
Journal of Solution Chemistry p. 775 - 784 (1981)
Update date:2022-08-11
Topics:
 Hoeiland, H.
Hoeiland, H.
 Hald, L.H.
Hald, L.H.
 Kvammen, O.J.
Kvammen, O.J.
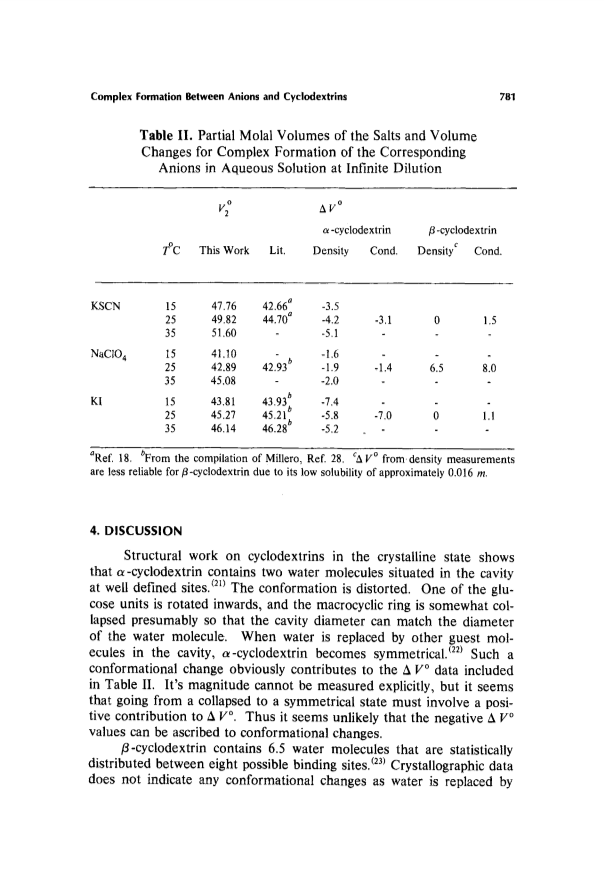
Partial molal volume changes during complex formation between SCN(1-), I(1-), and ClO4(1-) and α- and β-cyclodextrin have been determined by two independent methods of measurements; one based on density measurement and subsequent calculation of apparent molal volumes, the other on differentiating the association constants with respect to pressure.Results from the two methods are in good agreement.Negative volume changes were observed for complex formation between the anions and α-cyclodextrin while zero or slightly positive values were observed for complex formation with β-cyclodextrin.The result is consistent with the idea that the anions do not become dehydrated as they form complexes with cyclodextrins.
View More








DANYANG ZHONGTAI TRADE CO .,LTD.
Contact:0086-511-86985511
Address:No.25,Dongfang Rd., Danyang City.Jiangsu P.R.China
zhuzhou zhongle chemical co. ltd.
Contact:+86-0731 28228409
Address:Zhuzhou, Hunan, China
Taizhou Elitechemie MediPharma Technology Co.,Ltd.
Contact:+86-523-86810021
Address:Building G14,NO.1 Avenue,China Medical City, Taizhou, Jiangsu,China
Chengdu Pukang Biotechnology Co., Ltd
Contact:+86-28-82550498
Address:No. 558 Rulin Road,Xinjin county,Chengdu city, China
Beijing Hope Pharmaceutical Co., Ltd.
Contact:86-10-67886402
Address:309,Block 1 District B,No.12 Hongda North Road , Beijing Economic-Technological Development Area, Beijing, China
Doi:10.1016/j.tetasy.2009.07.048
(2009)Doi:10.1016/j.molcata.2015.05.009
(2015)Doi:10.1021/jo01362a028
(1957)Doi:10.1039/j29710001625
(1971)Doi:10.1021/ja01245a513
(1943)Doi:10.1021/jm00333a045
(1964)