Communications
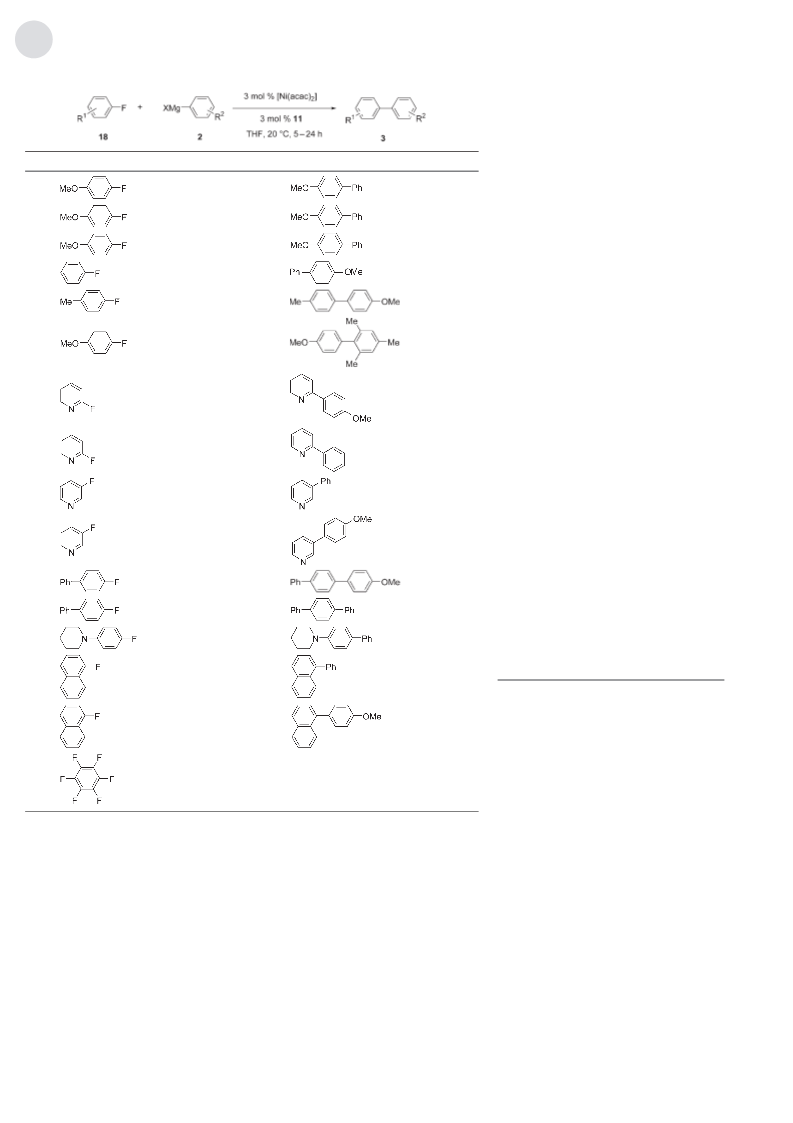
Table 3: Kumada cross-coupling with aryl fluorides.[a]
regioselective functionalization of fluoro-
benzene 1h (Scheme 1).
In summary, we reported the first use of
air-stable secondary phosphine oxides for
ꢀ
the activation of C F bonds in aryl fluo-
Entry
1
Aryl fluoride
Grignard reagent
18a ClMgPh
Product
Yield [%]
3a 80
rides. In particular, a complex derived from
the sterically congested preligand 11
showed activity superior to that of known
protocols for the nickel-catalyzed cross-
coupling reactions of aryl Grignard reagents
at ambient temperature.
2a
2a
2a
2[b]
3[c]
4
18a ClMgPh
18a ClMgPh
3a 55
3a 64
3a 88
3c 88
18b 4-BrMgC6H4OMe 2b
18c 4-BrMgC6H4OMe 2b
Experimental Section
Representative procedure for nickel-catalyzed
cross-coupling reactions of aryl fluorides
(Table 3, entry 15): A solution of [Ni(acac)2]
(7 mg, 0.03 mmol, 3 mol%) and 11 (13 mg,
0.03 mmol, 3 mol%) in dry THF (0.5 mL) was
stirred for 10 min at ambient temperature under
5[d]
6[c]
18b 4-BrMgMes
2c
3i
88
N2.
1-Fluoronaphthalene
(18h)
(148 mg,
1.01 mmol) was added, and the solution was
stirred for 5 min. Thereafter, 2b (0.5m in THF,
3.0 mL, 1.50 mmol) was added. The resulting dark
solution was stirred at ambient temperature for
15 h. Et2O (75 mL) and H2O (75 mL) were added
to the reaction mixture. The separated aqueous
phase was extracted with Et2O (2 75 mL). The
combined organic layers were dried over MgSO4
and concentrated in vacuo. The remaining residue
was purified by column chromatography on silica
gel (n-pentane/Et2O, 300:1!200:1!150:1!
7
18d 4-BrMgC6H4OMe 2b
3 f
3j
90
63
8
9
18d ClMgPh
18e ClMgPh
2a
2a
3g 72
100:1) to yield 3o as
a pale yellow solid
10
18e 4-BrMgC6H4OMe 2b
18 f 4-BrMgC6H4OMe 2b
3h 95
3k 88
(223 mg, 94%).
Received: May 29, 2005
Published online: October 17, 2005
11
12
18 f ClMgPh
18g ClMgPh
2a
2a
3l
89
ꢀ
ꢀ
Keywords: C C coupling · C F activation ·
Grignard reaction · nickel · phosphine oxides
.
13[e]
3m 61
3n 84
14
15
18h ClMgPh
2a
[1] K. Tamao, K. Sumitani, M. Kumada, J. Am.
Chem. Soc. 1972, 94, 4374 – 4376.
[2] R. J. P. Corriu, J. P. Masse, J. Chem. Soc.
Chem. Commun. 1972, 144.
[3] K. Tamao, J. Organomet. Chem. 2002, 653,
23 – 26.
[4] P. Knochel, I. Sapountzis, N. Gommermann,
in Metal-Catalyzed Cross-Coupling Reac-
tions, 2nd ed. (Eds.: A. de Meijere, F. Die-
derich), Wiley-VCH, Weinheim, 2004,
pp. 671 – 698.
[5] A. de Meijere, F. Diederich, Metal-Cata-
lyzed Cross-Coupling Reactions, 2nd ed.,
Wiley-VCH, Weinheim, 2004.
18h 4-BrMgC6H4OMe 2b
18i 4-BrMgC6H4OMe 2b
3o 94
16
–
–
–
[a] Reaction conditions: 18 (1.00 mmol), 2 (1.50 mmol), [Ni(acac)2] (3 mol%), 11 (3 mol%), THF
(2 mL), 208C; yield of isolated product; Mes=2,4,6-Me3C6H2. [b] 16 (3 mol%) instead of 11. [c] 17
(3 mol%) instead of 11. [d] [Ni(acac)2] (5 mol%), 11 (5 mol%). [e] [Ni(acac)2] (8 mol%), 11 (8 mol%).
[6] A. F. Littke, G. C. Fu, Angew. Chem. 2002,
114, 4350 – 4386; Angew. Chem. Int. Ed.
2002, 41, 4176 – 4211.
lene fluoride 18h (entries 14 and 15), was feasible with
phosphine oxide 11. However, electron-poor polyfluorinated
18i was not converted.
Finally, a combination of a palladium-catalyzed amina-
tion[14] and a nickel-catalyzed Kumada cross-coupling of the
resulting electron-rich aryl fluoride 18j was used for the
[7] T. G. Richmond, Angew. Chem. 2000, 112, 3378 – 3380; Angew.
Chem. Int. Ed. 2000, 39, 3241 – 3244.
[8] T. Braun, R. N. Perutz, Chem. Commun. 2002, 2749 – 2757.
[9] T. Takahashi, K. Kanno in Modern Organonickel Chemistry
(Ed.: Y. Tamaru), Wiley-VCH, Weinheim, 2005, pp. 41 – 55.
7218
ꢀ 2005 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Angew. Chem. Int. Ed. 2005, 44, 7216 –7219

 Ackermann, Lutz
Ackermann, Lutz
 Born, Robert
Born, Robert
 Spatz, Julia H.
Spatz, Julia H.
 Meyer, Daniel
Meyer, Daniel