408
KARAKHANOV et al.
EXPERIMENTAL
Materials
Guaiacol was hydrogenated in an aqueous solution
in the presence of a MPF–PPI-G3–Pd dendrimer-
containing palladium catalyst and Pd/C at
=
PH
2
The reactants were guaiacol (≥98%, Sigma-
50 atm and 200°C for 1–5 h. The catalytic test results
and the reaction conditions are shown in Table 1. It
was found that the main reaction route is the aromatic
ring hydrogenation to form 2-methoxycyclohexanol.
With an increase in the reaction time, the products are
2-methoxycyclohexanol and cyclohexanol; this find-
ing indicates the occurrence of a hydrogenation–
demethoxylation process.
Aldrich); triblock copolymer Pluronic F127 (Mn =
12600, EO106-PO70-EO106, Aldrich); phenol (chemi-
cally pure grade, Reakhim); formaldehyde (37%
aqueous solution, Sigma-Aldrich); sodium hydroxide
(reagent grade, Irea 2000); hydrochloric acid (reagent
grade, Irea 2000); sulfuric acid (reagent grade, Irea
2000); and chloromethyl methyl ether (technical
grade, Sigma-Aldrich). The solvents were methanol
(99+%, Acros Organics); ethanol (analytical grade,
Irea 2000); chloroform (analytical grade, Ecos-1); and
acetone (analytical grade, Ekos-1). Dendrimer
DAB(NH2)16 was synthesized under laboratory condi-
tions as described in [18]. Palladium(II) acetate
(Aldrich, 99.9%) was used as the metal source.
Figure 1 shows representative structures of palla-
dium catalysts based on ordered mesoporous phenol–
formaldehyde polymers modified with poly(propylene
imine) dendrimers (left) and poly(propylene imine)
dendrimers quaternized with sulfuric acid (right). The
addition of sulfuric acid to the reaction mixture leads
to an increase in the reaction rate and significantly
affects the reaction mechanism: deoxygenation pro-
cesses occur more vigorously in an acid medium.
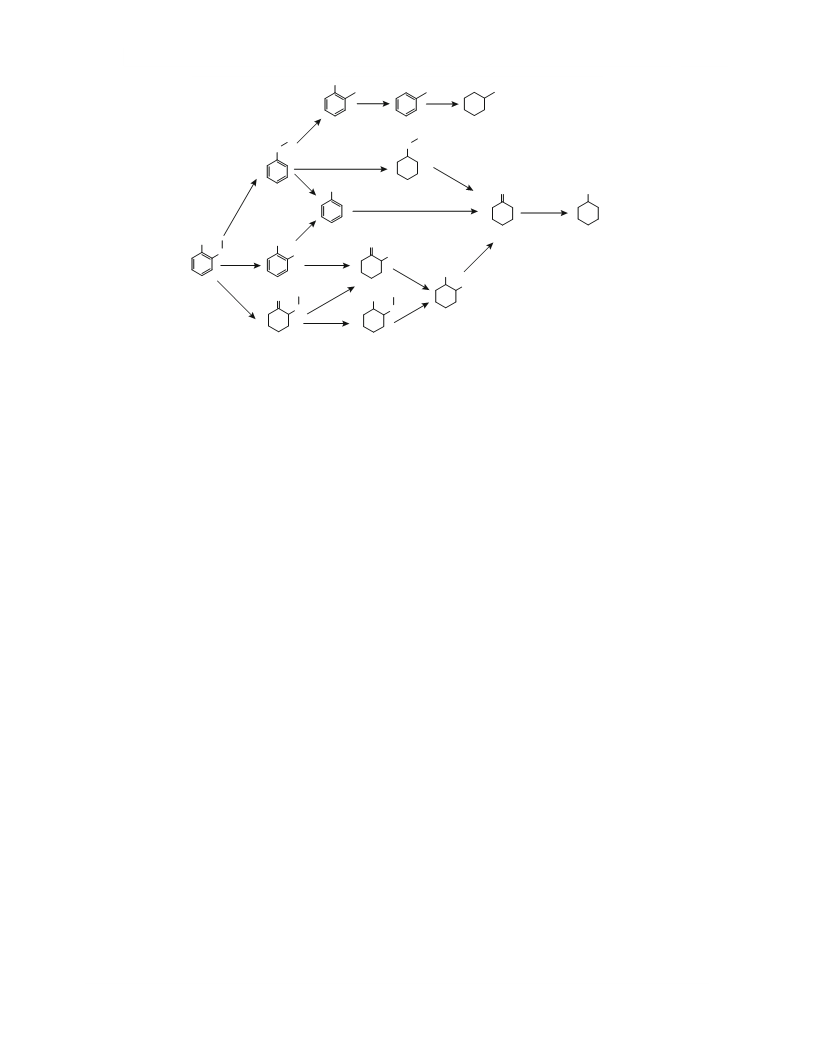
Thus, after the reaction run for 1 h, the products com-
prise, in addition to cyclohexanol and 2-methoxycy-
clohexanone, catechol, cyclohexanediol-1,2, 2-hyd-
roxycyclohexanone, anisole, o-cresol, methoxycyclo-
hexane, and toluene (Table 2). The addition of the
acid induces a more complex mechanism of guaiacol
hydrogenation, which involves the simultaneous
occurrence of different processes, namely, the aro-
matic ring hydrogenation to form 2-methoxycyclo-
hexanone, demethylation to form pyrocatechol, and
demethoxylation to form anisole. In turn, 2-methoxy-
cyclohexanone could be further converted to 2-me-
thoxycyclohexanol and 2-hydroxycyclohexanol,
which were subsequently hydrogenated to cyclohex-
anediol-1,2 and cyclohexanol. Anisole was converted
to phenol and methoxycyclohexane, which were sub-
sequently transformed to cyclohexanol and o-cresol,
which underwent dehydroxylation to form toluene.
After the reaction run for 5 h, the main products were
cyclohexanol (73%), methoxycyclohexane (19%), and
methylcyclohexane (6%). In this case, the reuse of the
catalyst led to a significant decrease in the substrate
conversion; this finding can be attributed to the elu-
tion of the metal from the pores of the support by the
acid. Thus, on the basis of the derived data, a possible
route of guaiacol hydrogenation using an MPF–PPI-
G3–Pd catalyst and sulfuric acid has been proposed
(Fig. 2). It should be noted, in the case of addition of
the acid to Pd/C, the reaction occurred in accordance
with a similar mechanism; however, after the reaction
run for 5 h, the products still comprised substances
with two oxygen-containing groups; this finding sug-
gests that hydrodeoxygenation occurred at a lower
rate.
Catalyst Testing Procedure
Catalytic tests on hydrogenation were conducted in
a steel autoclave at high hydrogen pressure under vig-
orous stirring of the reaction mixture. Calculated
amounts of the catalyst, the substrate, and water were
placed in an autoclave equipped with an insertable
glass test-tube and a magnetic stirrer. In cases stipu-
lated by the experimental procedure, 110 μL of sulfuric
acid was added to the reaction mixture. The autoclave
was filled with hydrogen to a pressure of 5.0 MPa at
room temperature and placed into a furnace preheated
to 200°C for a predetermined time. After reaction, the
autoclave was cooled below room temperature and
depressurized. The catalyst was separated from the
solution by centrifugation. To provide the complete
dissolution of organic compounds, acetone was added
to the sample before chromatographic studies. Hydro-
genation products were analyzed on a Kristallyuks
4000 M chromatograph equipped with a flame ioniza-
tion detector and a Petrocol DH 50.2 capillary column
coated with the polydimethylsiloxane stationary liquid
phase (dimensions, 50 m × 0.25 mm; carrier gas,
helium; split ratio, 1 : 90). In addition, liquid products
were analyzed by chromatography–mass spectrome-
try using a Finnigan MAT 95 XL instrument equipped
with a chromatograph with a Varian VF-5MS capillary
column (30 m × 0.25 mm × 0.25 μm) using helium as
a carrier gas (1.5 cm3/min). Results were processed
using the Xcalibur software package. Products were
identified by comparing their mass spectra with the
spectra of compounds available in the library attached
to the software.
RESULTS AND DISCUSSION
The synthesis procedure for the MPF–PPI-G3–
For comparison, Table 2 shows data on guaiacol
Pd catalyst (2.9% N, 11.6% of a third-generation hydrogenation in the presence of palladium catalysts
poly(propylene imine) dendrimer, 10.0 wt % Pd) and with additives of zeolite solid acids. It is evident from
the catalyst characteristics are described in [19, 20].
the table that, in the case of using a mixture of MPF–
PETROLEUM CHEMISTRY
Vol. 58
No. 5
2018

 Karakhanov
Karakhanov
 Boronoev
Boronoev
 Filippova, T. Yu.
Filippova, T. Yu.
 Maksimov
Maksimov