitoring of the UV−vis spectral changes revealed an
increase in absorbance at 375 nm, which was attributed to
(CF3C6H5, toluene, benzene) was studied by kinetic analysis
1CF Me, as the 408 nm band of 4CF Me decreased with an
isosbestic point at 395 nm. Surprisingly, this titration
experiment was reproducible at room temperature, indicating
3
3
using UV−vis spectroscopy and product analysis using GC
8
(Scheme 2). Without any substrate, the complex 1CF Me was
3
thermally robust even at 70 °C in benzene (first-order rate
the high thermal stability of the formed 1CF Me [Figure S2 in the
constant = 1.63 × 10−5 s−1), while 1Me2 decomposed below
3
−20 °C.12 This thermal stability of 1CF Me is a result of the
3
Supporting Information (SI)].
After the volatiles were removed from the reaction mixture of
steric protection around the Ni center and the electron-
HmCPBA (2.7 equiv) and 4CF Me in CH2Cl2, the dissociated
withdrawing nature of the CF3 groups in the TpCF Me ligand.
3
3
However, 1CF Me behaved as an O-atom transfer reagent toward
3
HmCBA and the remaining excess HmCPBA were washed out
with H2O and EtOH. The agreement between the UV−vis
spectra of the redissolved isolated solid and the final species in
the above-mentioned titration in CH2Cl2 suggests that isolation
was achieved (Figure S4). Recrystallization of the resulting
green-blue solid from CH2Cl2/n-hexane yielded a single crystal
nucleophiles such as phosphine and sulfide under mild
conditions. A reaction solution containing 10 equiv of
thioanisole stoichiometrically formed MeS(O)Ph (96%)
and MeS(O)2Ph (1%), and the use of 10 equiv of PPh3
gave OPPh3 (103%), as determined by GC analyses (yields
of 1CF Me suitable for X-ray analysis (Figure 1 and Table S3 in
based on 1CF Me; Table S4). In fact, the decay of 1CF Me was
accelerated even at −40 °C in the presence of excess
thioanisole. The pseudo-first-order reaction rate constant
3
3
3
the SI).8,9 The oxidation state of the Ni center is +2, and 1CF Me
3
is a neutral complex, as evidenced by the absence of any
counterions in the crystal lattice. The Ni center in 1CF Me is
(kobs) for the decay of 1CF Me became saturated as the
3
3
supported by a donor set containing three N atoms and two O
atoms. The O−O bond length [1.443(3) Å] falls in the typical
range for peroxide O−O lengths (Figure S4).3,4a,10 The
coordination mode of mCPBA is κ2, as indicated by the bond
lengths of the percarboxylate moiety [Ni1−O1, Ni−O3, and
concentration of thioanisole increased, implying that the
reaction of 1CF Me and thioanisole proceeds through a
3
Michaelis−Menten-type mechanism (Scheme 2 and Figures
S10−S12).13 A Lineweaver−Burk-type plot (1/[thioanisole] vs
1/kobs) gave values of the association equilibrium constant Kon
6011
dx.doi.org/10.1021/ja4020277 | J. Am. Chem. Soc. 2013, 135, 6010−6013

 Nakazawa, Jun
Nakazawa, Jun
 Terada, Shota
Terada, Shota
 Yamada, Masaki
Yamada, Masaki
 Hikichi, Shiro
Hikichi, Shiro