ChemComm
Communication
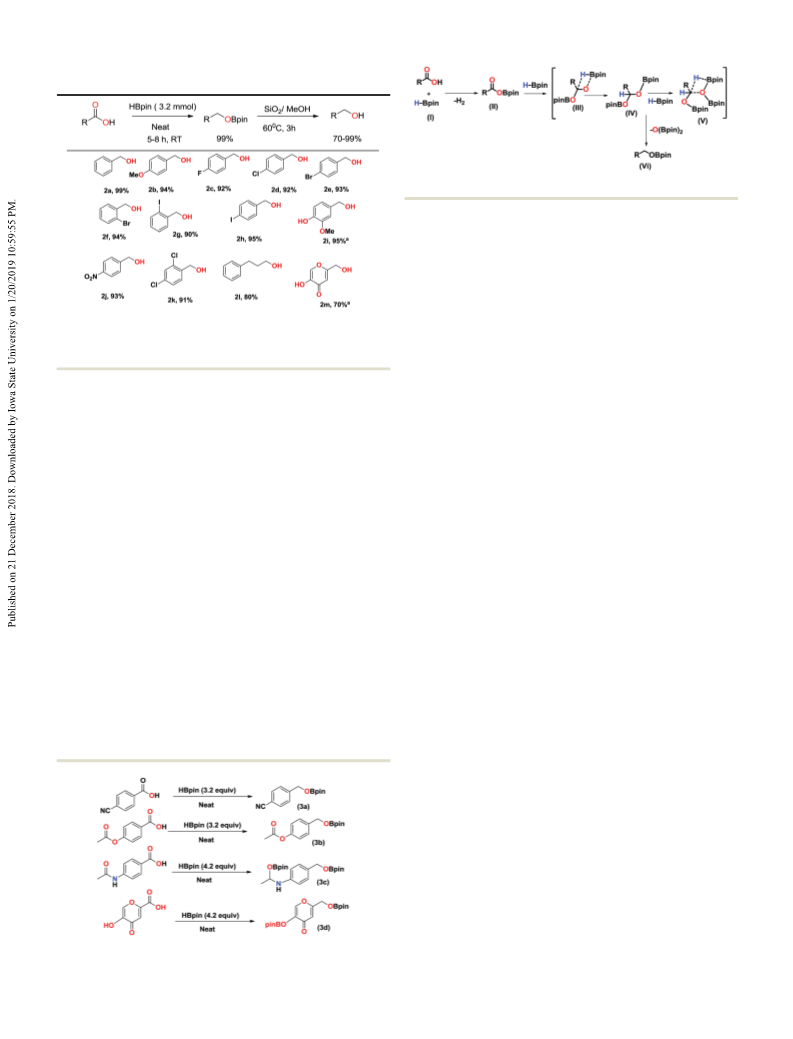
Table 3 Hydrolysis of boronate ester to achieve corresponding primary
alcohols
Scheme 2 Proposed mechanism for hydroboration of carboxylic acids.
hydroboration of carboxylic acid is depicted in Scheme 2. In the
first step, cross-dehydrocupling of the COOH group occurs with
HBpin, leading to the formation of intermediate species II with
the elimination of H2 gas. In the next step, the second molecule
of HBpin reacts with species II to form a four-membered cyclic
species III, which further rearranges itself to form IV. Species IV
Reaction conditions: carboxylic acids (0.25 mmol) and HBpin (0.80 mmol) further reacts with the third molecule of HBpin through another
were stirred together for 5–8 hours in room temperature; isolated yields and
four-membered cyclic intermediate to afford the desired boronate
a
products were purified by column chromatography. HBpin (1.05 mmol)
ester through the liberation of [O(Bpin)2] as a side product
was used.
which was confirmed by 1H, 11B NMR spectra of all the boronate
esters (1a–1z).
with dicarboxylic acids such as adipic acid and terephthalic
acid also occurred efficiently to yield boronate esters 1y and
1z, indicating the versatile substrate scope of this protocol
(Fig. FS74–79 in ESI†).
To summarise, we have developed a simple and facile
protocol for the deoxygenative hydroboration of carboxylic acids
to corresponding boronate esters under catalyst-free and solvent-
free conditions, which upon hydrolysis furnishes primary alcohols.
Further, we hydrolysed the selected boronate esters to their
This reaction proceeds with quantitative chemo selective yields,
alcohols using known methodology6 and in all cases, we obtained
exhibiting a broader substrate scope with both aliphatic and
near-quantitative yields (2a–2m) (Fig. FS80–104, ESI†). The results
are summarised in Table 3. Additionally, to examine the chemo-
aromatic carboxylic acids, in short reaction times.
This work is supported by the JICA FRIENDSHIP Project
selectivity of this methodology, we treated 4-cyano benzoic acid
under the Collaboration Kick-starter Programme (CKP). A. H.
and 4-acetoxy benzoic acid with HBpin and observed the for-
and J. B. thank CSIR and UGC India respectively for their PhD
mation of exclusive deoxygneative hydroboration with acid func-
fellowships. We thank Prof. Kazushi Mashima and Prof. Hayato
tionality after 8 h. No trace of a corresponding product from
Tsurugi, Osaka University, Japan, for their generous support.
hydroboration of the nitrile or ester moiety was found, indicating
We are also thankful to Dr S. R. Dey and Prof. Ch. Subrahmanyam,
chemoselective hydroboration of the acid group (Fig. FS106–111
IIT Hyderabad for their help.
in ESI†). Additionally, in the reaction with 4-acetamido benzoic
acid, both the acid and amido functionalities underwent hydro-
boration over dehydrocoupling of NH with HBpin in the substrate
Conflicts of interest
(Fig. FS112–114 in ESI†).13 However, 5-hydroxy-4-oxo-4H-pyran-
There are no conflicts to declare.
2-carboxylic acid underwent chemo-selective deoxygenative
hydroboration, yielding only the corresponding boronate ester
(Scheme 1, 3a–3d, Fig. FS115–118 in ESI†).
In light of this outcome and previously reported literature
reports13,17 the most plausible mechanism for the deoxygenative
Notes and references
1 (a) N. Miyaura and A. Suzuki, Chem. Rev., 1995, 95, 2457–2483;
(b) A. J. Lennox and G. C. Lloyd-Jones, Chem. Soc. Rev., 2014,
43, 412–443; (c) H. C. Brown, G. W. Kramer, A. B. Levy and
M. M. Midland, Organic Syntheses via Boranes, Wiley-Interscience,
New York, 1975, vol. 1; (d) A. Pelter, K. Smith and H. C. Brown,
Borane Reagents, Academic Press, London, 1988.
2 L. J. Gooßen, L. Rodriguez and K. Gooßen, Angew. Chem., Int. Ed.,
2008, 47, 3100–3120.
3 (a) J. Seyden-Penne, Reductions by the Alumino- and Borohydrides in
Organic Synthesis, Wiley, New York, 2nd edn, 1997; (b) P. N.
Rylander, Catalytic Hydrogenation in Organic Syntheses, Academic
Press, New York, 1979; (c) G. Kokotos and C. Noula, J. Org. Chem.,
1996, 61, 6994–6996; (d) M. Bagheri and M. Karimkoshteh, Iran.
J. Chem. Chem. Eng., 2017, 36, 37–43; (e) Y. Suseela and M. Periasamy,
Tetrahedron, 1992, 48, 371–376; ( f ) J. V. B. Kanth and M. Periasamy,
J. Org. Chem., 1991, 56, 5964–5965; (g) S. Narasimhan, S. Madhavan and
K. Ganeshwar Prasad, J. Org. Chem., 1995, 60, 5314–5315.
4 (a) T. Vom Stein, M. Meuresch, D. Limper, M. Schmitz, M. Holscher,
J. Coetzee, D. J. Cole-Hamilton, J. Klankermayer and W. Leitner,
Scheme 1 Chemoselective hydroboration of carboxylic acids (3a–3d).
This journal is ©The Royal Society of Chemistry 2019
Chem. Commun.

 Harinath, Adimulam
Harinath, Adimulam
 Bhattacharjee, Jayeeta
Bhattacharjee, Jayeeta
 Panda, Tarun K.
Panda, Tarun K.