Milwaukee, Wisconsin, 2003; (b) N. Miyaura and A. Suzuki, Chem.
Rev., 1995, 95, 2557; (c) A. Suzuki, J. Organomet. Chem., 1999, 576,
147.
orange and after 20 min became deep-red again. The mixture was
cooled and extracted with petroleum ether (5 × 10 cm3). The com-
bined extracts were washed with water (2 × 10 cm3), and dried over
anhydrous magnesium sulfate. Evaporation of the solvent in vacuo
6 (a) A. H. Soloway, W. Tjarks, B. A. Barnum, F. G. Rong, R. F. Barth,
I. M. Codogni and J. G. Wilson, Chem. Rev., 1998, 98, 1515; (b)
T. D. James, P. Linnane and S. Shinkai, Chem. Commun., 1996, 281.
7 (a) H. C. Brown and T. E. Cole, Organometallics, 1983, 2, 1316;
(b) H. C. Brown, N. G. Bhat and M. Srebrnik, Tetrahedron Lett.,
1988, 29, 2631; (c) W. Li, D. P. Nelson, M. S. Jensen, R. S. Hoerrner,
D. Cai, R. D. Larsen and P. J. Reider, J. Org. Chem., 2002, 65, 5394.
8 (a) T. Ishiyama, M. Murata and N. Miyaura, J. Org. Chem., 1995,
60, 7508; (b) T. Ishiyama, Y. Itoh, T. Kitano and N. Miyaura, Tetra-
hedron Lett., 1997, 19, 3447; (c) T. Ishiyama and N. Miyaura, in
Advances in Boron Chemistry, ed. W. Siebert, The Royal Society of
Chemistry, Cambridge, 1997, p. 92.
1
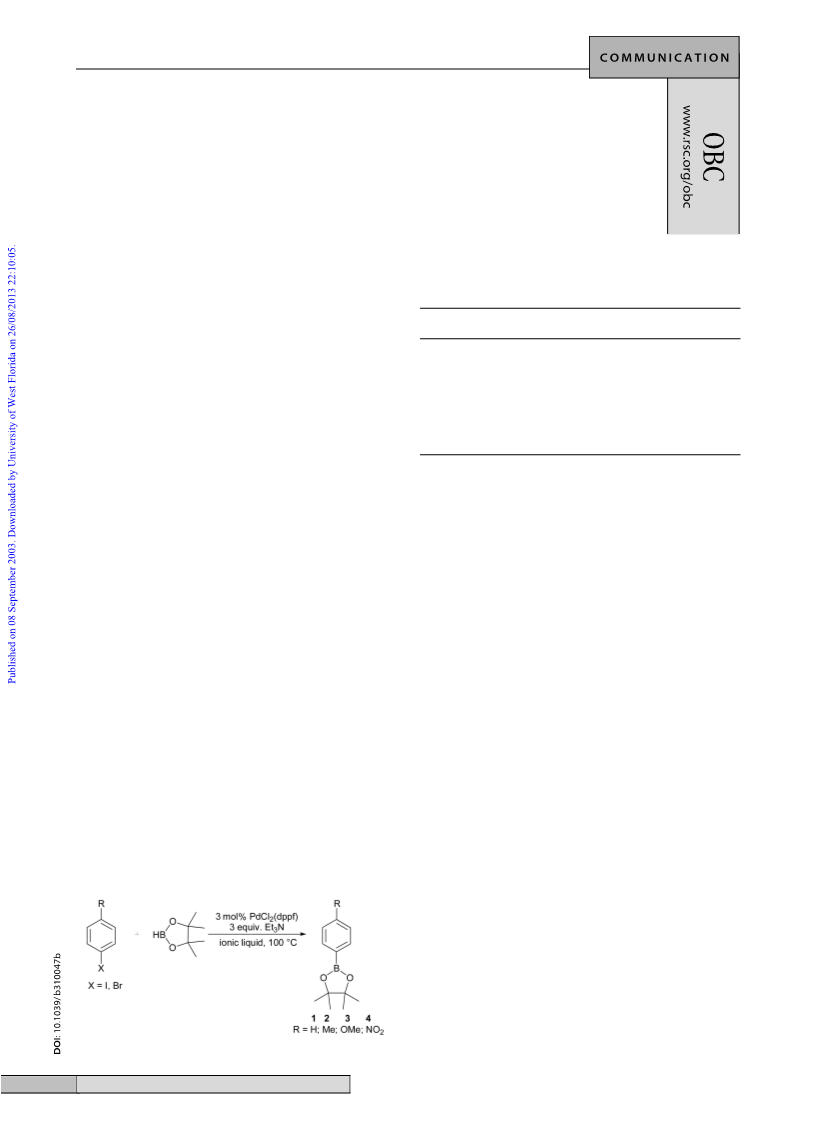
afforded 2 (0.57 g, 87% yield); H NMR (300 MHz, CDCl3) δ 1.34
(s, 12H, CH3), 2.37 (s, 3H, CH3), 7.19 (d, J = 8.2 Hz, 2H, CH), 7.71
(d, J = 8.2 Hz, 2H, CH); 13C NMR (75.5 MHz, CDCl3) δ 21.6 (CH3),
24.8 (CH3), 83.5 (C), 128.5 (CH), 134.8 (CH), 144.3 (C); 11B NMR
(64.2 MHz, CDCl3) δ 31.7. The spectral data were identical with the
data reported in the literature.9a The ionic liquid solution containing
the catalyst was dissolved in dichloromethane (10 cm3), washed with
water (3 × 5 cm3) and dried over anhydrous magnesium sulfate. The
solution can be recycled after evaporation of dichloromethane in
vacuo.
2-(4,4,5,5-Tetramethyl-[1,3,2]dioxaborolan-2-yl)benzoic
acid
9 (a) M. Murata, T. Oyama, S. Watanabe and Y. Masuda, J. Org.
Chem., 2000, 65, 164; (b) M. Murata, S. Watanabe and Y. Masuda,
J. Org. Chem., 1997, 62, 6458; (c) O. Baudoin, D. Guénard and
F. Guéritte, J. Org. Chem., 2000, 65, 9268; (d ) M. Melaimi,
F. Mathey and P. Le Floch, J. Organomet. Chem., 2001, 640, 197;
(e) M. Doux, N. Mézailles, M. Melaimi, L. Ricard and P. Le Floch,
Chem. Commun., 2002, 1566.
10 L. Xu, W. Chen and J. Xiao, Organometallics, 2000, 19, 1123.
11 J. McNulty, A. Capretta, J. Wilson, J. Dyck, G. Adjabeng and
A. Robertson, Chem. Commun., 2002, 1986.
12 Representative procedures: 4,4,5,5-Tetramethyl-2-p-tolyl-[1,3,2]di-
oxaborolane (2). 4-Iodotoluene (0.65 g, 3 mmol) was added to a
suspension of PdCl2(dppf ) (73 mg, 0.09 mmol) in degassed [bmim]-
[BF4] (3 cm3) under argon atmosphere. The mixture was vigorously
stirred at 100 ЊC, and the deep-red solution was obtained. It was
cooled to ambient temperature, triethylamine (0.91 g, 9 mmol) was
added, followed by pinacolborane (0.50 g, 3.9 mmol). The mixture
was heated at 100 ЊC with vigorous stirring. The colour changed to
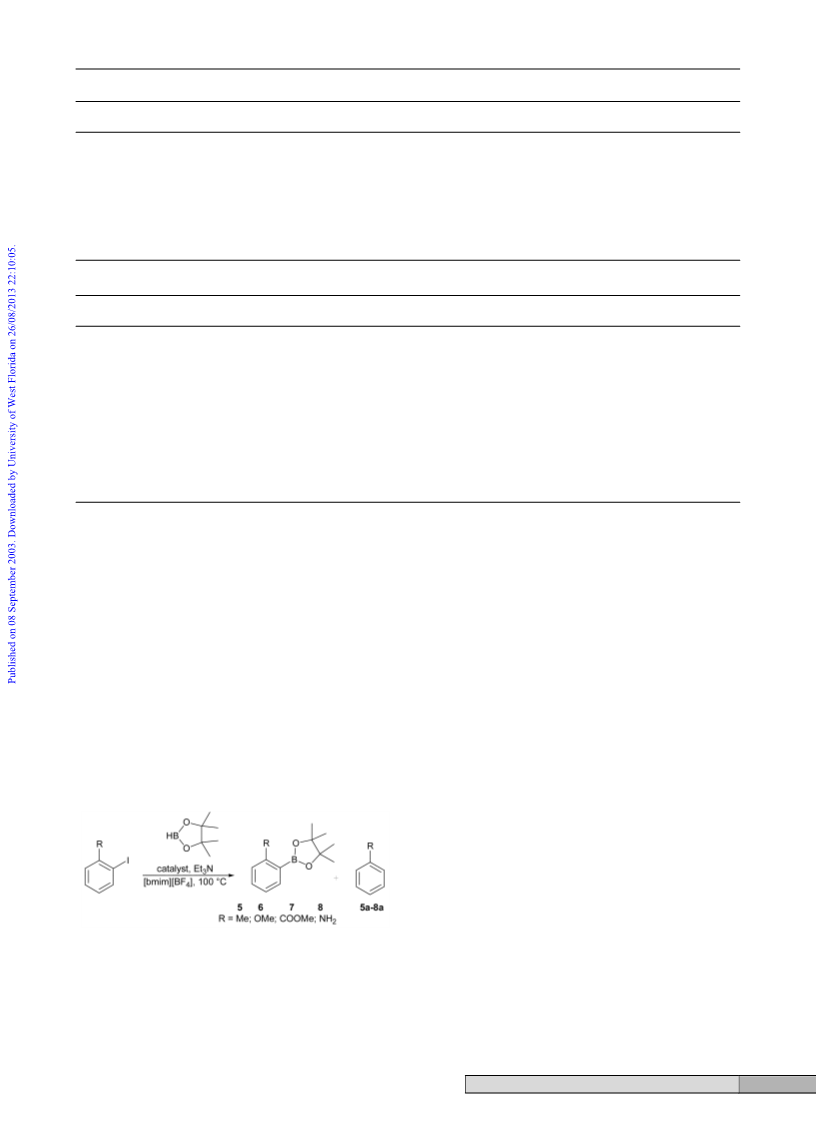
methyl ester (7). 2-Iodobenzoic acid methyl ester (0.77 g, 3 mmol)
was added to a suspension of Pd(OAc)2 (34 mg, 0.15 mmol) in
degassed [bmim][BF4] (3 cm3) under argon atmosphere. The mixture
was vigorously stirred at 100 ЊC, and a brown solution was obtained.
It was cooled to ambient temperature, triethylamine (0.91 g,
9 mmol) was added, followed by pinacolborane (0.50 g, 3.9 mmol).
The mixture was heated at 100 ЊC with vigorous stirring. The colour
changed to yellow and after 6 h became brown again. The mixture
was cooled and extracted with diethyl ether (5 × 10 cm3). The
combined extracts were washed with water (2 × 10 cm3), and dried
over anhydrous magnesium sulfate. Ether was removed in vacuo and
the product was isolated by flash chromatography on silica gel
(hexane–EtOAc, 9 : 1) (0.49 g, 62% yield); 1H NMR (200 MHz,
CDCl3) δ 1.42 (s, 12H, CH3), 3.91 (s, 3H, CH3), 7.41 (m, 1H, CH),
7.51 (m, 2H, CH), 7.94 (ddd, J = 7.4, 1.2, 1.0 Hz, 1H, CH); 13C
NMR (50.3 MHz, CDCl3) δ 24.8 (CH3), 52.1 (CH3), 83.9 (C), 128.7
(CH), 128.9 (CH), 131.7 (CH), 132.1 (CH), 133.4 (C), 168.4 (C); 11
B
NMR (64.2 MHz, CDCl3) δ 31.9 (s).
O r g . B i o m o l . C h e m . , 2 0 0 3 , 1, 3 2 7 4 – 3 2 7 6
3276

 Wolan, Andrzej
Wolan, Andrzej
 Zaidlewicz, Marek
Zaidlewicz, Marek