5208
A. Pyo et al. / Tetrahedron Letters 54 (2013) 5207–5210
H
I
1 mol% catalyst a~h
O
+
Base A~L (1.5 equiv)
H
H
DMSO,80°C
(a)
$ % & ' ( ) *
+ , - . /
D
E
F
G
H
Scheme 1. Optimization of hydrodehalogenations by using PBCIS.
Table 1
Pd-catalyzed hydrodehalogenationa
I
J
H
I
cat. Pd
O
+
H
Cs2CO3
K
H
DMSO, 80°C
(b)
100
80
60
40
20
0
Entry
Pd (mol %)
Mol %
Conversion (%)
Yieldb (%)
1
2
3
4
5
6
7
8
Pd2(dba)3
Pd(PPh3)4
PdCl2
Pd(OAc)2
PdCl2
Pd(OAc)2
Pd(OAc)2
Pd(OAc)2
1.0
1.0
1.0
1.0
100
100
100
100
75
100
100
35
93
96
99
99
73
99
95
32
0.1
0.01
0.005
0.001
0
12 24 36 48 60 72 84 96
Number of the sample
a
Reaction condtions: 1-iodonaphthalene (0.3 mmol), paraformaldehyde
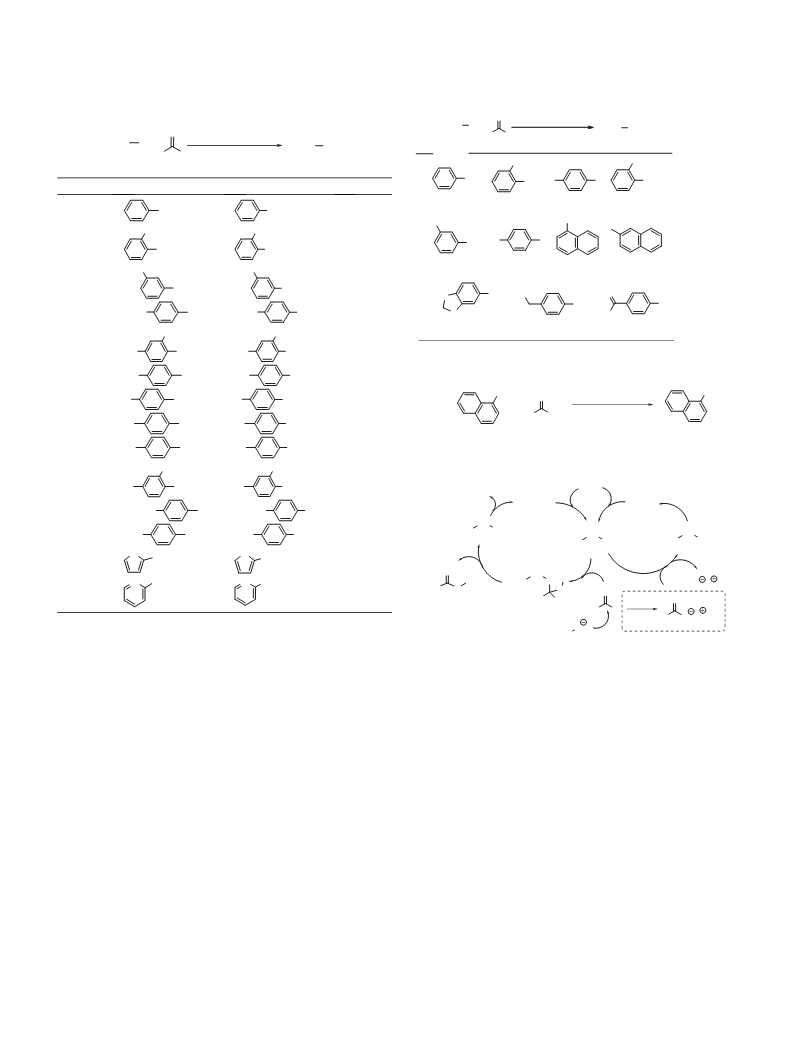
Figure 1. Screening of catalysts and bases for the hydrodehalogenation using PBCIS.
Catalyst: a–h, Base: A–L. (a) Picture of the PBCIS results of the reactions. (b) The
extent of conversion of 1-iodonaphthalene was determined by equation [conver-
sion (PBCIS)% = (À1.0506) Â (grayscale intensity À 243.9713)] which is obtained
from linear plot of standard samples.
(0.3 mmol), and Cs2CO3 (0.45 mmol) were reacted in DMSO at 80 °C for 12 h.
b
Determined by gas chromatography with an internal standard.
To find suitable catalytic system, as shown in Table 1, 1-iodo-
naphthalene was chosen as a model substrate and reacted with
paraformaldehyde under 96 reaction conditions. The reaction of
iodonaphthalene and paraformaldehyde was carried out in the
presence of catalyst and base in DMSO at 80 °C for 6 h. As a cata-
lyst, PdCl2 (a), Pd(OAc)2 (b), NiCl2 (c), Ni(OAc)2 (d), CuCl (e), CuCl2
(f), FeCl3 (g), and CoBr2 (h) were employed. Five kinds of weak
inorganic bases [Cs2CO3 (A), K2CO3 (B), Na2CO3 (C), K3PO4 (D),
and K2HPO4 (E)], four kinds of organic bases [DBU (F), Et3N (G),
DBN (H), and TMEDA (I)], and three kinds of strong bases [NaH
(J), KOtBu (K), and NaOtBu (L)] were tested. First, we monitored
the conversion of iodonaphthalene by using PBCIS, which is sim-
pler and easier than using gold nanoparticles, and much faster than
gas chromatography. The reaction mixture was diluted and treated
with 0.2 N HCl, and this solution was spotted into the PBCIS. All the
reactions provide different strengths of color, as shown in Fig-
ure 1(a). The spots on the scanned PBCIS were converted to gray-
scale values using Adobe Photoshop. Then, they were converted
to the extent of conversion using the conversion equation shown
in Figure 1 (b).20
As shown in Figure 1, we found several things as followings: (1)
PBCIS is a very useful tool to find the optimized condition for the
hydrodeiodination. (2) PBCIS is a suitable assay for the transition
metal catalyst such as palladium, copper, nickel, iron, and cobalt.
(3) At seeing the darkness of spots, the extent of conversion was
obtained quickly, and the value of that was obtained from their
grayscale intensity. From the results of Figure 1, the reactions with
palladium showed higher conversion extents than other catalysts.
Among the palladium-catalyzed reactions, weak inorganic bases
such as Cs2CO3, K2CO3, and Et3N and strong bases such as NaH
and NaOtBu showed high conversion extents (sample numbers 1,
2, 7, 10, 12, 13, 14, 19, 22, and 24). Considering the mild reaction
conditions, PdCl2 and Pd(OAc)2 were chosen. In addition, Pd2(dba)3
and Pd(PPh3)4 were added as palladium sources due to their good
activities in the Pd-catalyzed hydrodehalogenation of aryl bro-
mide.13b They were tested for the hydrodehalogenation of iodo-
naphthalene in the presence of Cs2CO3.21 Their conversions and
product yields were monitored by gas chromatography with an
internal standard.
Pd2(dba)3 and Pd(PPh3)4 showed 100% conversion of 1-iodo-
naphthalene, however, the yields of products were 93% and 96%,
respectively (entries 1 and 2). Both PdCl2 and Pd(OAc)2 showed good
yields when 1.0 mol % of them was used (entries 3 and 4). These re-
sults were similar to those from PBCIS. When the amount of palla-
dium was decreased to 0.1 mol %, PdCl2 afforded a 73% yield of the
product (entry 5). The employment of 0.01 and 0.005 mol % of
Pd(OAc)2 produced naphthalene in 99% and 95% yields, respectively
(entries 6 and 7). However, the product yield was decreased to 32%
when the amount of Pd(OAc)2 was decreased to 0.001 mol % (entry
8). Based on these results, the optimized conditions of the hydrode-
halogenation of aryl iodides involve aryl iodides (1.0 equiv) and
paraformaldehyde (1.0 equiv) reacted with Pd(OAc)2 (0.01 mol %)
and Cs2CO3 (1.5 equiv) in DMSO at 80 °C for 12 h. In addition, when
the reaction was conducted in the presence of formaline instead of
paraformaldehyde, the desired hydrodehalogenated product was
formed. A variety of aryl iodides were reacted with paraformalde-
hyde under the optimized conditions to explore them further. The
results are summarized in Table 2.
Alkyl and alkoxy-substituted aryl iodides were converted into
the hydrodehalogenated arenes in good yields (entries 1–6). Halo-
substituted aryl iodides were transformed into the deiodinated
arenes in good yields (entries 7–9). The hydrodebromination did
not proceed in these reaction conditions (entry 9). Aryl iodides
bearing the amino or ester group produced the corresponding
product in good yields (entries 10 and 11). 4-Nitro iodobenzene
was converted to nitrobenzene without the reduction of the nitro
group (entry 12). Heteroaromatic iodides such as 2-iodothiophene
and 2-iodopyridine also produced the hydrodeiodinated arenes in
good yields (entries 13 and 14).
Next, we attempted to apply this reaction method to the aryl
bromides. However, the hydrodehalogenated product was formed
in low yield in the presence of Pd(OAc)2. To increase the yield of

 Pyo, Ayoung
Pyo, Ayoung
 Kim, Sudeok
Kim, Sudeok
 Kumar, Manian Rajesh
Kumar, Manian Rajesh
 Byeun, Aleum
Byeun, Aleum
 Eom, Min Sik
Eom, Min Sik
 Han, Min Su
Han, Min Su
 Lee, Sunwoo
Lee, Sunwoo