794
Helvetica Chimica Acta – Vol. 89 (2006)
Method C: Elimination with Chloral/TsOH. A mixture of (ꢀ)-8a,b (0.11 g, 0.26 mmol), chloral (0.07
ml, 0.75 mmol), and TsOH·H2O (20 mg, 0.13 mmol) in benzene (10 ml) was stirred at 358 for 3 h. The
mixture was filtered through Alox B (act. I) and evaporated. The residue was purified by FC (silica
gel, hexane/CH2Cl2 1:1): 90 mg (88%) of (ꢀ)-9. Colorless oil. Data of (ꢀ)-9: see above.
(ꢀ)-Wiedendiol B (=4-Methoxy-3-{[(1E,2RS,4aRS,8aRS)-octahydro-2,5,5,8a-tetramethylnaphtha-
len-1(2H)-ylidene]methyl}benzene-1,2-diol; (ꢀ)-1) and 4-Methoxy-3-{[(1RS,2SR)-1,2,3,4,5,6,7,8-octahy-
dro-1,2,5,5-tetramethylnaphthalen-1-yl]methyl}benzene-1,2-diol ((ꢀ)-10). Compound (ꢀ)-9 (30 mg,
0.078 mmol) was treated with EtOH/HCl 7:1 (20 ml ) at 508 for 21 h. The mixture was diluted with
H2O and Et2ACHTREUONG , the org. layer washed with H2O, dried (Na2SO4), and evaporated. Further purification
was carried out by MPLC (hexane/CH2Cl2 95 :5 !50 :50, 60 min): 17 mg (65%) of (ꢀ)-1 and 4 mg
(15%) of (ꢀ)-10. Light yellow oils.
Data of (ꢀ)-1: Rf (CH2Cl2): 0.38. 1H-and 13C-NMR: Table 3. MS: 344 (27, M+), 206 (31), 191 (100),
153 (50), 152 (39), 95 (23), 81 (23), 69 (43), 59 (42), 57 (21), 55 (54), 43 (50), 41(86). HR-MS: 344.2351
Oþ3 ; calc. 344.2351).
(C22ACHTERUNGH32ACHTREGUN
Data of (ꢀ)-10: Rf (CH2Cl2): 0.25. 1H-and 13C-NMR: Table 3. MS: 344 (8, M+), 192 (50), 191 (100),
190 (37), 153 (23), 135 (32), 121 (25), 109 (21), 95 (25), 69 (24). HR-MS: 344.2351 (C
344.2351).
Oþ3 ; calc.
22ACHTRUEGNH32ACHTREUNG
2. Protected (ꢀ)-Siphonodictyal B. Sesamol Methyl Ether (=5-Methoxy-1,3-benzodioxole; 15). Sesa-
mol (5.47 g, 39.60 mmol) was treated with 1.5M Et4ANOH (39.6 ml, 59.40 mmol) in MeOH. After evapo-
CTHREUNG
ration, the residue was dissolved in THF (150 ml), the soln. cooled to 08, DMS (38.42 ml, 396.05 mmol)
added, and then the mixture stirred at r.t. for 1 h. The mixture was hydrolyzed with conc. NH3 soln./H2O/
ice 1:1:1, and the diluted with Et2ACHTREOUNG and H2O and the org. layer washed with H2O, dried (Na2SO4), and
evaporated: 6.16 g (100%) of 15. Yellowish oil. Rf (hexane/AcOEt 4 :1) 0.77. 1H-NMR (300 MHz): 6.64
(d, J=8.5, HꢁC(6)); 6.42 (d, J=2.5, HꢁC(3)); 6.25 (dd, J=8.5, 2.5, HꢁC(5)); 5.84 (s, OCH2O); 3.68 (s,
MeO). MS: 152 (98, M+), 137 (100), 107 (41), 79 (42), 53 (22), 51 (20). HR-MS: 152.0473 (C Oþ3 ; calc.
8ACHTRENUHG 8ACHTREUGN
152.0473).
5-Bromosesamol Methyl Ether (=5-Bromo-6-methoxy-1,3-benzodioxol; 16). A soln. of 15 (1.08 g,
7.10 mmol) in dry THF (100 ml) was treated with Br2 (0.36 ml, 7.10 mmol) at 08. After stirring for 5
min at 08, Et2ACHTREUONG and sat. Na2CO3 soln. were added. The org. layer was separated, washed with H2O
and brine, filtered through Na2SO4 and evaporated: 1.55 g (95%) of 16. White solid. Rf (toluene): 0.72.
1H-NMR (300 MHz): 6.93 (s, HꢁC(6)); 6.50 (s, HꢁC(3)); 5.88 (s, OCH2O); 3.76 (s, MeO). MS: 232
(100, M+), 230 (93), 217 (77), 215 (77), 187 (27). HR-MS: 229.9579 (C BrOþ3 ; calc. 229.9579).
8ACHTRENUHG 7ACHTREUGN
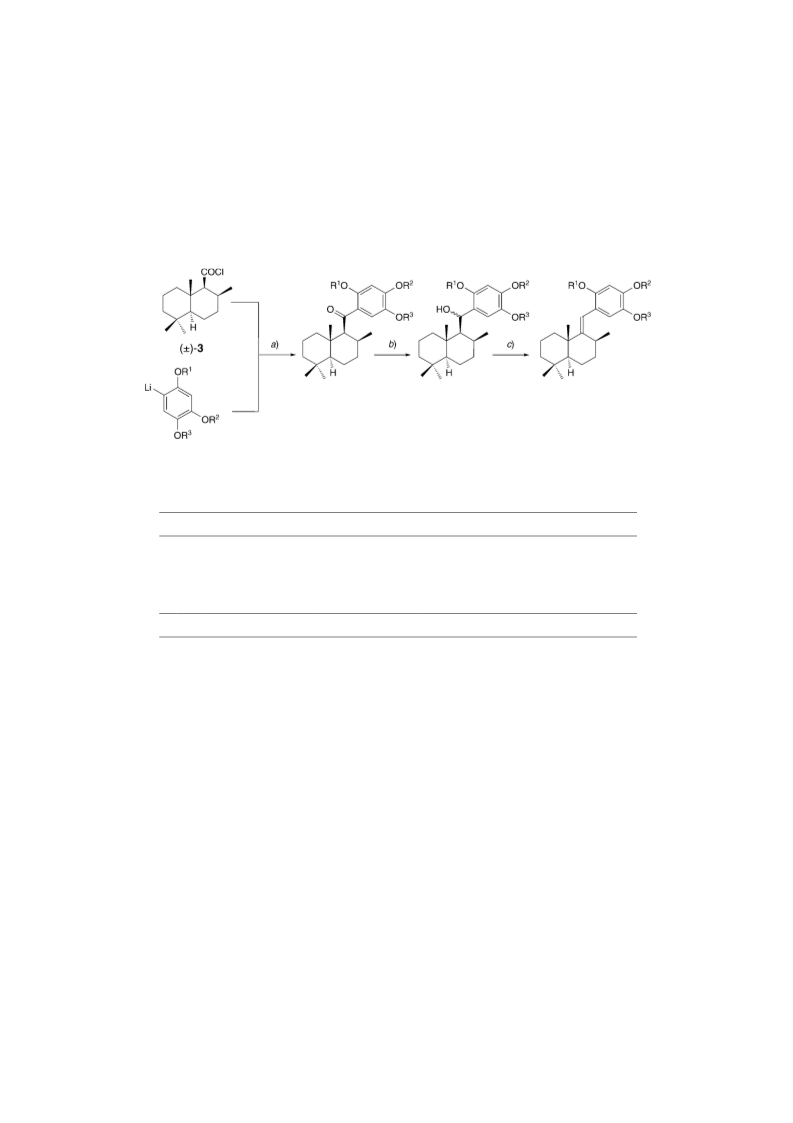
[(1RS,2RS,4aRS,8aRS)-Decahydro-2,5,5,8a-tetramethylnaphthalen-1-yl](6-methoxy-1,3-benzo-
dioxol-5-yl)methanone ((ꢀ)-17). A soln. of 16 (1.00 g, 4.36 mmol) in dry THF (50 ml) was cooled to
ꢁ1108 and treated with 1.6M BuLi in hexane/cyclohexane (2.73 ml, 4.36 mmol). After 5 min, a freshly
prepared soln. of (ꢀ)-3 (2.18 mmol) in dry THF (20 ml) was added dropwise. The mixture was allowed
to warm up to r.t. for ca. 1.5 h, and sat. NH4Cl soln. (2.0 ml) was added. For purification, the mixture was
adsorbed on silica gel and submitted to FC (hexane/toluene 1:1 for the eluation of 15 and 16, then hex-
ane/AcOEt 7:1): 0.79 g (97%) of (ꢀ)-17. Yellow oil. Rf (hexane/Me
CO 3 :1): 0.67. 1H-and 13C-NMR:
2ACHTREUNG
Table 4. MS: 372 (13, M+), 221 (34), 194 (16), 180 (10), 179 (100). HR-MS: 372.2300 (C Oþ4 ; calc.
23ACHTERNUGH32ACHTREGUN
372.2301).
(aRS)-a-[1RS,2RS,4aRS,8aRS)- and (aRS)-a-[(1SR,2SR,4aSR,8aSR)-Decahydro-2,5,5,8a-tetrame-
thylnaphthalen-1-yl]-6-methoxy-1,3-benzodioxol-5-methanol ((ꢀ)-18a,b). To a soln. of (ꢀ)-17 (0.31 g,
0.83 mmol) in dry THF (100 ml), 1.0M LiBHEt3 in THF (4.16 ml, 4.16 mmol) was added at 08. After stir-
ring at r.t. for 1 h, sat. NH4Cl soln. was added, the mixture stirred for additional 5 min and diluted with
Et
was further purified by FC (hexane/Me
Me2ACO 3 :1) 0.54. Since the product consists of two diastereoisomers, no further spectroscopic character-
ization was carried out.
O/H2O. The org. layer was washed with H2O, filtered through Na2SO4, and evaporated. The residue
CTHREUNG
CTHREUNG
6-Methoxy-5-{[(2RS,4aRS,8aRS)-octahydro-2,5,5,8a-tetramethylnaphthalen-1(2H)-ylidene]methyl}-
1,3-benzodioxole ((ꢀ)-19). A soln. of (ꢀ)-18a,b (0.31 g, 0.83 mmol) in benzene (20 ml) was treated with
TsOH·H2O (0.16 g, 0.83 mmol) at r.t. for 1 h. The mixture was diluted with sat. Na2CO3 soln., Et2CATHREOUNG , and
H2O. The org. layer was washed twice with sat. Na2CO3 soln. and H2O, dried (Na2SO4), and evaporated:

 Bernet, Andreas
Bernet, Andreas
 Seifert, Karlheinz
Seifert, Karlheinz