Tetrahedron Letters
Direct bromodeboronation of arylboronic acids with CuBr in water
2
Yan-Ling Tang a,1, Xian-Song Xia b,1, Jin-Chun Gao , Min-Xin Li , Ze-Wei Mao
a
a
a,
⇑
a
College of Pharmaceutical Science, Yunnan University of Chinese Medicine, Kunming 650500, PR China
Teaching Affairs Department, Yunnan University of Chinese Medicine, Kunming 650500, PR China
b
a r t i c l e i n f o
a b s t r a c t
Article history:
An efficient and practical method has been developed for the preparation of aryl bromides via the direct
bromodeboronation of arylboronic acids with CuBr in water. This strategy provides several advantages,
such as being ligand-free, base-free, high yielding, and functional group tolerant.
Ó 2020 Elsevier Ltd. All rights reserved.
Received 5 October 2020
Revised 24 November 2020
Accepted 2 December 2020
Available online 30 December 2020
2
Keywords:
Bromodeboronation
Arylboronic acids
CuBr
Aryl bromides
2
Aryl halides are valuable synthetic intermediates in organic
complexes for the bromination of arylboronic acids in the presence
of NaNO (Scheme 1d) [10].
chemistry that have been widely applied in carbon–carbon and
carbon-heteroatom bond formation [1]. Especially, aryl bromides
can be utilized in various coupling reactions to obtain functional-
ized cores [2]. Therefore, the exploration of efficient, regioselective
and concise methods for the preparation of aryl bromides is of
great importance. Traditional methods for the synthesis of aryl
bromides such as the Sandmeyer reaction [3] and direct elec-
2
Although various bromodeboronation reactions of arylboronic
acids has been reported, there are often limitations in these meth-
ods: (1) specific reagents need to be prepared, (2) ligands or bases
are required, (3) requirement for special reaction condition, such as
an oxygen atmosphere, and (4) toxic reagents and solvents. There-
fore, it is still worthwhile to develop inexpensive and effective
reaction conditions for the formation of aryl bromides from aryl-
boronic acids.
Copper is an abundant and inexpensive metal, which is widely
used in transition metal-catalyzed coupling reactions, such as
Sonogashira cross-coupling, Ullmann coupling and Chan-Lam cou-
pling [11]. In the reported studies, copper complexes have shown
good catalytic activity toward the preparation of biaryls [12] and
phenols [13] from arylboronic acids. Based on this, we envisioned
2
trophilic aromatic substitution with Br [4] or NBS [5] often suffer
from limitations including harsh reaction conditions, toxic
reagents and solvents, and low yields. In addition, although transi-
tion metal-catalyzed CAH bromination of arenes represents an
efficient choice, expensive palladium salts and phosphine ligands
restricts the application [6].
Arylboronic acids are important organic chemical intermedi-
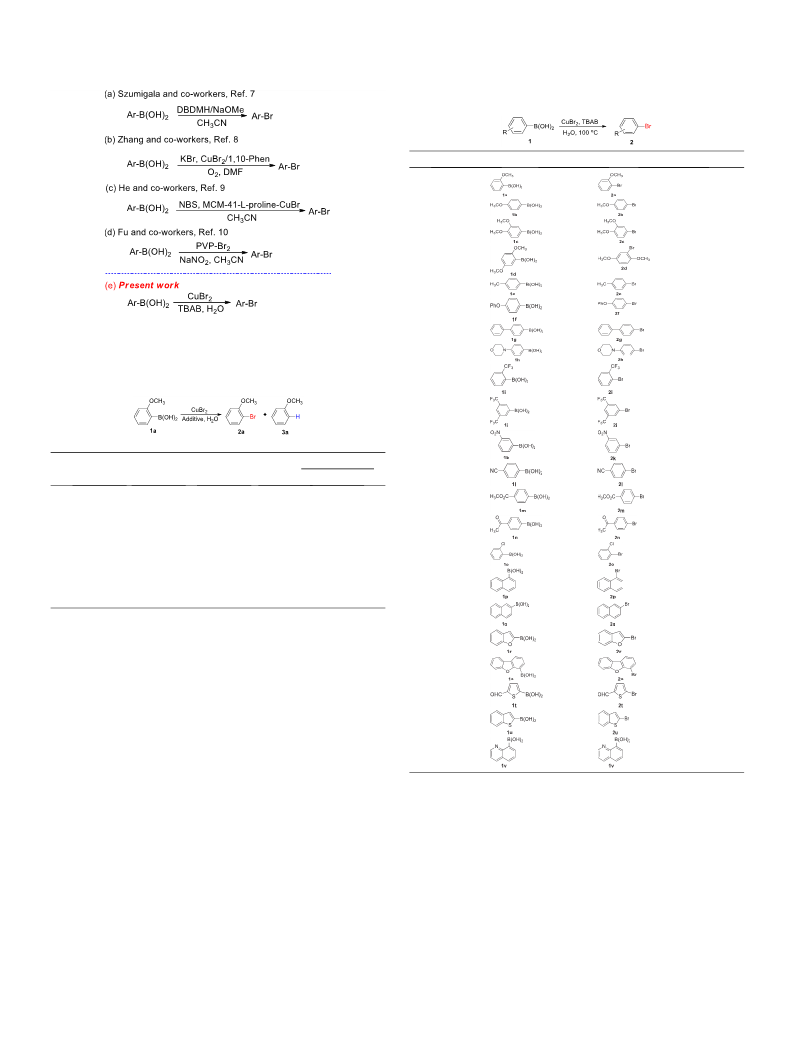
ates, which can be converted into aryl halides. In 2004, Szumigala
and co-workers reported the synthesis of aryl bromides from aryl-
boronic acids with 1,3-dihalo-5,5-dimethylhydantoin via NaOMe-
catalyzed bromodeboronation (Scheme 1a) [7]. In 2011, Zhang
and co-workers developed a copper-catalyzed halogenation reac-
tion between arylboronic acids and halogen salts under oxygen
that CuX
the best of our knowledge, there are no reports on the direct bro-
modeboronation of arylboronic acids with CuBr to aryl bromides
2
could promote the halogenation of arylboronic acids. To
2
in water. In the present work, we have developed an efficient bro-
modeboronation of arylboronic acids for the synthesis of aryl bro-
mides in water under ligand-free and base-free conditions.
2-Methoxyphenylboronic acid (1a) was selected as a model
substrate to identify the optimal reaction conditions (Table 1).
(
Scheme 1b) [8]. In 2017, He and co-workers developed the cop-
per(I)-catalyzed bromodeboronation of arylboronic acids with N-
bromosuccinimide (NBS) using a MCM-41-immobilized CuCl com-
plex as the catalyst (Scheme 1c) [9]. In 2019, Fu and co-workers
reported the use of poly (4-vinyl pyridine) supported bromine
2
Firstly, when 0.2 eq. CuBr was used, 2-methoxyphenylboronic
acid was not converted into the desired product in three different
solvents (Table 1, Entries 1–3). When 10 mol% tetrabutylammo-
nium bromide (TBAB) was added [14], aryl bromide 2a was
⇑
These authors contributed equally to this work.
obtained in 12% yield in H
2
O, and anisole 3a was obtained in 26–
1
33% yield via protodeboronation (Table 1, Entries 4–6). These
040-4039/Ó 2020 Elsevier Ltd. All rights reserved.
0

 Tang, Yan-Ling
Tang, Yan-Ling
 Xia, Xian-Song
Xia, Xian-Song
 Gao, Jin-Chun
Gao, Jin-Chun
 Li, Min-Xin
Li, Min-Xin
 Mao, Ze-Wei
Mao, Ze-Wei