RSC Advances
Page 4 of 5
DOI: 10.1039/C6RA04919B
ESI). A little increase in the SP3 carbon and –COOH groups was
observed in XPS of the spent catalyst (Fig S&, ESI) perhaps due
to the oxidation of some carbon in SP2 state to bear extra –COOH
groups at reaction conditions. This is in consistant with the
relatively higher weak acid sites observed in TPD of the spent
catalyst. Small amount of an additional component related to CꢀN
was also appeared at 287.5 eV in the spent catalyst.30 But, these
changes do not influenced the activity of the spent catalyst.
The catalyst also exhibited good activity towards
10 esterification reaction of levulinic acid with butanol (Table 3).
The levulinic acid conversion is ~70% at 70°C and increased with
the reaction temperature up to 120°C. The highest conversion of
91% levulinic acid was obtained at 120°C after 8 h reaction time.
The selectivity towards nꢀbutyl levulinate is always 100% that
15 makes the product useful for fuel applications.
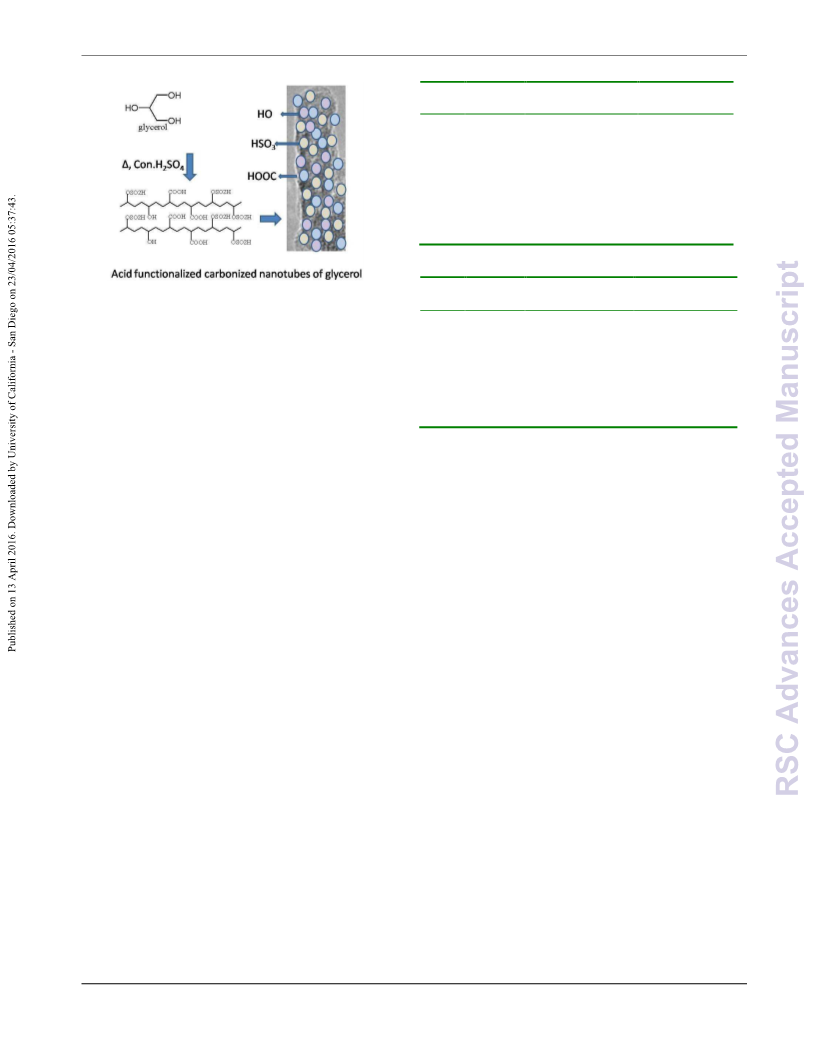
In conclusion, the present communication demonstrates a
facile and efficient carbonizationꢀacidification of bioꢀwaste
glycerol to produce high acidꢀbearing carbonized nanotubes of
glycerol that can catalyze important reactions such as
20 acetalization of glycerol and esterificaitn of levulinic acid to
produce valuable bioꢀfuel suitable for blending applications.
Moreover, the active material does not suffer from leaching
problems and can be efficiently reused in consecutive catalytic
cycles.
We acknowledge the CSIR for the research funding of the
project under 12th FYP. We are thankful to XRD, IR, SEM, GC
and GCꢀMass groups at IIP for analysis and Dr. Myo Tay Zar
Myint, Department of Physics, College of Science, Sultan
Qaboos University, Muscat, Sultanate of Oman for XPS analysis.
5. S. Suganuma, K. Nakajima, M. Kitano, D. Yamaguchi, H. Kato, S.
Hayashi and M. Hara, J. Am. Chem. Soc., 2008, 130, 12787.
70 6. A.M.B. Furtado, J. Liu, Y. Wang and M.D. LeVan, J. Mater. Chem.,
2011, 21, 6698.
7. P. Gupta and S. Paul, Green Chem., 2011, 13, 2365.
8. K. Komura, Y. Nakano and M. Koketsu, Green Chem., 2011, 13, 828.
9. S. Van de Vyver, L. Peng, J. Geboers, H. Schepers, F. de clippel, C.J.
5
75
Gommes, B. goderis, P. A. Jacobs and B. F. Sels, Green Chem., 2010,
12, 1560.
10. S. Gu and M. Jaroniec, J. Mater. Chem., 2011, 21, 6389.
11.M.H. Zong, Z.Q. Duan, W.Y. Lou, T.J. Smith and H. Wu, Green
Chem., 2007, 9, 434.
80 12. A. Takagaki, M. Toda, M. Okamura, J.N. Kondo, S. Hayashi and K.
Domen, Catal. Today, 2006, 116, 157.
13. X.H. Mo, D.E. Lopez, K. Suwannakarn, Y. Liu, E. Lotero and J.G.
Goodwin, J. Catal., 2008, 254, 332.
14. X.H. Mo, E. Lotero and C.Q. Lu, Catal. Lett., 2008, 123, 1.
85 15. B.L.A.P.Devi, K.N.Gangadhar, P.S.S.Prasad, B.Jagannadh and
R.B.N. Prasad, Chem.Sus.Chem., 2009, 2, 617.
16.J.A. MaciáꢀAgulló, M. Sevilla, M.A. Diez and A.B. Fuertes,
ChemSusChem.,2010, 3, 1352.
17 W.Y. Lou and M.H. Zong, Bioresour. Technol., 2008, 99,8752.
90 18 R. Liu, X.Q. Wang, X. Zhao and P.Y. Feng, Carbon., 2008,46, 1664.
19. Y. Gao, X. Chen, J. Zhang and N. Yan, Chem Plus Chem., 2015, 80,
1556.
20. N. Yan and X. Chen, Nature, 2015, 524, 155.
21. E.L.S. Ngee, Y. Gao, X. Chen, T.M. Lee, Z. Hu and D. Zhao, Ind.
95
Eng. Chem. Res., 2014, 53, 14225.
25
22. D. Nandan and N. Viswanadham, RSC Adv.,2011, 47, 11537.
23. O. Ioannidou and A. Zabaniotou, Renw. Sustain. Energ. Rev.,
2007, 11, 1966.
24. S. Y. Chen, T. Yokoi, C. Y. Tang, L. Y. Jang, T. Tatsumi, J.C.C.
Chana and S, Cheng, Green Chem., 2011,13, 2920.
25. J. Deng, L. P. Mo, F. Y. Zhao, L. L. Hpu, L. Yang and Z. H. Zhang,
Green Chem., 2011, 13, 2576.
100
30
Notes and reference
26. K.Y.Nadiwale, V.V.Bokade, Chem. Eng.Technol., 2015, 38, 246.
27. C. Mattevi, S. Hofmann, M. Cantoro, A.C. Ferrari, J. Robertson, C.
105 CastellarinꢀCudia, S. Dolafi, A. Goldoni and C. Cepek, Physica E: Low-
dimensional Systems and Nanostructures, 2008, 40, 2238.
1. L. Khezami, A. Chetouani, B. Taouk and R. Capart, Podwer Technol.,
2005, 157, 48.
28. S.K. Jerng, D.S. Yu, J.H. Lee, C. Kim, S. Yoon and S.H. Chun, ,
Nanoscale Res. Lett., 2011, 6, 1.
35 2. M. Toda, A. Takagaki, M. Okamura, J. N. Kondo, S. Hayashi, K.
Domen and M. Hara, Nature, 2005, 438, 178.
29. Y. Liu, C. Tian, B. Yan, Q. Lu, Y. Xie, J. Chen, R. Gupta, Z. Xu,
110 S.M. Kuznicki, Q. Liu and H. Zeng, RSC Adv., 2015, 5, 15634.
30. A.P. Dementjev, A. deꢀGraaf, M.C.M. vanꢀdeꢀSanden, K.I. Maslakov,
A.V. Naumkin and A.A. Serov, Diamond Relat. Mater., 2000, 9, 1904.
3. Yamaguchi, M. Kitano, S. Suganuma, K. Nakajima, H. Kato and M.
Hara, J. phys. Chem. C, 2009, 113, 3181.
4. M. Kitano, D. Yamaguchi, S. Satoshi, K. Nakajima, H. Kato, S.
40
45
50
55
60
65
Hayashi and M. Hara, Langmuir, 2009, 25, 5068.
115
4
| Journal Name, [year], [vol], 00–00
This journal is © The Royal Society of Chemistry [year]

 Viswanadham, Nagabhatla
Viswanadham, Nagabhatla
 Debnath, Suman
Debnath, Suman
 Saxena, Sandeep K.
Saxena, Sandeep K.
 Al-Muhtaseb, Ala'A H.
Al-Muhtaseb, Ala'A H.