The Journal of Organic Chemistry
Article
1
4-Chloroaniline (Table 6, entry 3): (254 mg, 99%); H NMR
(300 MHz, CDCl3) δ 7.11 (d, J = 9.0 Hz, 2H), 6.62 (d, J = 9.0 Hz,
2H), 3.58 (s, 2H); 13C NMR (75 MHz, CDCl3) δ 144.9, 129.1, 123.1,
116.2; MS-EI: m/z 129 (30%), 127 (100%), 100 (12%), 92 (16%), 65
(24%).
ACKNOWLEDGMENTS
■
This work was supported by a grant from the Christian
Doppler Research Society (CDG). D.C. thanks the Ministerio
́
de Ciencia e Innovacion of Spain for a scholarship.
1
4-Methoxyaniline (Table 6, entry 4): (243 mg, 98%); H NMR
(300 MHz, CDCl3) δ 6.77 (d, J = 8.9 Hz, 2H), 6.67 (d, J = 9.0 Hz,
2H), 3.76 (s, 3H), 3.41 (s, 2H). 13C NMR (75 MHz, CDCl3) δ 152.7,
139.9, 116.4, 114.8, 55.7; MS-EI: m/z 123 (72%), 108 (100%), 80
(43%).
REFERENCES
■
(1) Nanoparticles and Catalysis; Astruc, D., Ed.; Wiley-VCH:
Weinheim, Germany, 2008.
Benzylamine (Table 6, entry 5): (212 mg, 99%); 1H NMR (300
MHz, CDCl3) δ 7.38−7.25 (m, 5H), 3.87 (s, 2H). 13C NMR (75
MHz, CDCl3) δ 143.5, 128.5, 127.1, 126.8, 46.5; MS-EI: m/z 107
(50%), 106 (100%), 79 (35%), 77 (25%).
(2) For general reviews see: (a) Polshettiwar, V.; Varma, R. S. Green
Chem. 2010, 12, 743−754. (b) Astruc, D.; Lu, F.; Ruiz Aranzaes, J.
Angew. Chem., Int. Ed. 2005, 44, 7852−7872. (c) Kalidindi, S. B.;
Jagirdar, B. R. ChemSusChem 2012, 5, 65−75. (d) Zahmakiran, M.;
Ozkar, S. Nanoscale 2011, 3, 3462−3481. (e) Wang, D. S.; Xie, T.; Li,
Y. D. Nano Res. 2009, 2, 30−46. (f) Schlogl, R.; Abd Hamid, S. B.
Angew. Chem., Int. Ed. 2004, 43, 1628−1637.
Preparation and Isolation of Fe3O4 Nanoparticles. A solution
of Fe(acac)3 0.02 mmol (7.1 mg) and hydrazine hydrate (2 mmol) in
methanol (1.5 mL) was placed into a 10 mL microwave vial and
heated at 150 °C for 1 min. The resulting mixture was cooled at room
temperature and after 20−30 min the black precipitate was retrieved
with magnetic separation or centrifugation (5000 rpm, 5 min). The
solid was washed three times with fresh methanol and dried overnight
in a drying oven at 70 °C. The obtained Fe3O4 nanocrystals were
characterized by means of X-ray powder diffraction (XRD) and high-
resolution transmission electron microscopy (HRTEM).16
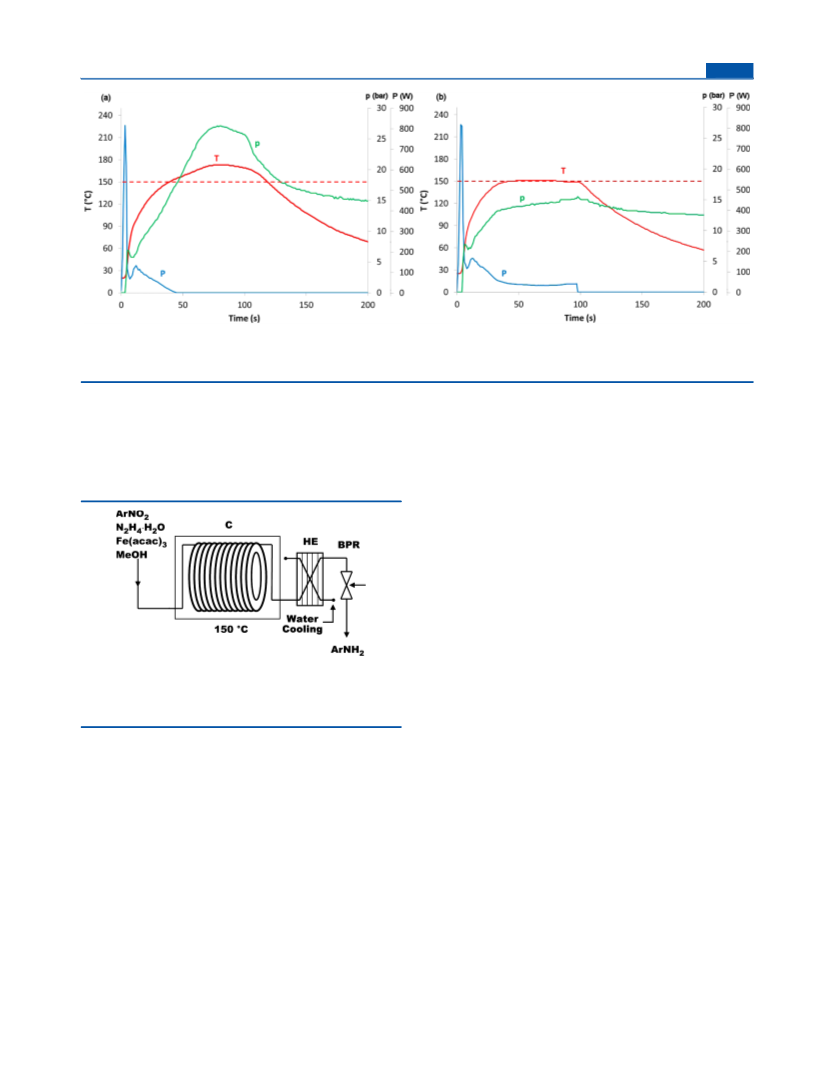
Representative Procedure for the Reduction of Nitroarenes
Using Continuous Flow Conditions. Flow experiments were
performed using a Uniqsis FlowSyn43 reactor equipped with a 20
mL stainless steel coil and a 34 bar backpressure regulator. After the
desired reaction parameters were achieved, a mixture containing
methanol (2 mL), the substrate (2 mmol), hydrazine hydrate (3.6
mmol), and Fe(acac)3 (0.25 mol %) was pumped through the system.
Due to the generation of N2 gas during the reaction, careful control of
the residence time is necessary because the extra pressure generated
will transport the reaction mixture at a different flow rate than the
nominal flow rate stated by the HPLC pumps. The residence time was
estimated by observing the total residence time in the reactor (visual
inspection, the reaction mixture is dark after the processing) and
taking into account the volume of the “cold” zones of the reactor
(pumps, tubing, etc) and the flow rate. The crude reaction mixture was
collected at the reaction outlet and purified as described above for the
microwave chemistry experiments.
(3) Plietker, B. Iron Catalysis in Organic Chemistry; Wiley-VCH:
Wienheim, Germany, 2008.
(4) For some recent examples of iron oxide nanocatalysts see:
(a) Luque, R.; Baruwati, B.; Varma, R. S. Green Chem. 2010, 12, 1540−
1543. (b) Zeng, T.; Chen, W.-W.; Cirtiu, C. M.; Moores, A.; Song, G.;
Li, C.-J. Green Chem. 2010, 12, 570−573. (c) Koukabi, N.; Kolvari, E.;
Khazaei, A.; Zolfigol, M. A.; Shirmardi-Shaghasemi, B.; Khavasi, H. R.
Chem. Commun. 2011, 47, 9230−9232. (d) Yang, C.; Wu, J.; Hou, Y.
Chem. Commun. 2011, 47, 5130−5141.
(5) For recent reviews on magnetic nanocatalysts see: (a) Shylesh, S.;
Schunemann, V.; Thiel, W. R. Angew. Chem., Int. Ed. 2010, 49, 3428−
̈
3459. (b) Polshettiwar, V.; Luque, R.; Fihri, A.; Zhu, H.; Bouhrara, M.;
Basset, J.-M. Chem. Rev. 2011, 111, 3036−3075. (c) Nasir, R. B.;
Varma, R. S. Chem. Commun. 2013, 49, 752−770.
(6) (a) Downing, R. S.; Kunkeler, P. J.; van Bekkum, H. Catal. Today
1997, 37, 121−136. (b) Ono, N. The Nitro Group in Organic Synthesis;
Wiley-VCH: New York, 2001.
(7) (a) Nishimura, S. Handbook of Heterogeneous Catalytic Hydro-
genation for Organic Synthesis; Wiley-Interscience: New York, 2001.
(b) Arnold, H.; Dobert, F.; Gaube, J. In Handbook of Heterogeneous
̈
Catalysis; Wiley-Interscience: New York, 2008; pp 3266−3284.
(8) Tafesh, A. M.; Weiguny, J. Chem. Rev. 1996, 96, 2035−2052.
(9) (a) Corma, A.; Serna, P. Science 2006, 313, 332−334. (b) He, L.;
Wang, L.-C.; Sun, H.; Ni, J.; Cao, Y.; He, H.-Y.; Fan, K.-N. Angew.
Chem., Int. Ed. 2009, 48, 9538−9541. (c) Li, M.; Hu, L.; Cao, X.;
Hong, H.; Lu, J.; Gu, H. Chem.Eur. J. 2011, 17, 2763−2768.
(d) Wu, H.; Zhuo, L.; He, Q.; Liao, X.; Shi, B. Appl. Catal., A 2009,
366, 44−56. (e) Amali, A. J.; Rana, R. K. Green Chem. 2009, 11, 1781−
1786. (f) Li, J.; Shi, X.; Bi, Y.; Wei, J.; Chen, Z. ACS Catal. 2011, 1,
657−664.
4-Chloro-2′-aminobiphenyl (8): (7.9 g, 97% yield, in a 40 mmol
scale reaction) for characterization data, see above.
4-Chloro-1,2-diaminobenzene (10): (5.6 g, 98%, in a 40 mmol
scale reaction); 1H NMR (300 MHz, DMSO-d6) δ 6.50 (d, J = 2.4 Hz,
1H), 6.45 (d, J = 8.2 Hz, 1H), 6.34 (dd, J = 2.4, 8.2 Hz, 1H), 4.63 (sa,
4H). 13C NMR (75 MHz, DMSO-d6) δ 137.1, 134.3, 120.8, 116.5,
115.4, 113.7. MS-EI: m/z 144 (30%), 142 (100%), 141 (22%), 114
(22%), 80 (25%)
3-Fluoro-4-morpholinylaniline (12): (3.7 g, 95%, a 20 mmol
scale reaction); 1H NMR (300 MHz, DMSO-d6) δ 6.76 (t, J = 8.6 Hz,
1H), 6.37−6.29 (m, 2H), 4.99 (s, 2H), 3.68 (t, J = 4.6 Hz, 4H), 2.80
(t, J = 4.6 Hz, 4H). 13C NMR (75 MHz, DMSO-d6) δ 158.4, 155.2,
146.0, 145.9, 129.6, 129.4, 121.0, 120.9, 110.0, 102.5, 102.2, 66.9, 52.1,
52.0. MS-EI: m/z 196 (60%), 138 (100%), 136.9 (50%).
(10) (a) Junge, K.; Wendt, B.; Shaikh, N.; Beller, M. Chem. Commun.
́
2010, 46, 1769−1771. (b) Pehlivan, L.; Metay, E.; Laval, S.; Dayoub,
W.; Demonchaux, P.; Mignani, G.; Lemaire, M. Tetrahedron Lett.
2010, 51, 1939−1941.
(11) Bae, J. W.; Cho, Y. J.; Lee, S. H.; Yoon, C. O. M.; Yoon, C. M.
Chem. Commun. 2000, 1857−1858.
(12) Lin, X.; Wu, M.; Wu, D.; Kuga, S.; Endoe, T.; Huang, Y. Green
Chem. 2011, 13, 283−287.
(13) Berthold, H.; Schotten, T.; Honig, H. Synthesis 2002, 1607−
̈
1610.
ASSOCIATED CONTENT
■
(14) (a) Wienhofer, G.; Sorribes, I.; Boddien, A.; Westerhaus, F.;
̈
S
* Supporting Information
Junge, K.; Junge, H.; Llusar, R.; Beller, M. J. Am. Chem. Soc. 2011, 133,
1
Copies of H NMR spectra of all prepared compounds. This
12875−12879. (b) Sorribes, I.; Wienhofer, G.; Vicent, C.; Junge, K.;
̈
Llusar, R.; Beller, M. Angew. Chem., Int. Ed. 2012, 51, 7794−7798.
(15) (a) Vass, A.; Dudas, J.; Toth, J.; Varma, R. S. Tetrahedron Lett.
2001, 42, 5347−5349. (b) Kumarraja, M.; Pitchumani, K. Appl. Catal.
A: Gen. 2004, 265, 135−139. (c) Shi, Q.; Lu, R.; Jin, K.; Zhang, Z.;
Zhao, D. Green Chem. 2006, 8, 868−870. (d) Kim, S.; Kim, E.; Kim, B.
M. Chem. Asian J. 2011, 6, 1921−1925. (e) Sharma, U.; Verma, P. K.;
Kumar, N.; Kumar, V.; Bala, M.; Singh, B. Chem.Eur. J. 2011, 17,
material is available free of charge via the Internet at http://
AUTHOR INFORMATION
■
Corresponding Author
5903−5907. (f) Jagadeesh, R. V.; Wienhofer, G.; Westerhaus, F. A.;
̈
Notes
Surkus, A.-E.; Pohl, M.-M.; Junge, H.; Junge, K.; Beller, M. Chem.
Commun. 2011, 47, 10972−10974. (g) Shi, Q.; Lu, R.; Lu, L.; Fu, X.;
The authors declare no competing financial interest.
4541
dx.doi.org/10.1021/jo400556g | J. Org. Chem. 2013, 78, 4530−4542

 Cantillo, David
Cantillo, David
 Moghaddam, Mojtaba Mirhosseini
Moghaddam, Mojtaba Mirhosseini
 Kappe, C. Oliver
Kappe, C. Oliver