Table
(
1
Epoxidation and hydration of styrene derivatives
) catalyzed by Mn–salen/TiP–PhGly
1
2
4-R PhCR QCH
2
Epoxide
selectivity
%ee
Diol
selectivity
%ee
Conversion
(%)
Substrate
1
2
R = H, R = H
17
84
20
86
25, 36
35, 7
15, 10
25, 5
65, >99
20, >99
40, >99
25, >99
1
R = H, R = Me
2
1
R = Me, R = H
2
1
2
R = Me, R = Me
1
2
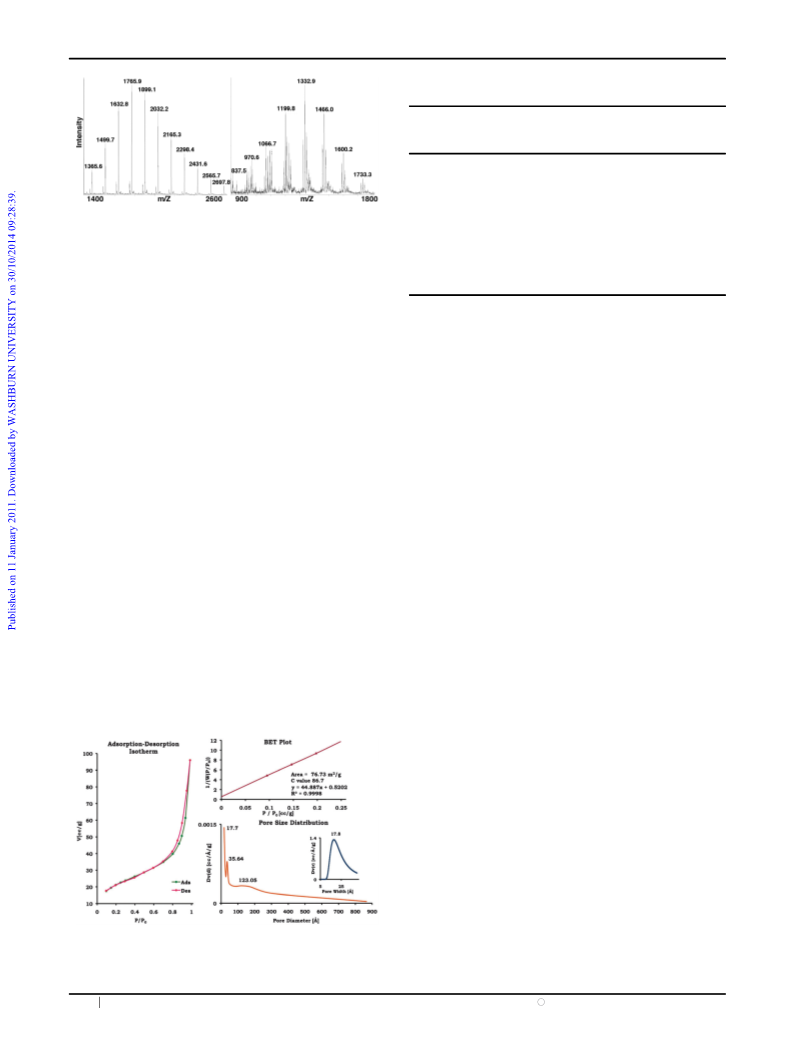
Fig. 1 MALDI MS of P1 (left) and P3 (right). For P1 there are M
and M + Na (major) peaks. For P3 there are peaks for M, M + Na,
M + 2Na, M + 3Na, M + Na + K and M + 2Na + K with Z = 2,
and M, M + Na, M + K with Z = 1.
2
Reaction conditions: 4-R PhCR QCH (0.1 mmol), NaOCl (0.5 mL
buffered solution, 0.2 mmol oxidant), THF (0.5 mL), Mn–salen/
TiP–PhGly (10 mg), 3 1C, 48 h. Conversion and selectivity were
determined by GC; mostly chlorohydrins and dichlorostyrene derivatives
were the by-products in a non-catalytic reaction.y The %ee was
determined by HPLC. The S isomer was the major isomer in all cases.
phosphonate functionalities (P2 and P3) with mild hydrolysis
III
carried out by Me SiBr. Finally in the presence of Mn –salen,
3
Ti–iso-propoxide was used for the non-hydrolytic condensation
with the phosphonate P3 to yield the desired tripodal
polypeptide scaffolds which were then immobilized by
the titanium phosphonate network (TiP–PhGly) with
titanium phosphonate octahedral coordination sphere. The
solid-state absorption spectra of Mn–salen/TiP–PhGly and of
TiP–PhGly alone, see ESIw, clearly showed the incorporation
of Mn–salen in the material.
III
encapsulated Mn –salen.
The Mn–salen/TiP–PhGlyz material was tested for a
The degree of polymerization of L-PhGly was determined by
MALDI mass spectrometry and showed a distribution of
peaks attributable to 9–19 L-PhGly moieties in P1, Fig. 1, with
a maximum at m/z = 1766 assigned to the molecular cation
with 12 L-PhGly units plus Na. The MS of P3 reveals a
distribution of peaks with a repeating unit of 66 indicating a
range of doubly charged cation adducts for longer polymers.
These are overlapped by peaks with a repeating unit of 132 for
the shorter polymers.
consecutive reaction sequence where Mn–salen catalyzes the
1
4
epoxidation of a styrene derivative with NaOCl and the
homochiral TiP–PhGly scaffold catalyzes the highly enantio-
selective ring opening of the intermediate epoxide to the
1
1
corresponding diol. The results are shown in Table 1.
There are several notable points concerning the catalytic
results. (1) Although Mn–salen is achiral, its encapsulation
within the homochiral TiP–PhGly led to the formation of the
S epoxide with low to moderate enantioselectivity. This is a
rather unusual case where the chirality of the reaction medium
induces an enanatioselective transformation using an achiral
Mn–salen/TiP–PhGly is not soluble in any solvent due to
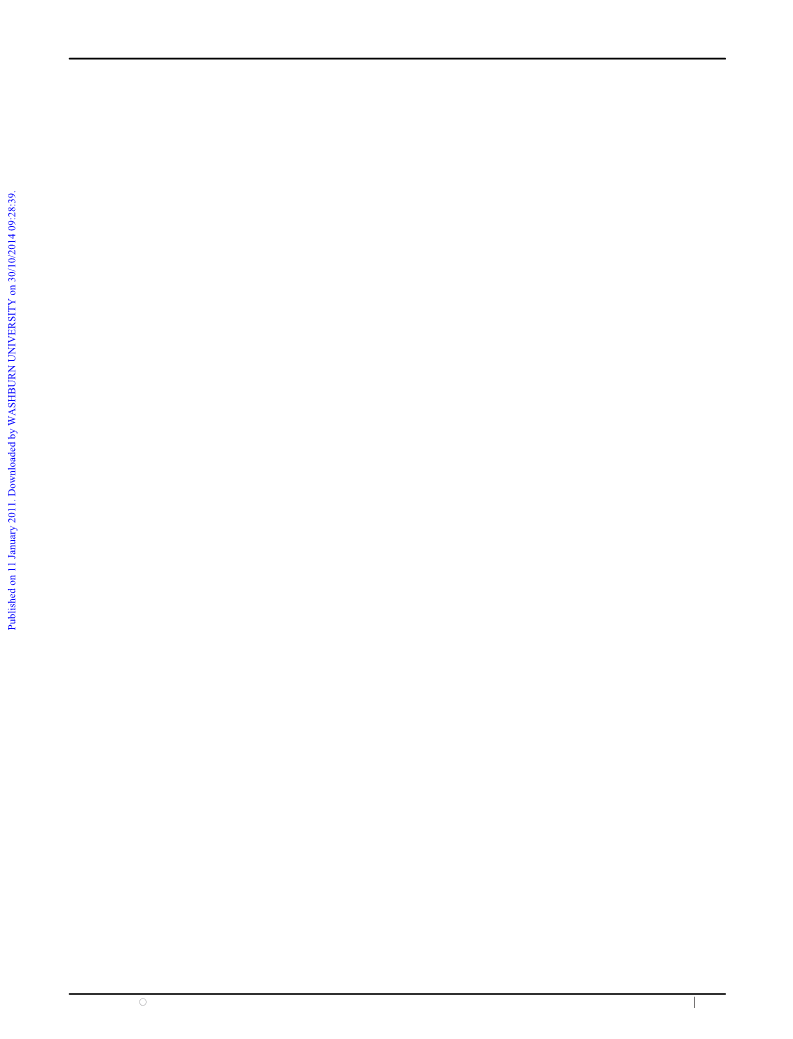
the cross-linked nature of metal–phosphonate structures. BET
measurements showed a moderately high specific surface area
1
5,16
11
catalyst.
(2) As previously described for TiP–Leu, y the
2
ꢀ1
of 77 m g with a wide distribution of nanometric pore sizes,
˚
notably micropores with an average diameter of 18 A and
hydration of the epoxide to the diol proceeds with almost
exclusive formation of the S diol. We have suggested that the
epoxide is activated by the polypeptide scaffold, followed by
an enantioselective attack of water at the benzylic position
directed by the homochiral peptide to yield S-styrene glycol.
˚
mesopores of 35 and 123 A, Fig. 2; see ESIw for details.
Mn–salen/TiP–PhGly was analyzed by elemental analysis
and thermogravimetrically, see ESI.w The EDS measurement
showed a Mn : P ratio of 1 : 9 indicating on average one
Mn–salen complex per 3 tripodal polypeptides. The Ti : P
ratio was 5 : 1. This ratio suggests that a cross-linked polymer
may contain tripod arms linked via ten titanium groups
between each two phosphorus atoms, and organized in a
1
8
This was supported by using H2 O in the hydration reaction
of racemic epoxides by a non-encapsulated polypeptide
1
1
titanium–phosphonate scaffold. (3) The catalytic system is
stable towards deactivation and leaching; recovery-recycle was
carried out 3 times using styrene as the substrate with
negligible loss to conversion and selectivity, neither in the case
when the catalyst was washed and dried between recycles nor in
the case when it was reused as is. The later method was also used
to see if the pores were ‘‘blocked’’ by the substrate or product.
This research presents a proof of principle that an achiral
catalyst encapsulated within a homochiral, insoluble porous
material can lead to heterogeneous reactions with very high
enantioselectivity and effective catalyst recovery.
Notes and references
z Mn–salen/TiP–Leu was also active for epoxidation–hydration
reaction cascade, but the epoxide enantioselectivities were slightly
lower. One can hypothesize that this is due to a better interaction
between Mn–salen and PhGly versus Leu.
y In reactions without Mn–salen, that is with TiP–PhGly only, no
epoxide or diols were obtained although the chlorohydrins and
dichlorostyrene derivatives were obtained as may be expected for
Fig. 2
2
N adsorption isotherm (left), BET surface area (top right)
and Barrett Joyner Halenda and Dubinin–Astakhov (inset) plots for
mesopores and micropores, respectively (right bottom).
2
non-catalytic addition of HOCl and Cl to the double bonds.
2
536 Chem. Commun., 2011, 47, 2535–2537
This journal is c The Royal Society of Chemistry 2011

 Milo, Anat
Milo, Anat
 Neumann, Ronny
Neumann, Ronny