Evaluation Only. Created with Aspose.PDF. Copyright 2002-2021 Aspose Pty Ltd.
10.1002/adsc.201800050
Advanced Synthesis & Catalysis
and bromoperoxidations using hydrogen peroxide as
[23b,27]
oxidant.
More recently V7 has been
successfully used as catalysts for the synthesis of
cyclic carbonates via CO2/internal epoxide
[24d]
cycloaddition.
In particular, it has been
Scheme 1. Aerobic C-C oxidative cleavage of diols to the
corresponding carbonyl derivatives.
demonstrated that the tetradentate nature of the
complex and the presence of bulky t-butyl or chloro
groups ortho to the phenol play a fundamental role
for the catalyst stability and activity under turnover
conditions. Furthermore, the nature of the multiple
substituents in the ortho/para positions of the phenol
rings can effectively tune the reactivity of the catalyst.
Only vanadium-based polyoxometalates or Pt/C-V2O5
with high O2 pressures and temperature, strong acidic
conditions could convert cyclohexane-1,2-diols, 1,2-
ethanediol or monosaccharides forming mixtures of
the corresponding carboxylic acids and over-oxidized
products.[18,19]
[23a,b]
Therefore, V(V)-amino triphenolate complexes
V4-V7 seemed to us to be good candidates for
exploring the aerobic oxidative C-C bond cleavage of
vicinal diols.
More recently Baker, Hanson and Thorn reported that
vanadium complexes V1-V3 (Figure 1) are able to
Vanadium complexes V4-V7 can be easily prepared
by mixing the appropriate triphenolamine[28] with an
equimolar amount of V(O)(OiPr)3 in dry CH2Cl2 or
THF under nitrogen. In all cases, high yields of
reddish brown (V4), dark-red (V5), reddish violet
(V6) or dark-blue (V7) crystalline solids were
obtained. For all the complexes, characterizations (1H,
oxidize pinacol under stoichiometric conditions to
[20]
yield acetone.
Complex V1[21] is effective also
under aerobic, catalytic conditions in pyridine or 1-
methyl-2-pyrrolidinone. Under the best conditions,
97% yield of acetone was obtained using 5% of
catalyst at 100°C at atmospheric pressure. [20a]
51
13C and V NMR, FT-IR and ESI-MS) in agreement
with literature data were obtained. [23b,28c]
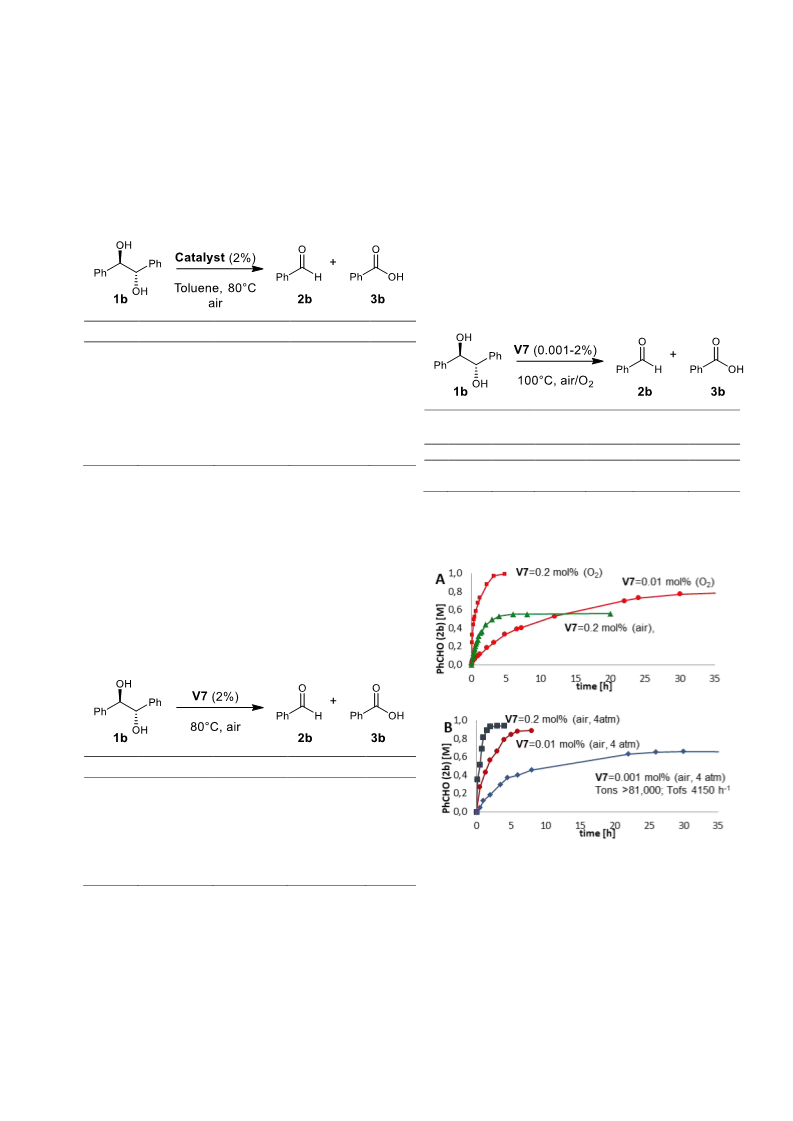
The effectiveness of our systems as catalysts for the
aerobic carbon-carbon cleavage was first tested for
the oxidation of pinacol 1a [0.05M] (Scheme 1,
R=R’=R”=R”’=Me), in the presence of 10% V4 at
80°C under air and in different solvents (toluene,
pyridine and NMP). The course of the reaction was
1
monitored via H NMR. We were very pleased to
observe that V4 was effective, as complete
conversion of the substrate into acetone occurred in
only 2 hours.
Figure 1. Vanadium complexes V1-V7 as catalysts for the
aerobic oxidative carbon-carbon cleavage of diols.
A more detailed study, undertaken in order to define
the performances of V4-V7 was carried out with
meso-hydrobenzoin 1b as model substrate in toluene.
The reactions were carried out at 80°C under air with
2% catalyst loading. For comparison, the behavior of
other vanadium complexes such as V1 and
commercially available vanadium oxo tri-isopropoxy
(VO(OiPr)3) and vanadium oxodiacetylacetonate
(VO(acac)2) were also tested under the same
experimental conditions (Table 1).
The oxidative C-C bond cleavage of 1b affords two
equivalents of benzaldehyde 2b, which, at longer
reaction times, can be over-oxidized to benzoic acid
3b. The reaction courses were monitored via
quantitative HPLC. The results in Table 1 not only
show that V4-V7 can effectively catalyze the process
but also that the electron-withdrawing character of
the TPA ligand plays a major role: V7 (R=R’=Cl),
bearing the most electron-withdrawing substituents,
was significantly more active than the V4-V6
complexes (reaction fourteen-fold faster with respect
to V6, bearing the most electro-donating substituents
(t-Bu/MeO)), as well as the commercially available
VO(OiPr)3 and (VO(acac)2) complexes and catalyst
V1 (Table 1, entries 5-7). Furthermore, the
acceleration of the reaction was found to have a
beneficial effect on the chemoselectivity of the
While the capability of homogeneous vanadium
complexes to catalyze the aerobic C-C bond cleavage
of glycols has been explored to some detail, the
development of an effective and sustainable
vanadium-based catalytic process with broad
substrate scope is still missing. Herein we report that
V(V) amino triphenolate complexes V4-V7 are
efficient catalysts for the oxidative aerobic C-C bond
cleavage of glycols, not only tertiary and benzylic
diols but also cyclic and linear aliphatic diols, both
internal and terminal ones, affording the
corresponding carbonyl derivatives with high yields
and selectivity, with catalyst loadings down to
0.001%, TONs of up to 81,000 and TOFs of up to
4150 h-1.
Results and Discussion
In recent years TPA complexes[22] have been reported
to be effective and tunable Lewis acid catalysts,
affording processes with high turnover numbers and
frequencies, by us[23] and other research groups.
[24,25,26] Among the systems investigated, V(V) tris-t-
butylphenolate complex V4 (Chart 1) is one of the
most active homogeneous catalyst for sulfoxidations
2
This article is protected by copyright. All rights reserved.

 Amadio, Emanuele
Amadio, Emanuele
 González-Fabra, Joan
González-Fabra, Joan
 Carraro, Davide
Carraro, Davide
 Denis, William
Denis, William
 Gjoka, Blerina
Gjoka, Blerina
 Zonta, Cristiano
Zonta, Cristiano
 Bartik, Kristin
Bartik, Kristin
 Cavani, Fabrizio
Cavani, Fabrizio
 Solmi, Stefania
Solmi, Stefania
 Bo, Carles
Bo, Carles
 Licini, Giulia
Licini, Giulia