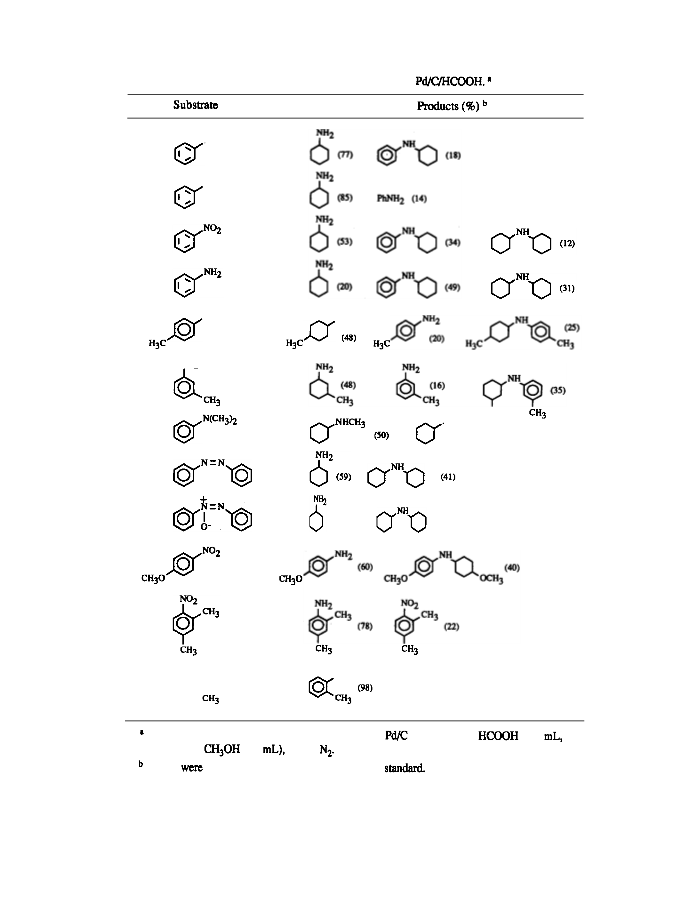
gave aniline in 57% yield, the remainder being recovered nitro compound), while formanilide resulted (from
the reaction of aniline and formic acid) at higher ratios of formic acid to reactant (e.g. at
formanilide was isolated in 77% yield and
as well as aniline and phenyl
in 13% yield). The reduction is
applicable to
(Table I). Good selectivity for the benzene
No reaction
in the absence of either
ring reduction was obtained in the case of
formic acid or With one exception
and
afforded no reaction took place when an
atmosphere of air or carbon monoxide was used for the reaction. A nitrogen containing functionality is
needed in order to achieve reduction of the arene ring, although such a reduction does not occur in all cases.
No hydrogenation of the benzene ring is observed with compounds containing other functional groups, such
as
-OH, or with organophosphorus compounds (e.g.
We found that the reduction was rapidly terminated by the presence of an anion (e.g. Cl-) of a strong
acid. This observation is consistent with the results of Entwistle and
who investigated the
conversion of nitroarenes to aromatic
were run, for 15 minutes at
using 10% palladium-charcoal in formic acid. These reactions
using
acid as the solvent (i.e. a very high ratio of formic
acid/substrate).
It is likely that reduction of the arene ring in the present case is due to the use of a specific,
relatively low ratio of
acid/substrate
Also, the reaction was effected for an extended period of
time compared with the previous methodology.
The
acid likely acts as the hydrogen source in the reduction
with oxidative addition of
The sequence of events (i.e.
the formic acid to palladium resulting in the generation of a palladium hydride.
reduction of the arene and nitrogen functionalities) remains to be determined.
In conclusion the reduction of the arene ring of a number of organonitrogen compounds occurs under
exceptionally mild conditions using formic acid and palladium on carbon.
The following general procedure was used: a mixture of substrate (2 mmol),
and formic acid (0.5 14 mmol) in 10 of methanol was stirred at room temperature for 70 hr under a
nitrogen atmosphere. Work up was effected by filtration and the filtrate was concentrated by evaporation of
(50 mg, 0.05 mmol)
the methanol. The residue was extracted with ether and the solution concentrated to give the
was further purified by silica gel column chromatography.
which
Acknowledgment: We are grateful to British Petroleum and to the Natural Sciences and Engineering
Research of Canada for support of this research.

 Alper, Howard
Alper, Howard
 Vampollo, Guiseppe
Vampollo, Guiseppe