Heterogeneous Permanganate Oxidations
J . Org. Chem., Vol. 62, No. 25, 1997 8771
Activity 1, 8.8 g) with stirring to give a permanganate/alumina
mixture. This mixture was dried in an uncovered Petri dish
overnight at room temperature before use.
involve two-electron transfers, is +5. The cleavage
reaction depicted in Scheme 3, however, involves an
overall transfer of four electrons, leaving manganese with
a formal oxidation state of +3. Although it is not possible
to verify these oxidation states experimentally, it is
obvious that a definite change in the appearance of the
oxidant occurs in every case. The heterogeneous oxidant,
a bright purple color initially, is a dull brown at the end
of the reaction, suggesting that it may be a form of
manganese dioxide, which is known to be the usual
product from homogeneous permanganate oxidations
under neutral conditions.26 Since manganate(V) is known
to be a very vigorous oxidant under neutral conditions,27
it is quite likely that the products depicted in Schemes
1, 2, and 5 would be rapidly reduced by the solvent to a
+4 oxidation state. Predicting the fate of the manganese-
(III) product produced from the cleavage reaction depicted
in Scheme 3 is more difficult. It could either be reduced
by the solvent to give manganese(II) or oxidized by excess
oxidant to manganese dioxide.28
KMn O4/Cu SO4‚5H2O: Equal weights of potassium per-
manganate and copper sulfate pentahydrate were ground
together in a mortar. The resulting fine, highly colored
product was then used as a heterogeneous oxidant in meth-
ylene chloride solutions.
Typ ica l Exp er im en ta l P r oced u r es. Th e Oxid a tion of
F lu or en e w ith KMn O4/Al2O3‚xH2O: Fluorene (0.54 g, 3.2
mmol), dissolved in 1,2-dichloroethane (120 mL), was placed
in a round-bottomed flask with a magnetic stirrer and the
permanganate/alumina mixture added in small portions over
a 10 min period. The mixture was stirred at room temperature
(about 20 °C) and the extent of reaction monitored by use of
TLC. After 118 h, analysis of the liquid phase by TLC
indicated complete conversion to 9-fluorenone. The product
was filtered through a fritted glass funnel and the residue
washed with portions of CH2Cl2 (3 × 50 mL). The solvent was
then removed by flash evaporation to give a product (0.59 g,
3.2 mmol, 100%) which produced only one TLC spot and melted
at 82.5-85 °C (lit.20 85 °C). The infrared and NMR spectra
were identical with those of an authentic sample of fluorenone.
Oxid a t ion of Dip h en ylm et h a n e w it h KMn O4/Al2O3‚
xH2O: Diphenylmethane (0.61 g, 3.6 mmol) was dissolved in
1,2-dichloroethane (120 mL) and placed in a round-bottomed
flask. KMnO4/Al2O3 reagent (11.5 g), prepared as described
above, was added in small portions over a period of 15 min.
The mixture was stirred at room temperature for 282 h. The
spent oxidant and solid support were removed by filtration
through a fritted glass funnel and the residue washed with
methylene chloride (3 × 50 mL). Evaporation of the solvent
gave impure product (0.62 g) which was purified by elution
chromatography to yield benzophenone (0.59 g, 3.3 mmol,
91%). The melting point of the 2,4-DNP derivative was 235.5-
237.5 °C (lit.29 239 °C). The infrared and NMR spectra were
identical with those of an authentic sample.
Con clu sion s
1. Aliphatic side chains are selectively oxidized at the
benzylic position by permanganate adsorbed on solid
supports (copper sulfate pentahydrate or moist alumina).
The products are alcohols if the benzylic carbon is
tertiary, or ketones if it is secondary; no carbon-carbon
bond cleavage is observed. The reactions are carried out
under mild conditions using inexpensive reagents and
equipment. Although the reaction times are long, the
yields are good and no side products are obtained.
Furthermore, the products can be easily isolated in
relatively pure form.
Oxid a tion of Isoch r om a n (3,4-d ih yd r o-1H-2-ben zop y-
r a n ) w ith KMn O4/Cu SO4‚5H2O: Isochroman (0.188 g, 1.40
mmol), dissolved in 20 mL of dichloromethane, and oxidant
(3.2 g) were placed in a 50 mL round-bottomed flask and
stirred vigorously under gentle reflux. After 72 h, the product
was filtered through a Celite pad and the residue washed
successively with dichloromethane (3 × 20 mL) and ether (3
× 20 mL). Evaporation of the solvent gave the lactone,
2-chromanone (0.198 g, 1.35 mmol, 96%). The identity of the
product was confirmed by NMR and mass spectrometry. 1H
NMR (CDCl3) δ: 7.1-8.0 (m, 4H), 4.45 (t, 2H), 3.95 (t, 2H).
Ms: m/z (%): 148 (M+, 46), 118 (100), 90 (91), 62 (25), 50 (17),
39 (19).
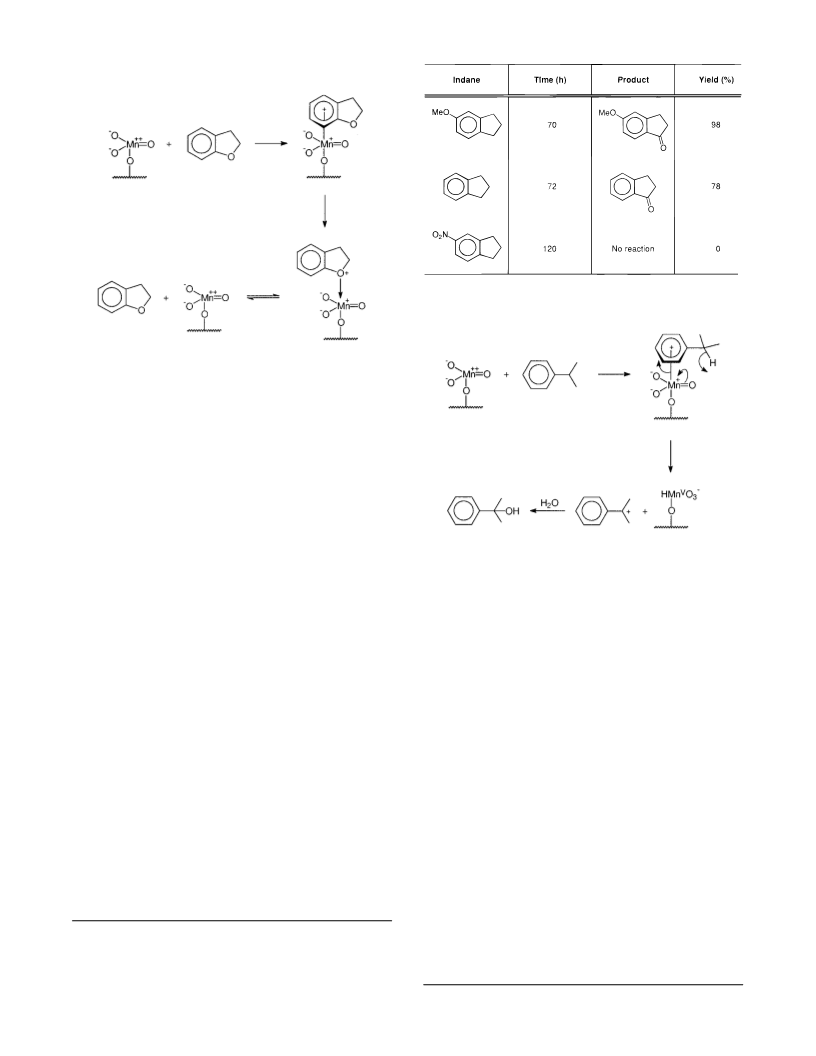
2. A unique selectivity is observed for the oxidation
of cyclic side chains containing an oxygen as part of the
ring. If the oxygen atom is in the 1-position, no oxidation
occurs; if it is in the 2-position, the reaction proceeds
normally to give the corresponding lactone.
3. The reaction is very sensitive to the presence of
substituents on the aromatic ring. p-Nitro groups com-
pletely inhibit the reaction while methoxy groups pro-
mote the reaction and are strongly para-directing.
4. The reaction is inhibited by the presence of cyclo-
hexene which appears to bind irreversibly to the active
sites on the surface of the heterogeneous oxidant.
5. The proposed mechanism involves an initial inter-
action between the aromatic ring and manganese(VII)
giving an η6 complex which reacts by hydride transfer to
produce a benzylic carbocation and reduced manganese.
The benzylic carbocation then reacts with water to give
the corresponding alcohol. If the alcohol is tertiary, it is
resistant to further oxidation; if it is secondary, oxidation
to a carbonyl occurs readily.
Oxid a tion of 6-Meth oxytetr a lin (6-Meth oxy-1,2,3,4-
t et r a h yd r on a p h t h a len e) w it h KMn O4/Cu SO4‚5H 2O:
6-Methoxytetralin (0.227 g, 1.40 mmol), dissolved in 20 mL of
dichloromethane, and oxidant (3.2 g) were placed in a 50 mL
round-bottomed flask and stirred vigorously under gentle
reflux. After 72 h, the product was filtered through a Celite
pad and the residue washed successively with dichloromethane
(3 × 20 mL) and ether (3 × 20 mL). Evaporation of the solvent
gave an impure product (0.235 g). Analysis of this mixture
by GC/MS indicated three components, 6-methoxy-1-tetralone
(85%), 7-methoxy-1-tetralone (5%), and unidentified material
(10%). These products were separated by elution chromatog-
raphy, and the two isomers were identified by comparison of
their 1H NMR spectra with published spectra.30
Exp er im en ta l Section
P r ep a r a tion of Oxid a n ts. KMn O4/Al2O3‚xH2O: Finely
ground potassium permanganate (2.2 g, 14.0 mmol), dissolved
in 5 mL of water, was added to alumina (Acid, Brockman
Attem p ted Oxid a tion of Eth ylben zen e w ith KMn O4/
Cu SO4‚5 H2O in th e P r esen ce of Cycloh exen e: The effect
of cyclohexene on this reaction was assessed by running
parallel experiments, one containing cyclohexene, the other
(26) Perez-Benito, J . F.; Lee, D. G. Can. J . Chem. 1985, 63, 3545-
3550.
(27) Zahonyi-Budo, E.; Simandi, L. I. Inorg. Chim. Acta 1996, 248,
81-84; 1995, 237, 173-5; 1992, 191, 1-2. Simandi, L. I.; J aky, M.;
Schelly, Z. A. J . Am. Chem. Soc. 1984, 106, 6866.
(28) De Klein, W. J . In Organic Syntheses by Oxidation with Metal
Compounds; Mijs, W. J ., De J onge, C. R. H. I., Eds.; Plenum: New
York, 1986; pp 261-314.
(29) Adams, R.; J ohnson, J . R.; Wilcox, C. F. Laboratory Experiments
in Organic Chemistry; 7th ed.; MacMillan: New York, 1979; p 224.
(30) Pouchert, C. J .; Campbell, J . R. The Aldrich Library of NMR
Spectra; Aldrich Chemical Co.: Milwaukee, 1974; Volume 6, No. 12B
and No. 12C.

 Noureldin, Nazih A.
Noureldin, Nazih A.
 Zhao, Dongyuan
Zhao, Dongyuan
 Lee, Donald G.
Lee, Donald G.