Angewandte
Chemie
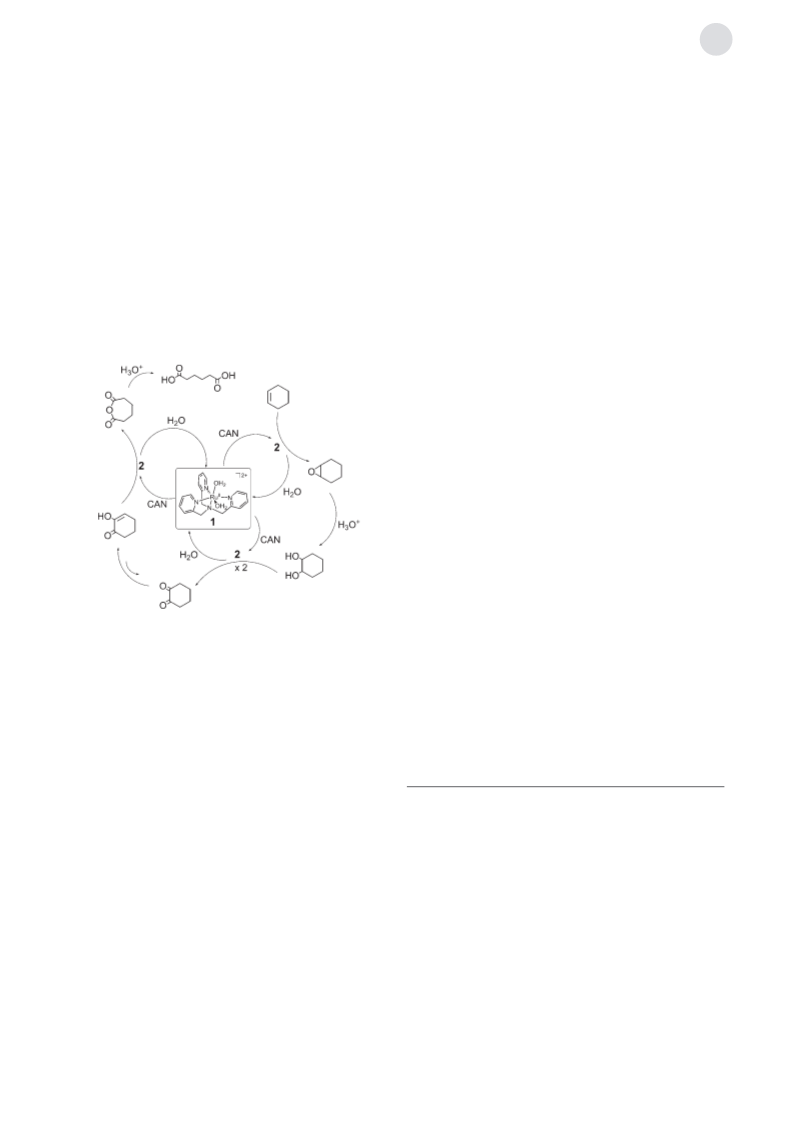
oxide, and cyclohexanedione may also be formed by two 2e
oxidations of the diol. These plausible intermediates en route
to adipic acid were submitted to the catalytic conditions to
afford adipic acid as the sole product. This supports their
involvement as intermediates toward adipic acid. In both
cases, the absence of the catalyst 1 afforded a complicated
mixture of products, including small amount of adipic acid.
This lends credence to the indispensable role of 1 in the
selective oxygenation of cyclohexene to form adipic acid.
Thus, we propose a reaction mechanism involving the
epoxidation of cyclohexene, the hydrolysis of the epoxide to
give cyclohexane-1,2-diol, the 4e oxidation of the diol to give
cyclohexane-1,2-dione, its subsequent Baeyer–Villiger-like
oxidation to give an acid anhydride, and a final hydrolysis
under strongly acidic conditions to give adipic acid
(Scheme 1).[30] All the oxidation steps are 2e oxidations,
which can be performed by 2 as the responsible species.
oxygenation or oxidation. Indeed, we observed a large
turnover number of 2560 for adipic acid formation at a
lower concentration of 1 and higher concentration of CAN
(entry 2, Table 1).
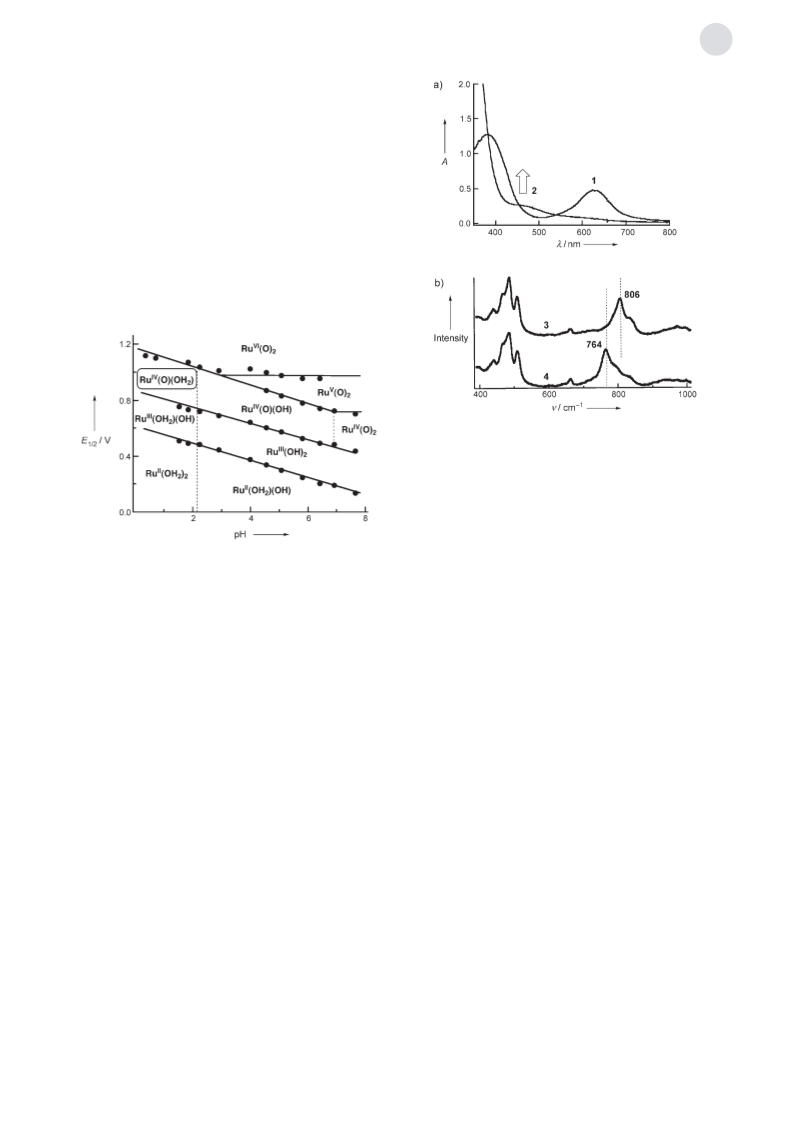
In summary, we have clarified the redox properties of 1
and the formation of the catalytically reactive intermediate-
spin RuIV–oxo species 2 from 1 by PCET. With 1 as a catalyst,
we have established a selective and efficient catalytic oxy-
genation system involving PCET to form reactive species in
water, which acts as both the solvent and as the oxygen
source. Since catalyst durability has been assured, larger
TONs are expected with this system. In addition, according to
the Pourbaix diagram we can control the reactive species in
accordance with the pH value of the solution. This makes it
VI
possible to form RuV O or Ru (O)2 species that probably
=
show higher reactivity than RuIV O species and allows us
=
access to a wider range of oxidation reactions through the
control of pH and the reduction potential of the oxidant.
Experimental Section
Synthesis of [Ru(tpa)(OH2)2](PF6)2·(H2O) (1·H2O):
A mixture
including [RuCl(tpa)]2(PF6)2 (378.0 mg, 0.33 mmol) and AgPF6
(167.1 mg, 0.66 mmol) in H2O (33 mL) was refluxed for 12 h. The
deep-green solution was filtered through a membrane filter to remove
insoluble salt. The filtrate was condensed by rotatory evaporation to
give a green precipitate of 1. The precipitate was filtered and washed
with EtOH followed by ether and then dried in vacuo. The yield of
isolated
1 was 59% (286 mg). Elemental analysis: calcd for
C18H22N4O2RuP2F12·H2O: C 29.32, H 2.96, N 7.72; found: C 29.40,
H 3.29, N 7.62.
General procedure for catalytic oxygenation reactions and
quantitative method: A solution was prepared in 1 mL of D2O to
contain 0.10 mmol of the substrate and 0.001 mmol of the catalyst 1
and a fixed amount of sodium trimethylsilylpropionate-d4 ([D4]TSP)
as an internal standard for NMR quantification. The solution was
treated with 0.20 mmol of CAN, and the mixture was stirred at room
temperature for 30 min. The solution was directly analyzed by
1H NMR spectroscopy. Quantitative analysis was made on the basis
of calibration of peak integration of authentic sample relative to TSP.
Scheme 1. A proposed mechanism for the catalytic oxygenation of
cyclohexene to produce adipic acid in H2O.
Received: March 11, 2008
Published online: June 20, 2008
The stepwise mechanism is also supported by the results
of the oxidations of the water-soluble styrene derivative given
in Table 1 (entries 4 and 5). The amount of CAN added
regulates the product distribution; this confirms that the
initial product is the diol derived from hydrolysis of the
Keywords: cerium · homogeneous catalysis · oxidation ·
.
redox chemistry · ruthenium
À
epoxide and that the diol undergoes the C C bond cleavage
to form the aldehyde, which is further converted to benzoic
acid and formic acid (see Figure S5 in the Supporting
Information).
[3] Cytochrome P450, Structure, Mechanism, and Biochemistry, 2nd
ed. (Ed: P. R. Ortiz de Montellano), Prenum, New York, 1995.
[4] I. Schlichting, J. Berendzen, K. Chu, A. M. Stock, S. A. Maves,
D. E. Benson, R. M. Sweet, D. Ringe, G. A. Petsko, S. G. Sliger,
[5] S.-K. Lee, B. G. Fox, W. A. Froland, J. D. Lipscomb, E. Münck, J.
[6] G. L. Berglund, G. H. Carlsson, A. T. Smith, H. Szöke, A.
We evaluated the durability of 1 in the course of the
catalysis. An aqueous solution of 1 (5.0 10À4 m) was treated
with cyclohexene (1.0 10À1m) and CAN (1.2 10À2 m), and
after the reaction was complete more oxidant was added
several times. The production of adipic acid could be
duplicated and triplicated with no decrease in the product
amount upon addition of further portions of CAN to the same
reaction mixture (see Figure S6 in the Supporting Informa-
tion). The stability of 1 in solution was ensured by UV/Vis and
NMR spectroscopy after catalysis was complete. This enables
us to establish a persistent catalytic cycle for substrate
Angew. Chem. Int. Ed. 2008, 47, 5772 –5776
ꢀ 2008 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
5775

 Hirai, Yuichirou
Hirai, Yuichirou
 Kojima, Takahiko
Kojima, Takahiko
 Mizutani, Yasuhisa
Mizutani, Yasuhisa
 Shiota, Yoshihito
Shiota, Yoshihito
 Yoshizawa, Kazunari
Yoshizawa, Kazunari
 Fukuzumi, Shunichi
Fukuzumi, Shunichi